chemical kinetics notes pdf
|
Chemical Kinetics
➢ As the concentration of reactants increases so does the likelihood that reactant molecules will collide PDF Created with deskPDF PDF Writer - Trial :: http |
|
General Chemistry II Chapter 13 Lecture Notes Chemical Kinetics
Chemical Kinetics is concerned with reaction rates Typical reaction rate Observing the allover course of a reaction and measuring its rate usually tells |
|
Chemical Kinetics
Mechanisms provide a very detailed picture of which bonds are broken and formed during the course of a reaction Elementary Steps Molecularity • Elementary |
|
CHEMICAL KINETICS
It catalyses the spontaneous reactions but does not catalyse non-spontaneous reactions www spiroacademy com Page 14 CHEMISTRY NOTES www |
|
CHEMICAL KINETICS/RATE OF CHEMICAL REACTION 1
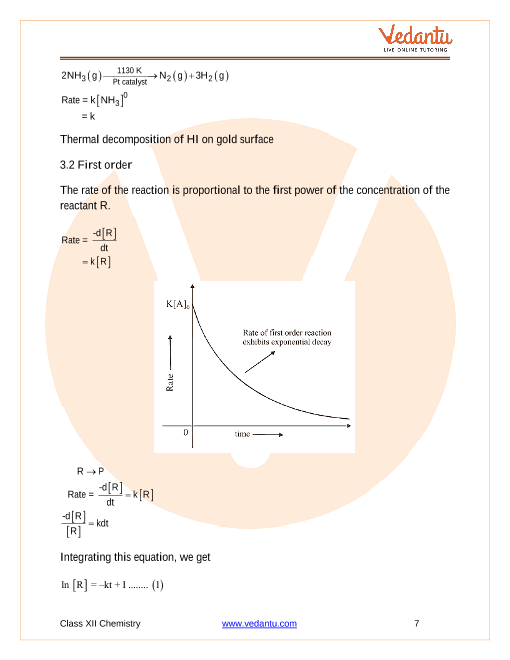
▷ This is a reaction whose rate is independent of concentration (i e initial concentration) ▷ A product ▷ Rate equation in differential form is R |
|
CHEMICAL KINETCS (Note-1) BSc 2nd Semester
Chemical kinetics is the branch of physical chemistry which deals with a study of the speed of chemical reactions Such studies also enable us to understand |
What is chemical kinematic?
chemical kinetics, the branch of physical chemistry that is concerned with understanding the rates of chemical reactions.
It is to be contrasted with thermodynamics, which deals with the direction in which a process occurs but in itself tells nothing about its rate.Unimolecular reactions are those involving a change in only one molecular of ionic structure.
Dissociation or isomerization of the molecule may be considered as characteristic examples of such reactions.
What is reaction rate Class 12 PDF?
(i) Rate of reaction is defined as the change in the concentration of any one of the reactants or products per unit time.
Rate constant of a reaction is equal to the rate of reaction when concentration of each reactant is taken as 1 mol L–1.
|
22.-Chemical-Kinetics-Notes.pdf
Chemical Kinetics y Study of rate of reactions and factors affecting it like temperature pressure |
|
CHEMICAL KINETICS
Thus in chemical kinetics we can also determine the rate of chemical reaction. From the kinetic stand point the reactions are classified into two groups: a) |
|
Chemical Kinetics What are Chemical Kinetics?
Summary of the Kinetics of Zero-Order First-Order and Second-Order Reactions. Order. Rate Law. Concentration-Time. Equation. Half-Life. 0. 1. 2 rate = k rate = |
|
Chemistry Notes for class 12 Chapter 4 Chemical Kinetics .pdf
the factors affecting the rate of reactions and the mechanism of the reaction. is called chemical kinetics. Chemical Reactions on the Basis of Rate of Reaction. |
|
UNIT 3 CHEMICAL KINETICS
38 |
|
CHB-201: CHEMICAL KINETCS (Note-1) BSc 2nd Semester- N(D
Chemical kinetics is the branch of physical chemistry which deals with a Produced with a Trial Version of PDF Annotator - www.PDFAnnotator.com. Page 2 ... |
|
Chemical Kinetics
➢ As the concentration of reactants increases so does the likelihood that reactant molecules will collide. PDF Created with deskPDF PDF Writer - Trial :: http |
|
M. Sc. I Semester PHYSICAL CHEMISTRY-I
This is the basic idea of chemical kinetics. Now let us discuss the factors Write short notes on the following: (i) Ionic atmosphere (ii) Electrophoretic ... |
|
PHYSICAL B. Sc. I YEAR PHYSICAL CHEMISTRY EMISTRY-I
H. A. Radi and J. O. Rasmussen Principles of Physics |
|
General Chemistry II Chapter 13 Lecture Notes Chemical Kinetics
Chemical Kinetics is concerned with reaction rates. Typical reaction rate units are M/s the change in concentration of a species per unit time. There is a huge |
|
Chemical Kinetics What are Chemical Kinetics?
Chemical Kinetics. Lecture notes edited by John Reif from PPT lectures by: Chung (Peter) Chieh University of Waterloo. Hana El-Samad |
|
CHEMICAL KINETICS
Chemical kinetics is the branch of physical chemistry which deals with a study of the speed of chemical reactions. Such studies also enable us to understand |
|
Chemical Kinetics
? As the concentration of reactants increases so does the likelihood that reactant molecules will collide. PDF Created with deskPDF PDF Writer - Trial :: http |
|
General Chemistry II Chapter 13 Lecture Notes Chemical Kinetics
Chemical Kinetics is concerned with reaction rates. Typical reaction rate units are M/s the change in concentration of a species per unit time. There is a huge |
|
CHB-201: CHEMICAL KINETCS (Note-1) BSc 2nd Semester- N(D
Chemical kinetics is the branch of physical chemistry which deals with a study of the speed of Produced with a Trial Version of PDF Annotator - www. |
|
Chemistry Notes for class 12 Chapter 4 Chemical Kinetics .pdf
the factors affecting the rate of reactions and the mechanism of the reaction. is called chemical kinetics. Chemical Reactions on the Basis of Rate of Reaction. |
|
Chemical Kinetics - Saurashtra University
She is co-author of three books. Page 2. 1. Preface. Chemical kinetics is a branch of physical chemistry. Various excellent. |
|
Chapter 14 Chemical Kinetics
Chemical kinetics: the area of chemistry dealing with the speeds or rates at which The concentrations of the reactants: Most chemical reactions proceed ... |
|
CHEMICAL KINETICS/RATE OF CHEMICAL REACTION 1
Branch of physical chemistry that deals with rate of reaction. ? Also known as reaction kinetics FACTORS AFFECTING RATE OF CHEMICAL REACTIONS. |
|
Notes - 08 Reaction Kinetics - CIE Chemistry A Level
Generally? increasing the concentration of reactants will increase the rate of reaction because there are more particles in the same volume so particles |
|
Chemical Kinetics
Chemical Kinetics Lecture notes edited by John Reif from PPT lectures by: Chung (Peter) Chieh, University of Waterloo Hana El-Samad, UCSB John D |
|
Reaction Rates: Chemical Kinetics
The reaction rate law expression relates the rate of a reaction to the concentrations of the reactants Each concentration is expressed with an order ( exponent) |
|
General Chemistry II Chapter 13 Lecture Notes Chemical Kinetics
Chapter 13 Lecture Notes Chemical Kinetics Chemical Kinetics is concerned with reaction rates Typical reaction rate units are M/s, the change in concentration |
|
Chapter 14 Chemical Kinetics
Chemical Kinetics Learning goals and key skills: ➢ Understand the factors that affect the rate of chemical reactions ➢ Determine the rate of reaction given time |
|
Physical Chemistry 3: — Chemical Kinetics — - Uni Kiel
26 jui 2019 · This scriptum contains lecture notes for the module “Physical Chemistry 3: Chemical Kinetics” (chem0405), the last part of the 3 semester |
|
Chemical Kinetics - NCERT
Kinetic studies not only help us to determine the speed or rate of a chemical reaction but also describe the conditions by which the reaction rates can be altered |
|
Chemical Kinetics
➢ The more homogeneous the mixture of reactants, the faster the molecules can react PDF Created with deskPDF PDF Writer - Trial :: http://www docudesk com |
|
CHEMICAL KINETICS Y - NIOS
Chemical Kinetics Notes Chemical Dynamics ○ explain the effect of temperature on reaction rate, and ○ explain Arrhenius equation and activation energy |
|
Chapter 14 Chemical Kinetics
Chemical kinetics: the area of chemistry dealing with the speeds or rates at The concentrations of the reactants: Most chemical reactions proceed faster if the |
|
CHEMICAL KINETICS
1 1 Chemical Kinetics Chemical kinetics is the branch of physical chemistry which deals with a study of the Thus, in chemical kinetics we can also determine the rate of chemical concentration 5 Write short notes on activation energy 6 |