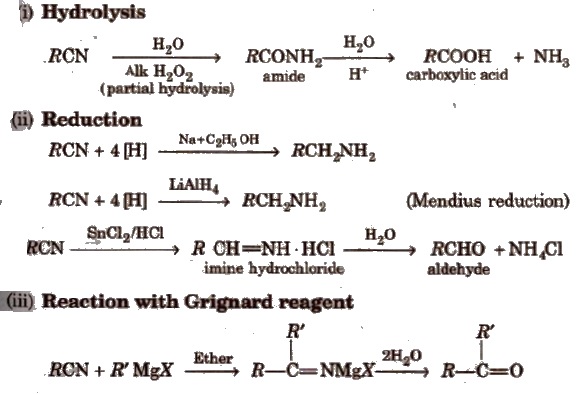
chemical properties of amines
|
Chapter 6 Amines and Amides
Chapter 6 Amines and Amides 33 Chemical Properties of Amines: Basicity • Amines are weak organic bases They react with water to produce alkylammonium ions |
What is the most important chemical property of amines?
The most important chemical property of amines is their basicity.
Amines, whether soluble or insoluble in water, react quantitatively with strong acids to form water-soluble salts.They are colourless and have an unpleasant odour.
They have high melting and boiling points compared to aliphatic amines.
They are completely soluble in organic solvents but insoluble in water due to the presence of an aromatic ring.
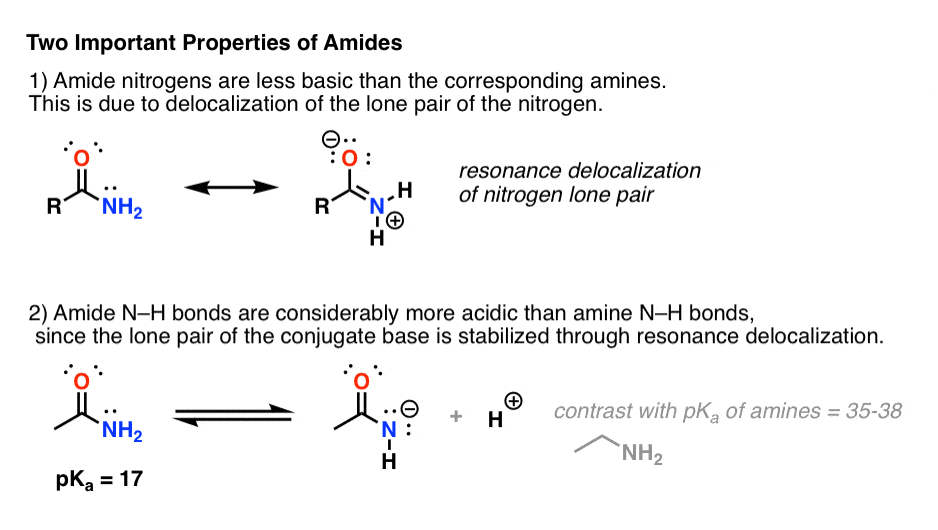
What are the chemical properties of amides?
Like the esters, solutions of amides in water usually are neutral—neither acidic nor basic.
The amides generally have high boiling points and melting points.
These characteristics and their solubility in water result from the polar nature of the amide group and hydrogen bonding (Figure 15.14.
What are the chemical reactions of amines?
Amines act as nucleophiles due to the presence of lone pair of electrons in the nitrogen atom.
They are involved in reactions such as acylation, electrophilic substitution, alkylation etc.
|
Chapter 6 Amines and Amides
Learn the important physical properties of the amines and amides. • Learn the major chemical reactions of amines and amides and learn how to predict the. |
|
Classification and Nomenclature of Amines
Learn the IUPAC system for naming amines and amides. •. Learn the important physical properties of the amines and amides. •. Learn the major chemical reactions |
|
THE EFFECT OF CHEMICAL STRUCTURE ON THE LOAD
The load-carrying properties of a range of dialkyl phosphates and their alkyl- amine salts were assessed using the four-ball machine. |
|
Amines
explain the properties of amines; ñ distinguish between primary secondary 9.6 Chemical. Reactions. Rationalised 2023-24. Page 9. 267 Amines. Amine salts on ... |
|
Chemical properties of phenanthrolinequinones and the mechanism
London 1956; p 881). Page 3. Phenanthrolinequinones and Amine Oxidation. J. Am. Chem. Soc. |
|
Amines Amines
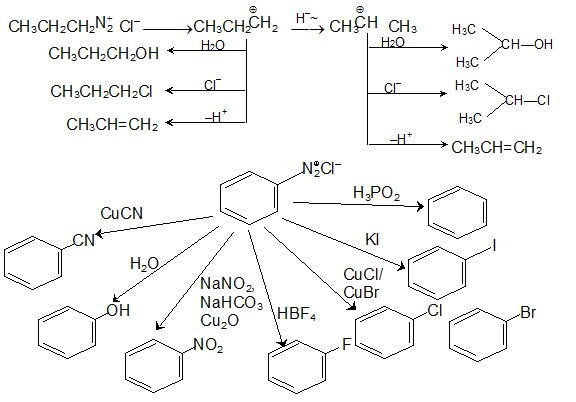
Properties. 13.9 Chemical. Reactions. 2. 2. 2. 2. 2015-16. Page 18. 398. Chemistry. 5. Replacement by hydroxyl group: If the temperature of the diazonium salt |
|
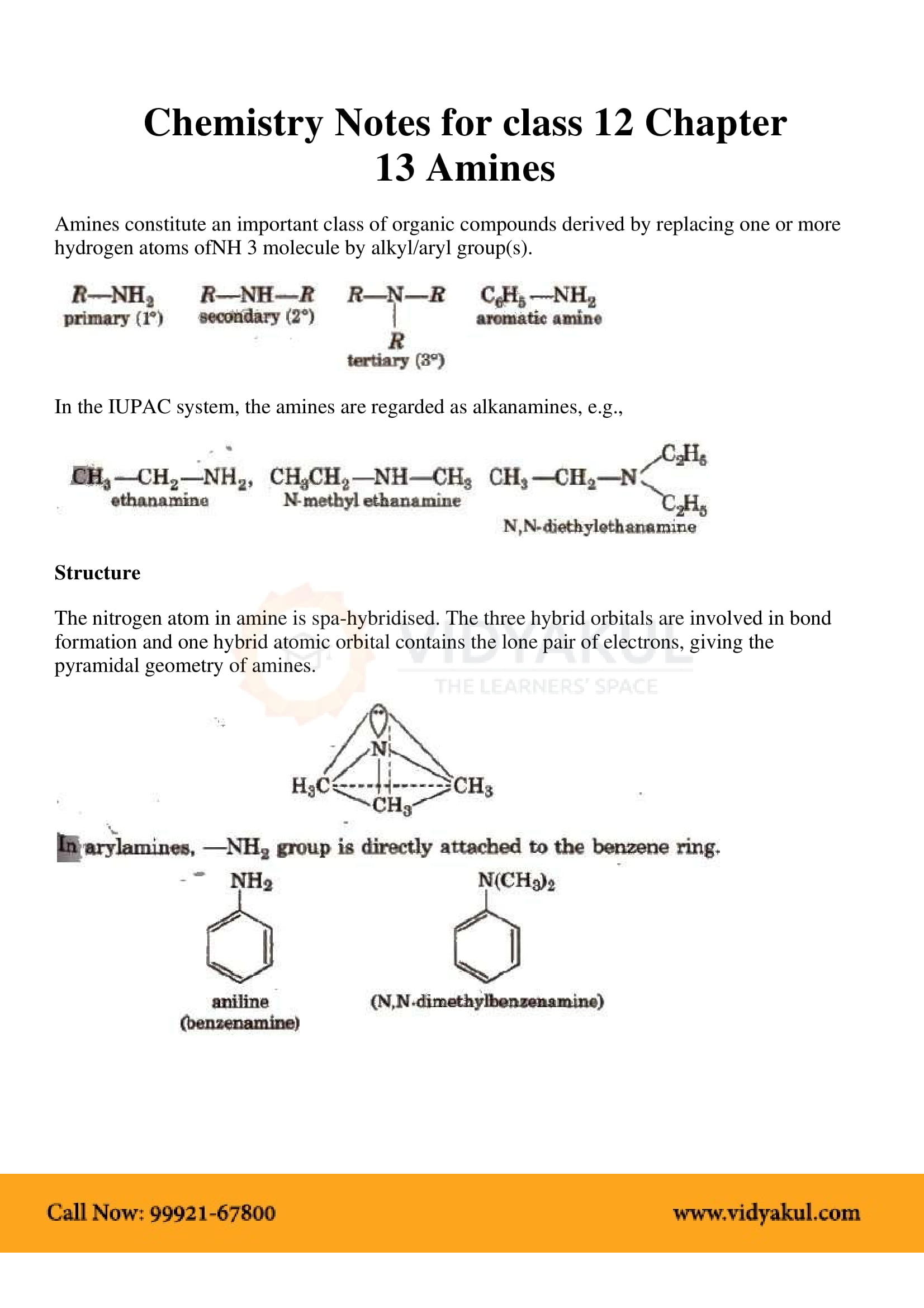
Chemistry Notes for class 12 Chapter 13 Amines
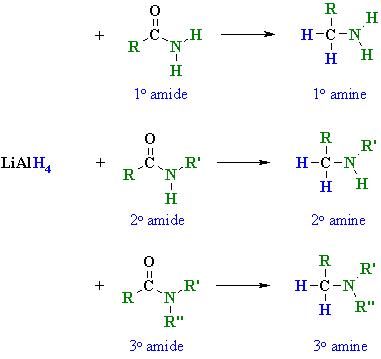
To obtain primary amine with same number of carbon atoms from primary amide reduction is done with LiAlH4/ether. Physical Properties of Amines. 1. The lower |
|
Olfactory properties of Amines and n-ButanoI
Olfactory properties of amines and n-boUuiol. ETHYLAMINE. OIETHYLAMINE. TRI chemical properties is needed. The aliphatic amines with their diversity of ... |
|
The Physical and Chemical Properties of the Methylhydroxylamines1
to variations found in the series ammonia and the methylamines. Although all the methyl derivatives of hydroxyl- amine have been mentioned in the early |
|
Carbon dioxide absorption into aqueous amine based solvents
and chemical properties of blended amines solutions (obtained by applying mixing rules detailed in a previous work. [2]) and to the kinetic term |
|
Chapter 6 Amines and Amides
Learn the important physical properties of the amines and amides. • Learn the major chemical reactions of amines and amides and learn how to predict the. |
|
Classification and Nomenclature of Amines
Learn the IUPAC system for naming amines and amides. •. Learn the important physical properties of the amines and amides. •. Learn the major chemical |
|
Olfactory properties of Amines and n-ButanoI
The chemical and olfactory purity of each odorant was determined using a gas chromatograph (150m x 0.75mm i.d.S.S. Carbowax 20M column; programmed for 30 min at |
|
Sch_206-amines.pdf
Physical Properties of Amines properties such as boiling point. ... dominates the chemistry of amines and is responsible for the. |
|
CHEM 212: Organic Chemistry II (3Credits) Chapter 1 Amines and
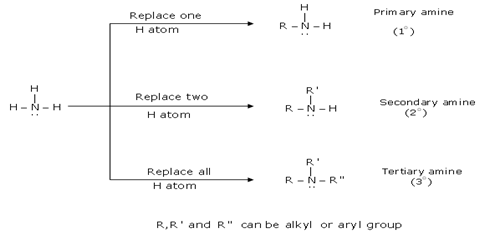
-Hydrogen Bond: Properties of amines depend on the degree of substitution of the nitrogen atom: tertiary amines do not have N-H bond hence it cannot form NH |
|
Chemical properties of phenanthrolinequinones and the mechanism
Chemical Properties of Phenanthrolinequinones and the. Mechanism of Amine Oxidation by o-Quinones of Medium. Redox Potentials. |
|
The Effect of the Physical and Chemical Characteristics of the
The chemical properties of fatty amines can be varied by changing the number of amine groups and their positions in the molecule (48). The balance between the |
|
Benzene and Its Derivatives
of the unique structure and chemical properties of benzene and its derivatives. properties of amines are their basicity and their nucleophilicity. |
|
Shaping amine-based solid CO2 adsorbents: effects of pelletization
chemical properties of an array of supported amine adsorbents based on mesoporous silica and ?-alumina supports. The virgin powders based on |
|
Organic Chemistry II / CHEM 252 Chapter 20 – Amines
Physical Properties and Structure of Amines • Primary and secondary amines can form hydrogen bonds to each other and water • Tertiary amines cannot form |
|
Amines
10 3 What Are the Characteristic Physical Properties of Amines? Amines are polar compounds, and both primary and secondary amines form intermolecu- lar |
|
Amine Chemistry Tutorial
In addition to basicity, amines are capable of functioning as nucleophiles and participating in displacement reactions with electrophilic compounds These |
|
Amines Amines - NCERT
Write chemical equations for the following reactions: (i) Reaction of ethanolic NH3 with C2H5Cl (ii) Ammonolysis of benzyl chloride and reaction of amine so |
|
Physical and chemical properties of amines pdf - Squarespace
Looking in detail at their simple physical properties, would be solubility and boiling points The easiest way to think of amine is as close to ammonia relatives, |
|
Chapter 17: Amines and Amides
and other physical disorders are amines and amides 17 1 Bonding Characteristics of Nitrogen Atoms in Organic Compounds Nitrogen atoms forms three |
|
AMINES
Physical Properties of Amines 13 Just like alcohols, amines are polar compounds However, because nitrogen is less electronegative than oxygen, the N-H |