chemistry solutions notes pdf
|
Chemistry Notes for class 12 Chapter 2 Solutions
www ncerthelp com (Visit for all ncert solutions in text and videos CBSE syllabus note and many more) Chemistry Notes for class 12 Chapter 2 Solutions |
|
Solution Chemistry Notes
Solution Chemistry Notes Page 2 Solutions • Basic definitions – Solution: homogenous mixture – Solute: substance that is dissolved – Solvent: substance |
|
Solutions Unacademy
Solution It is a homogeneous mixture of two or more chemically non-reacting substances whose composition can be changed within certain limits |
|
Solutions
Solvent determines the physical state of the solution Water is an universal solvent 2 Mole fraction (X) is a unitless quantity 3 Molality (m) and mole |
A solution is a homogeneous mixture.
The constituent particles of a solution are smaller than 10-9 metres in diameter.
Constituent particles of a solution cannot be seen by naked eyes.
Solutions do not scatter a beam of light passing through it.
What is a solution Class 12 chemistry?
A solution is a homogeneous mixture of two or more components in which the particle size is smaller than 1 nm.
Common examples of solutions are sugar in water and salt in water solutions, soda water, etc.
In a solution, all the components appear as a single phase.
What is a solution in chemistry notes?
A solution is a homogeneous mixture.
The major component is the solvent, while the minor component is the solute.
Solutions can have any phase; for example, an alloy is a solid solution.
Solutes are soluble or insoluble, meaning they dissolve or do not dissolve in a particular solvent.8 jui. 2022
|
Chemistry Notes for class 12 Chapter 2 Solutions
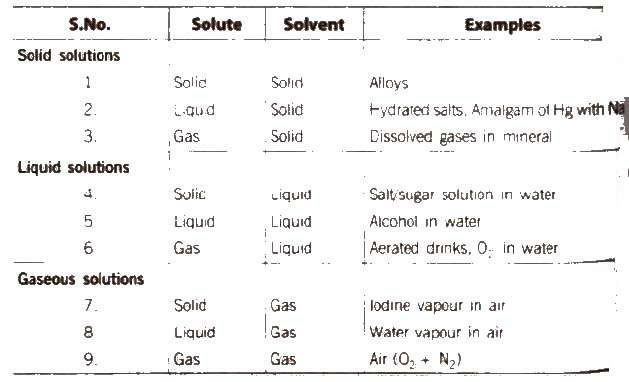
(A) Following types of solutions are seen on the basis of physical state of solute and solvent. [if water is used as a solvent the solution is called aqueous |
|
Notes Chapter 15: Solutions
Because so many things dissolve in water water is called the 'UNIVERSAL SOLVENT'. Page 2. Chemistry-2. Notes Chapter 14. Page 2 of 11. |
|
Solutions
Chemistry. Type of Solution. Solute. Solvent. Common Examples. Gaseous Solutions. Gas. Gas. Mixture of oxygen and nitrogen gases. |
|
LECTURE NOTES ON THERMODYNAMICS
Feb 14 2010 fugacities of gaseous solutions |
|
Analytical Chemistry 2.1 Solutions Manual
Solutions Manual for Analytical Chemistry 2.1. 6. Using the atomic weights from Appendix 18 we find that the formula weight for Ni(C4H14N4O4)2 is. |
|
Chapter 4: Calculations Used in Analytical Chemistry
Molar analytical concentration is the total number of moles of a solute regardless of its chemical state |
|
Introduction to Medical Laboratory Technology
circumvent these problems are developing of lecture notes on various subjects. Primary standard solution is a chemical solution that has the ... |
|
MATERIAL BALANCE NOTES Revision 3 Irven Rinard Department
Oct 2 1999 The material balance is the fundamental tool of chemical engineering ... complicate the solution of material balances models for the process ... |
|
Grade 12 Chemistry: A Foundation for Implementation
Jan 21 2011 Identifying Unknown Solutions (Teacher Notes and. Preparation Guide) 11. Appendix 1.6A: Process Notes for Writing Net Ionic Equations. |
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
When working with a dry chemical it is mixed as dry mass (g) per volume where #g/100 ml = percent concentration. A 10% NaCl solution is equal to 10 g dissolved |
|
Solution Chemistry Notes
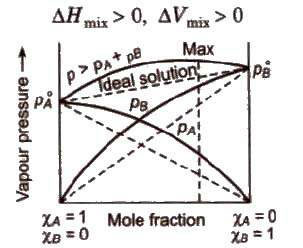
Solution Chemistry Notes Saturated solutions are at dynamic equilibrium – the solute is dissolving The solubility of a chemical determines how much can be |
|
Chemistry Notes for class 12 Chapter 2 Solutions - Ncert Help
(A) Following types of solutions are seen on the basis of physical state of solute and solvent [if water is used as a solvent, the solution is called aqueous solution |
|
Chapter 7 lecture notes: Solutions
10) Given a solution's initial concentration, be able to use the dilution equation to determine Chemistry 108 Lecture Notes Chapter 7: Solutions 3 Solutions |
|
Notes Chapter 9:
Most of the important chemistry that keeps plants, animals, and humans alive occur in aqueous solutions A solution is a homogeneous mixture – a mixture in |
|
Xii Chemistry Solution Notes - DITP
Solubility is defined as the [ ] Solutions Class 12 Notes Chemistry Chapter 2 - Learn CBSE NCERT Solutions for Class 12 Chemistry in PDF file format to free |
|
Solution Notes Class 12
(Part – I)class 12 maths notes chapter 1 - Tamil SolutionCBSE Notes Class 12 Chemistry Solutions AglaSem Schoolschemistry-notes-class-12- pdf - R K |
|
CHEMISTRY NOTES – CHAPTERS 17 AND 18 Water - Bonding
The chemical and physical properties of water 2 Aqueous solutions and their concentrations 3 Electrolytes and nonelectrolytes 4 Colligative properties of |
|
CHEMISTRY CLASS-XII - edudel
Chemical Kinetics 56-78 5 Surface Solutions 10 Unit III Electrochemistry 12 23 Unit IV Chemical Kinetics 10 Unit V Write short notes on the following : |
|
12th Class Chemistry Notes Cbse All Chapter imgnikoncentercl
1 mar 2021 · Chapter 2 Solutions Class 12 Notes Chemical Kinetics Class 12 Notes Students CBSE Chemistry Revision Notes for Class 12 PDF |