all mechanisms for a level chemistry edexcel
|
A Level Chemistry
The aims and objectives of the Pearson Edexcel Level 3 Advanced GCE in Chemistry are to enable students to develop: • essential knowledge and understanding of different areas of the subject and how they relate to each other • a deep appreciation of the skills knowledge and understanding of scientific methods |
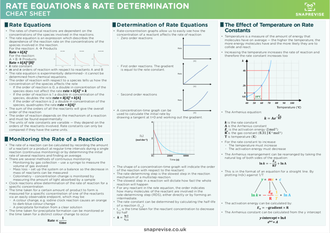
How can a rate equation be deduced from a reaction mechanism?
The order of a reactant and thus the rate equation can be deduced from a reaction mechanism if the rate-determining step is known For example, the reaction of nitrogen oxide (NO) with hydrogen (H 2) to form nitrogen (N 2) and water N2O2 (g) + H2 (g) → H2O (l) + N2O (g) slow (rate-determining step)
What is Edexcel chemistry?
Chemistry A level Edexcel a reaction in which one molecule combines with another to form a larger molecule with no other products. Involves breaking a pi bond The removal of a molecule from a saturated molecule to make an unsaturated molecule. A pi-bond is formed
Why are reaction mechanisms important?
Reaction mechanisms can help us understand fundamentals like order of reaction and rate-determining steps in a chemical process. Not all steps in a reaction mechanism occur at the same speed. The slowest step is known as the “rate-determining step” because it determines the overall rate of the reaction.
What is a one step reaction mechanism?
A one step reaction mechanism is simply the overall stoichiometric equation One of the two elementary steps for this two step mechanism can be taken from the question: The second elementary step must involve the reaction of the nitrogen dioxide formed with sulfur dioxide: Elementary step 2: NO 2 + SO 2 → NO + SO 3 (or 2NO 2 + 2SO 2 → 2NO + 2SO 3)

Quick Revision

How to remember organic chemistry mechanisms

Mechanisms Explained Year 12 or AS Chemistry Organic Chemistry A level Chemistry
|
REACTION MECHANISMS
In order to do this for organic chemistry it is useful to classify types of compound and how they behave. All reaction mechanisms involve looking at bond ... |
|
A-level-chemistry-2015-specification.pdf
All titles correct as at May 2014. “ ” Page 6. Page 7. Introduction. The Pearson Edexcel Level 3 Advanced GCE in Chemistry is designed for use in schools and |
|
Mechanism Summary for A-level AQA Chemistry :
The amine formed in the first reaction has a lone pair of electrons on the nitrogen and will react further with the haloalkane. |
|
Pearson Edexcel International Advanced Level in Chemistry (YCH11)
All Edexcel's academic qualifications including our new International AS and A level suite |
|
Reaction pathways - OCR A Level Chemistry A
OCR 2017. Version 1. Haloalkane. Alcohol. 1°. 2°. Nitrile. Amine. Alkene. Aldehyde. Ketone. Carboxylic acid. Acyl chloride. 1o Amide. 2o Amide. |
|
Pearson Edexcel Level 3 Advanced GCE in Chemistry (9CH0)
All information in this document is correct at the time of publication. (ii) Explain by considering the mechanism of the reaction |
|
Untitled
Edexcel A-level Chemistry. 6 Inorganic chemistry and the periodic table Edexcel A-level Chemistry. Secondary halogenoalkanes react by both mechanisms but SN1 ... |
|
Chemistry
Jun 19 2017 Show all your working in calculations and include units where appropriate. Pearson Edexcel. Level 3 GCE. Turn over. Page 2. 2. *P48059A0224*. DO ... |
|
Detailed Notes - Edexcel Chemistry A-level
This mechanism allows aromatic aminesand nitrobenzeneto be produced from benzene. Mechanism - general electrophilic substitution mechanism All amino acids ... |
|
Detailed Notes - Edexcel Chemistry A-level
This means that the two possible products of the reaction are mirror imagesand are therefore optical isomers. This mechanism |
|
REACTION MECHANISMS
The table below reviews the organic reactions studied for AS chemistry how they are All reaction mechanisms involve looking at bond breaking. |
|
6.-organic-chemistry-i-edexcel.pdf
Displayed formula: show all the covalent bonds present in a molecule The mechanism for this reaction is called a free radical substitution. |
|
Mechanism Summary for A-level AQA Chemistry :
Mechanism Summary for A-level AQA Chemistry Acid catalysed elimination mechanism: alcohols ? alkenes. The H+ comes from the conc H2SO4 or conc H3PO4. |
|
Specification - AS
Pearson Edexcel Level 3 Advanced Subsidiary GCE in Chemistry (8CH0) All our qualifications are designed to meet our World Class Qualification ... |
|
Pearson Edexcel International Advanced Level in Chemistry (YCH11)
The International Advanced Level can be claimed on completion of all the units Students start to use the conventions for mechanisms in organic chemistry ... |
|
Pearson Edexcel Level 3 Advanced GCE in Chemistry (9CH0)
A Level. Chemistry. Sample Assessment Materials. Pearson Edexcel Level 3 Advanced GCE in All the marks on the mark scheme are designed to be awarded. |
|
OCR A Level Chemistry A - Topic Exploration Pack - Reaction
OCR 2017. Version 1. Haloalkane. Alcohol. 1°. 2°. Nitrile. Amine. Alkene. Aldehyde. Ketone. Carboxylic acid. Acyl chloride. 1o Amide. 2o Amide. |
|
Chemistry
19 Jun 2017 Show all your working in calculations and include units where appropriate. Pearson Edexcel. Level 3 GCE. Turn over ... |
|
Mark Scheme (Results)
16 Aug 2017 GCE Chemistry June 2017 ... Edexcel and BTEC qualifications come from Pearson ... All marks on the mark scheme should be. |
|
Detailed Notes - Edexcel Chemistry A-level
This mechanism allows ?aromatic amines?and ?nitrobenzene?to be produced from benzene. Mechanism - general electrophilic substitution mechanism. The electrophile |
|
REACTION MECHANISMS - Physics & Maths Tutor
The table below reviews the organic reactions studied for AS chemistry, how they are classified and All reaction mechanisms involve looking at bond breaking |
|
Mechanism Summary for A-level AQA Chemistry : - chemrevise
The amine formed in the first reaction has a lone pair of electrons on the nitrogen and will react further with the haloalkane CH 3 C H H Br CH3CH2NH2 |
|
Organic Chemistry I - chemrevise
Displayed formula: show all the covalent bonds present in a molecule When drawing 6: Organic Chemistry I EDEXCEL does not split structural isomers into The mechanism for this reaction is called a free radical above the level of the |
|
MAPPING GUIDE - Pearson qualifications
Pearson Edexcel International Advanced Level in Chemistry (YCH11) First teaching September all Calculations are likely to be less structured AS Topic 1 : Formulae, Equations and Extension of radical mechanism to show formation of |
|
PEARSON EDEXCEL INTERNATIONAL A LEVEL Student Book 2
Copies of official specifications for all Edexcel qualifications may be found on the topic we will study the rates of chemical reactions and the mechanisms by |
|
SN1 versus SN2 (Edexcel) - The Exam Formula
Both are simple nucleophilic substitutions but the mechanisms differ slightly depending on the starting haloalkane This is also not just to do with organic chemistry |
|
George Facers A level Chemistry - Hodder Education
Edexcel A level Chemistry Year 1 Student Book 9781471807466 Mar 2015 £ 24 99 eTextbook elements that all work together to give you the ultimate classroom and There are two distinct mechanisms by which halogenoalkanes react |
|
Edexcel A level Chemistry Student Book 2 - Pearson Schools and
Edexcel A level Chemistry TOPIC 11 Answers to Student Book 2 questions Note: Two decimal places is correct for the final answer, since all of the data Mechanism 2 has one molecule of CH3CHO and one ion of CN− in the first step |
|
OCR A Level Chemistry A - Topic Exploration Pack - Reaction
OCR 2017 Version 1 Haloalkane Alcohol 1° 2° Nitrile Amine Alkene Aldehyde Ketone Carboxylic acid Acyl chloride 1o Amide 2o Amide Ester |











![June 2012 Edexcel Chemistry Unit 5 - [PDF Document] June 2012 Edexcel Chemistry Unit 5 - [PDF Document]](https://zbook.org/img/2012/unit-28-industrial-applications-of-organic-chemistry.jpg)





![Edexcel A2 Chemistry 43 - Notes - [PDF Document] Edexcel A2 Chemistry 43 - Notes - [PDF Document]](https://d1w7fb2mkkr3kw.cloudfront.net/assets/images/book/lrg/9781/8469/9781846905988.jpg)


![07 Organic Mechanisms EDEXCEL - [PDF Document] 07 Organic Mechanisms EDEXCEL - [PDF Document]](https://d1e4pidl3fu268.cloudfront.net/9b8d7b5a-cd50-4562-8c96-38661824dc93/Screenshot20200301at133827.crop_693x519_0)
