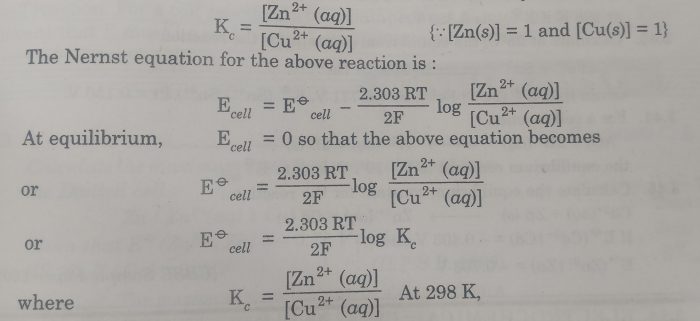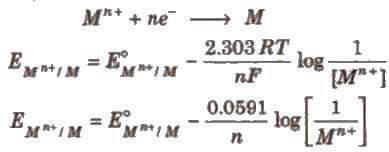all formulas of electrochemistry
How are electrochemical reactions in water analyzed?
Electrochemical reactions in water are better analyzed by using the ion-electron method, where H +, OH − ion, H 2 O and electrons (to compensate the oxidation changes) are added to the cell's half-reactions for oxidation and reduction. In acidic medium, H + ions and water are added to balance each half-reaction .
What is electrochemistry in chemistry?
Electrochemistry is the study of chemical processes which lead to electrons moving. This movement of electrons is called electricity, which can be generated in a reaction known as an oxidation-reduction reaction by electrons from one element to another. Electrochemistry Formulas: Get access to Electrochemistry Formulas for NEET 2023.
Who invented electrochemistry?
English chemist John Daniell ( left) and physicist Michael Faraday ( right ), both credited as founders of electrochemistry. Electrochemistry is the branch of physical chemistry concerned with the relationship between electrical potential difference and identifiable chemical change.
What are the two types of electrochemical cells?
There are two types of electrochemical cells: galvanic, also called Voltaic, and electrolytic. Galvanic cells derives its energy from spontaneous redox reactions, while electrolytic cells involve non-spontaneous reactions and thus require an external electron source like a DC battery or an AC power source.
Nernst Equation
Dependence of electrode potential and EMF on concentration and temperature. For electrode potential; For solids, liquids or gases at 1 atom. byjus.com
Gibbs Free Energy Change
Gibbs free energy change in an electrochemical reaction can be expressed as the equivalent of the potential difference. byjus.com
Concentration Cell
Concentration cells are electrochemical cells in which the emf arises from a transfer of material from one electrode to another due to a concentration difference between the two. In general for concentration cells. byjus.com
Electrolysis
The process of decomposition of an electrolyte by the passage of electricity through its aqueous solution or molten state. Electrodes used in the electrolysis of different electrolytes are of two types, inert electrodes and active electrodes. byjus.com
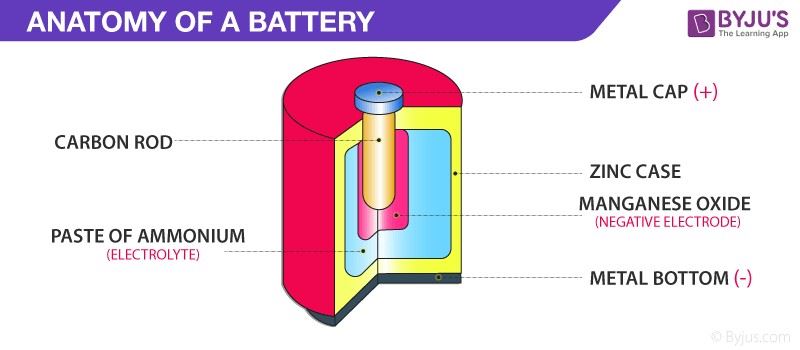
Faraday First Law of Electrolysis
The mass of primary products formed at an electrode by electrolysis is directly proportional to the quantity of electricity passed. byjus.com
Faraday Second Law of Electrolysis
The masses of different primary products formed by equal amounts of electricity are proportional to the ratio of molar mass to the number of electrons involved with a particular reaction. byjus.com
Conductance
In relation to electrolytes, the term conductance (C) is used more frequently than resistance. Conductance implies the case with which electric current can flow through a conductor. It is defined as the reciprocal of resistance. byjus.com
Kohlrausch Law
The molar conductivity of an electrolyte at infinite dilution can be expressed as the sum of the ionic conductivities of cations and anions each multiplied by the number of ions present in one formula unit of the electrolyte. byjus.com
Applications of Kohlrausch Law
1. In the calculation of molar conductivity at infinite dilution for a weak electrolyte. 2. In the calculation of the degree of dissociation. 3. In the calculation of solubility of a sparingly soluble salt. As the solution is saturated but infinitely dilute. Molarity = solubility Hence, Where ‘u’ are ionic mobilities at infinite dilution. Electroch

All Formulaes of electrochemistry Electrochemistry all Formulae how to solve electrochemistry Ques

Electrochemistry Review

Nernst Equation Explained Electrochemistry Example Problems pH Chemistry Galvanic Cell
|
Formulae For ELECTROCHEMISTRY
CHAPTER 3 - ELECTROCHEMISTRY u. Important Terms Definitions & Formulae. By Mir Mohammed Abbas II PCMB 'A'. Formulae For ELECTROCHEMISTRY. FORMULAS- |
|
Electrochemistry Formula Sheet
Electrochemistry. Formula Sheet. Page 2. Electrode Potential. Gibbs energy All the CSIR NET Test Series based on the latest pattern and the trend that ... |
|
Chapter 18: Electrochemistry
+ in the final equation add OH– to both sides o combine H. + and OH– to produce H2O all other half-cells. Page 9. Reference half-cell : standard hydrogen ... |
|
ELECTROCHEMISTRY ENGINEERING CHEMISTRY B.Tech 1 year
02-Apr-2020 All the plates are separated from adjacent plates by insulators like wood strips glass fiber etc. The entire combination is immersed in ... |
|
SINGULAR PERTURBATION THEORY APPLIED TO THE
Key words and phrases: Electrochemistry equations semiconductor device equations |
|
A Practical Beginners Guide to Cyclic Voltammetry
03-Nov-2017 The Nernst equation relates the potential of an electrochemical ... recommend having ferrocene present in all measurements rather than adding it ... |
|
The Electrochemistry Module Users Guide
44 |
| CHAPTER 2: MODELING WITH ELECTROCHEMISTRY equation of the electrolyte will make sure the total current over all electrode surfaces sums up to zero |
|
Electrochemical Corrosion Rate Measurement – A Comparison
EOC is the starting point for virtually all electrochemical corrosion The Butler-Volmer Equation is a general electrochemical equation that describes the ... |
|
Electrochemistr ochemistr ochemistry Electrochemistr ochemistr
Electrochemistry is the study of production of 3.13 Write the chemistry of recharging the lead storage battery highlighting all the materials that are ... |
|
Unacademy
Nature of solute : All ionic compounds. (strong electrolytes) have a ~ 1 at equations for the reactions taking place at cathode and anode. What happens if. |
|
Formulae For ELECTROCHEMISTRY
Unit of ?m in above formula is Scm2mol-1. 13. ? c m. 0 m. ?. = ?. 14. 2 a c?. K = 1-?. XII Chemistry. CHAPTER 3 - ELECTROCHEMISTRY. |
|
Electrochemistry Formula Sheet
Gradeup CSIR-NET. Super Subscription. Features: 1. Memory Based Test Series of the actual exam paper. 2. All the CSIR NET Test Series based on the latest |
|
1.3.10 Electrochemistry Name Symbol Definition SI unit Notes
(3) v+ and v- are the numbers of cations and anions per formula unit of an difference for zero current through the cell all local charage transfer ... |
|
ELECTROCHEMISTRY ENGINEERING CHEMISTRY B.Tech 1 year
02-Apr-2020 Application of electrochemical series. 6. Electromotive force (EMF). 7. Nernst equation. 8. Battery i). Primary batteries. |
|
Electrochemistry Electrochemistry
Equation. We have assumed in the previous section that the concentration of all the species involved in the electrode reaction is unity. This need not be. |
|
Understanding Voltammetry : Equilibrium Electrochemistry and the
Equilibrium Electrochemistry and the Nernst Equation 3 In all of the examples considered above a dynamic equilibrium is rapidly. |
|
Notes - 06 Electrochemistry - CIE Chemistry A Level
6 : Electrochemistry In an ion the sum of the oxidation states of all the atoms is ?equal to the ... Therefore the balanced equation can be written as:. |
|
CBSE NCERT Solutions for Class 12 Chemistry Chapter 3
Practice more on Electrochemistry Applying Nernst equation we have: ... Write the chemistry of recharging the lead storage battery highlighting all the. |
|
The Electrochemistry Module Users Guide
Domain Equations for the Electroanalysis Interface . . . . . . . . . 127 For a list of all the core physics interfaces included with a COMSOL. |
|
Test4 ch19 Electrochemistry Practice-answers-Marked
Spontaneous Voltaic Electrochemical Cells p4 Nonstandard Concentrations and Cell Key Equations Given for Test: ... Determine all oxidation numbers. |
|
Formulae For ELECTROCHEMISTRY - WordPresscom
Unit of Λm in above formula is Scm2mol-1 13 α c m 0 m ∧ = ∧ 14 2 a cα K = 1-α XII Chemistry CHAPTER 3 - ELECTROCHEMISTRY u Important Terms |
|
7 ELECTROCHEMISTRY - Mahesh Tutorials Science
Electrochemical Cells are of two types: It may be defined as the conducting power of all the ions formula unit of the electrolyte on complete dissociation 5 |
|
Electrochemistry all formulas pdf - f-static
Electrochemistry all formulas pdf Continue All important notes chemistry neet, JEE There are a total of 21 chapters in chemistry that are important to NEE and |
|
Electrochemistry - NCERT
When the concentrations of all the species involved in a half-cell is unity then the electrode potential is known as standard electrode potential According to IUPAC |
|
Some Formulas for E-Chem and Kinetics
All rates written as Aconc Atime or A[A] At Instantaneous rate is the slope of a concentration vs time plot and is shown by the differential equation: d[A] dt |
|
CHEMISTRY CLASS-XII - edudel
Analysis shows that a metal oxide has the empirical formula M 0 98 O 1 00 (b ) Atoms B occupy all the octahedral voids and half of the tetrahedral voids Electrochemistry may be defined as the branch of chemistry which deals with the |
|
Electrochemistry Formulas - WongChemistry
If all three conditions are not met, then it is nonstandard conditions Formulas Definitions E°cell = E°red + E°ox E°cell = cell potential at standard conditions, |
|
1310 Electrochemistry Name Symbol Definition SI unit - iupac
(3) v+ and v- are the numbers of cations and anions per formula unit of an difference for zero current through the cell, all local charage transfer equilibria and |
|
Electrochemistry
between them By 1812, the Swedish chemist Berzelius could propose that all Electrochemistry is the study of reactions in which charged particles (ions or electrons) to the next on the right is written in between the formulas (Note that the |