chemical kinetics lecture notes ppt
|
Chemical Kinetics
Chemical Kinetics Lecture notes edited by John Reif from PPT lectures by: • Chung (Peter) Chieh University of Waterloo • Hana El-Samad UCSB • John D |
What is chemical kinetics brief notes?
Chemical kinetics is an important topic in Physical Chemistry and basically deals with helping students understand the different aspects of a chemical reaction.
More specifically, the term 'kinetics' deals with the rate of change of some quantity.
For example, the rate of change of displacement is given as velocity.Which topics are important in chemical kinetics?
Related topics on Chemical kinetics
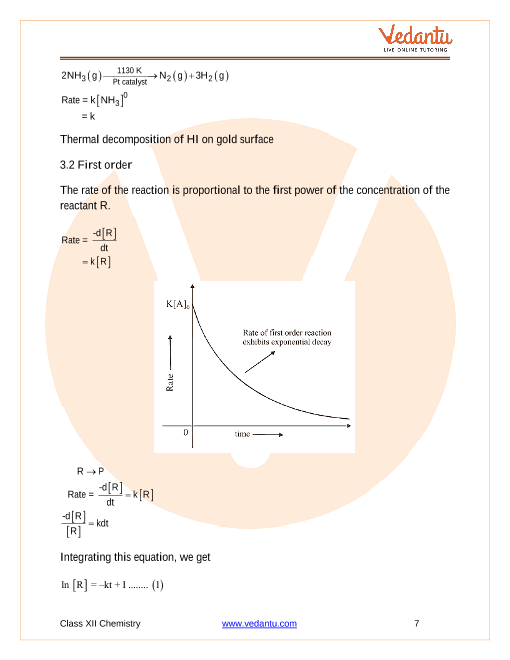
Rate of reaction.Rate law and rate constant.Collision theory of chemical reaction.Order and molecularity of reaction.Pseudo First order reaction.Zero order reaction.First order reaction.Second order reaction.The sequence of events that describes the actual process by which reactants become products is called the reaction mechanism.
Reaction Mechanisms.
Reactions may occur all at once or through several discrete steps.
|
Chemical Kinetics
Chemical Kinetics. Lecture notes edited by John Reif from PPT lectures by: • Chung (Peter) Chieh University of Waterloo. • Hana El-Samad |
|
Chemical Kinetics What are Chemical Kinetics?
Chemical Kinetics. Lecture notes edited by John Reif from PPT lectures by: Chung (Peter) Chieh University of Waterloo. Hana El-Samad |
|
LECTURE NOTES ON THERMODYNAMICS
7 days ago ... kinetics a = 0.1 s−1. the following |
|
Chemistry 1B General Chemistry
Sep 27 2020 The powerpoint lecture slides will be posted before the lecture on ... Course Description: This class will cover the principals of chemical ... |
|
Lecture 3: Enzyme kinetics
Jan 19 2009 The convention used for this slides is to use UPPERCASE for the ... Thought natural chemical reactions were fast? (e.g. interconversion ... |
|
Biological and Soft Matter Physics
Chemical Kinetics and Pattern Formation. Electrokinetic Phenomena. A Survey of Lectures will be given primarily with powerpoint slides. These will be. |
|
Lecture Atmospheric Chemistry
Most chemical processes in the atmosphere are started by energy input from the sun; the consequences are determined by kinetics. • [BrO] = 15 ppt [HO2] = 1 ... |
|
ECE 5320 Lecture #6 and 7
• Name files as lecture notes-your last name. • Do not make files of the notes that are already ppt only the The sol-gel process is a wet-chemical technique ... |
|
Laboratory Procedure Manual: Biochemistry Profile
The indicator reaction uses the oxaloacetate for a kinetic determination of NADH consumption. The. International Federation of Clinical Chemistry (IFCC) has now |
|
Chemical Kinetics What are Chemical Kinetics?
Chemical Kinetics. Lecture notes edited by John Reif from PPT lectures by: Chung (Peter) Chieh University of Waterloo. Hana El-Samad |
|
Chemical Kinetics
Chemical Kinetics. Lecture notes edited by John Reif from PPT lectures by: • Chung (Peter) Chieh University of Waterloo. • Hana El-Samad |
|
Lecture 2 - Chemical Kinetics.pptx
material is not to be sold reproduced or distributed without the prior wri en permission of the owner |
|
PowerPoint ????
?? ????? ?????? ???? ?? Lecture 1: Introduction to the course and basic kinetics. Yongdan Li. Mon 21th of Jan 10:15-12:00. Ke 5 D 311. Lecture 2: Ideal reactor ... |
|
LECTURE NOTES ON THERMODYNAMICS
?? ??? ???? ?? These are lecture notes for AME 20231 Thermodynamics |
|
Lecture 9 Elementary chemical kinetics
%20Bonding%20and%20Reactivity/Lecture%209%20Introductory%20kinetics.pdf |
|
Lecture 3: Enzyme kinetics
?? ???? ???? ?? parameters from the plot. • It is useful for analysis of multi- substrate and inhibited enzymatic reactions (see next slides). • ... |
|
Chemical Engineering Formulas
Chemical Kinetics Rate Laws – Chemistry Review – Order of Reaction You can also find Chemical engineering formulae GATE Notes |
|
CHAPTER 13. CHEMICAL KINETICS
Chapter 13 Kinetics Student notes page 1 of 8. CHAPTER 13. CHEMICAL KINETICS. Kinetics - Study of factors that affect how fast a reaction occurs and the |
|
Grade 12 Chemistry: A Foundation for Implementation
?? ??? ???? ?? Process Notes for Balancing Neutralization Reactions 20 ... Appendix 3.2A: Chemical Kinetics: Assignment 1 6. |
|
Chemical Kinetics
Chemical Kinetics Lecture notes edited by John Reif from PPT lectures by: Chung (Peter) Chieh, University of Waterloo Hana El-Samad, UCSB John D |
|
Physical Chemistry 3: — Chemical Kinetics — - Uni Kiel
26 jui 2019 · This scriptum contains notes for the lecture “Physical Chemistry 3: Origin,8 or QtiPlot) may be needed for data evaluation and presentation 9 |
|
Chapter 14 Chemical Kinetics
Chemical Kinetics Learning goals and key skills: ➢ Understand the factors that affect the rate of chemical reactions ➢ Determine the rate of reaction given time |
|
Lecture 2 - Chemical Kineticspptx
For example, the detailed kinetic mechanism of the primary reference fuel (PRF) contains 1034 species participating in 4236 elementary reactions1 15 16 Page 9 |
|
Chemical Kinetics
Chemical Kinetics Lecture notes edited by John Reif from PPT lectures by: • Chung (Peter) Chieh, University of Waterloo • Hana El-Samad, UCSB • John D |
|
Chapter 14 Kinetics - Central Lyon CSD
Lecture Presentation © 2012 Pearson Education, Inc Page 2 Chemical Kinetics Kinetics • In kinetics we study the rate at which a chemical process occurs |
|
Chemical Kinetics - NCERT
Kinetic studies not only help us to determine the speed or rate of a chemical reaction but also describe the conditions by which the reaction rates can be altered |
|
Kinetics: Rates and Mechanisms of Chemical Reactions Chapter 13
14 5 Theories of Chemical Kinetics 14 6 Reaction Mechanisms: The Steps from Reactant to The physical state of the reactants influences reaction rate |
|
CHEMICAL KINETICS
CHEMICAL KINETICS Objectives • Order and molecularity • Kinetics of first and second order reaction • Pseudo unimolecular reaction • Arrhenius equation 3 |
|
Chemical Reactor Design - PowerPoint 演示文稿
25 fév 2019 · The field that studies the rates and mechanisms of chemical reactions and the design of Lecture 1: Introduction to the course and basic kinetics Yongdan Li Mon 21th of Jan 10:15-12:00 Lecture slides – Exercises and |










![Herunterladen] Chemical Kinetics Stochastic Processes and Herunterladen] Chemical Kinetics Stochastic Processes and](https://images.springer.com/sgw/books/medium/9783540226840.jpg)















![Laidler Chemical Kinetics pdf [ylyx7wroxenm] Laidler Chemical Kinetics pdf [ylyx7wroxenm]](https://i2.wp.com/www.dronstudy.com/wp-content/uploads//2020/03/word-image-288.jpeg?fit\u003d472%2C404\u0026ssl\u003d1)






