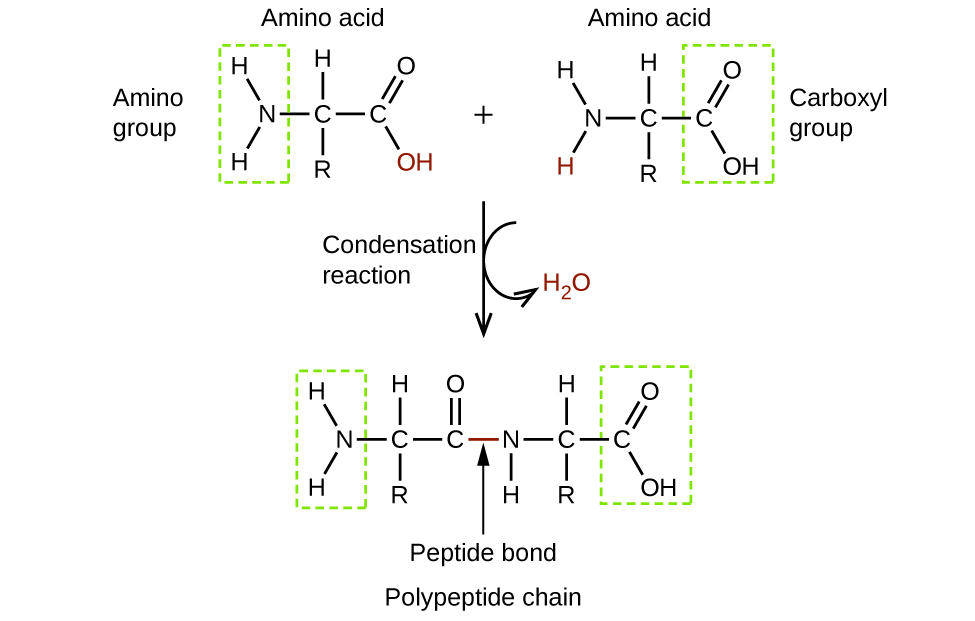
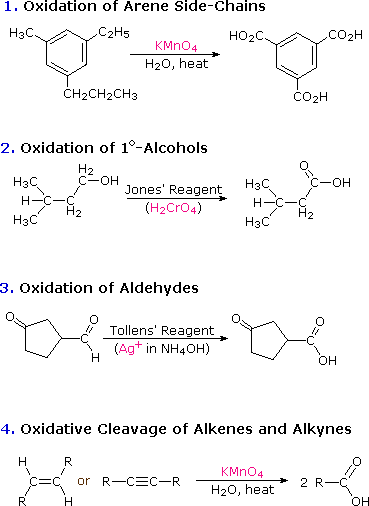
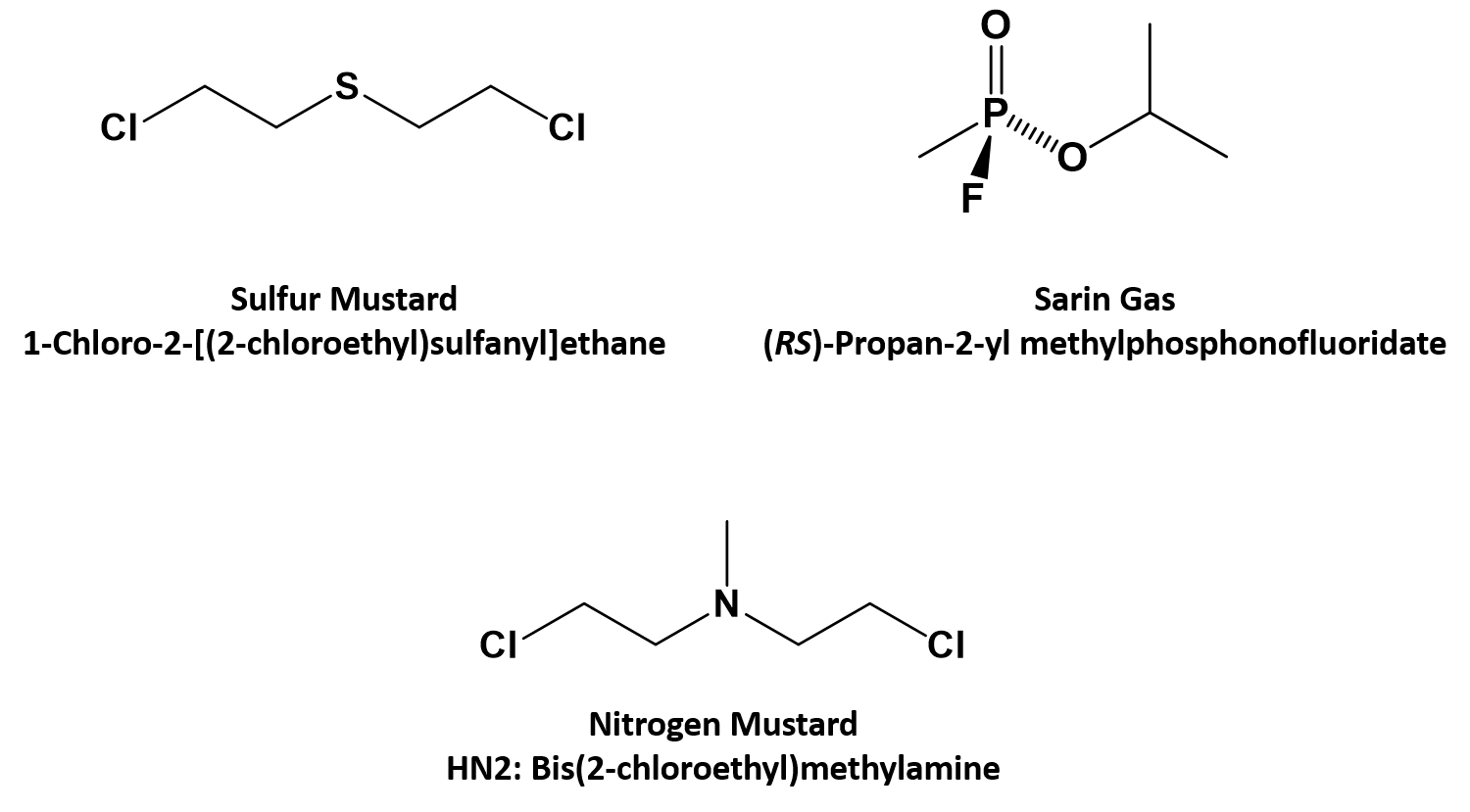
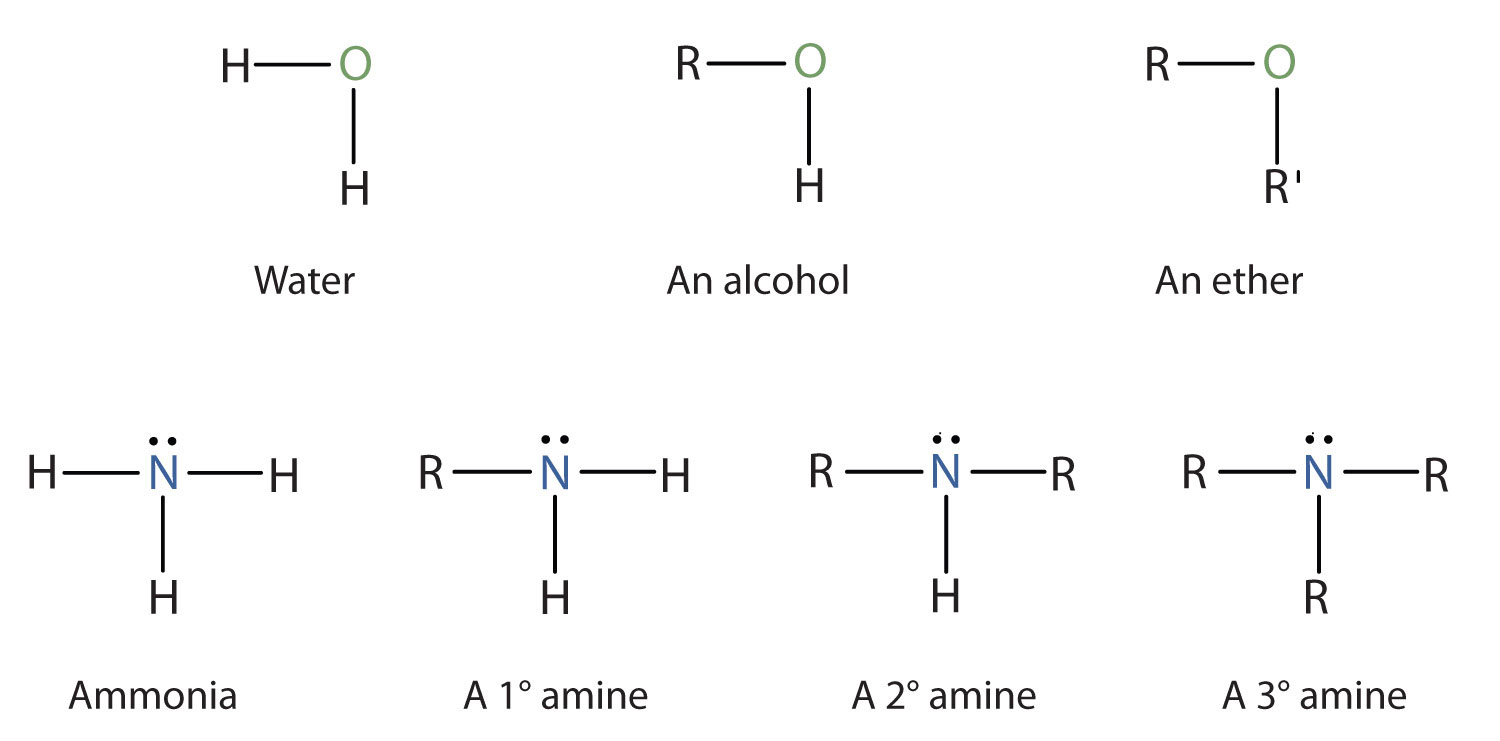
classify each formula or structure as a carboxylic acid an ester or an amide
|
Chapter 5 Carboxylic Acids and Esters
Learn to recognize the carboxylic acid ester and related functional groups • Learn the IUPAC system for naming carboxylic acids and esters |
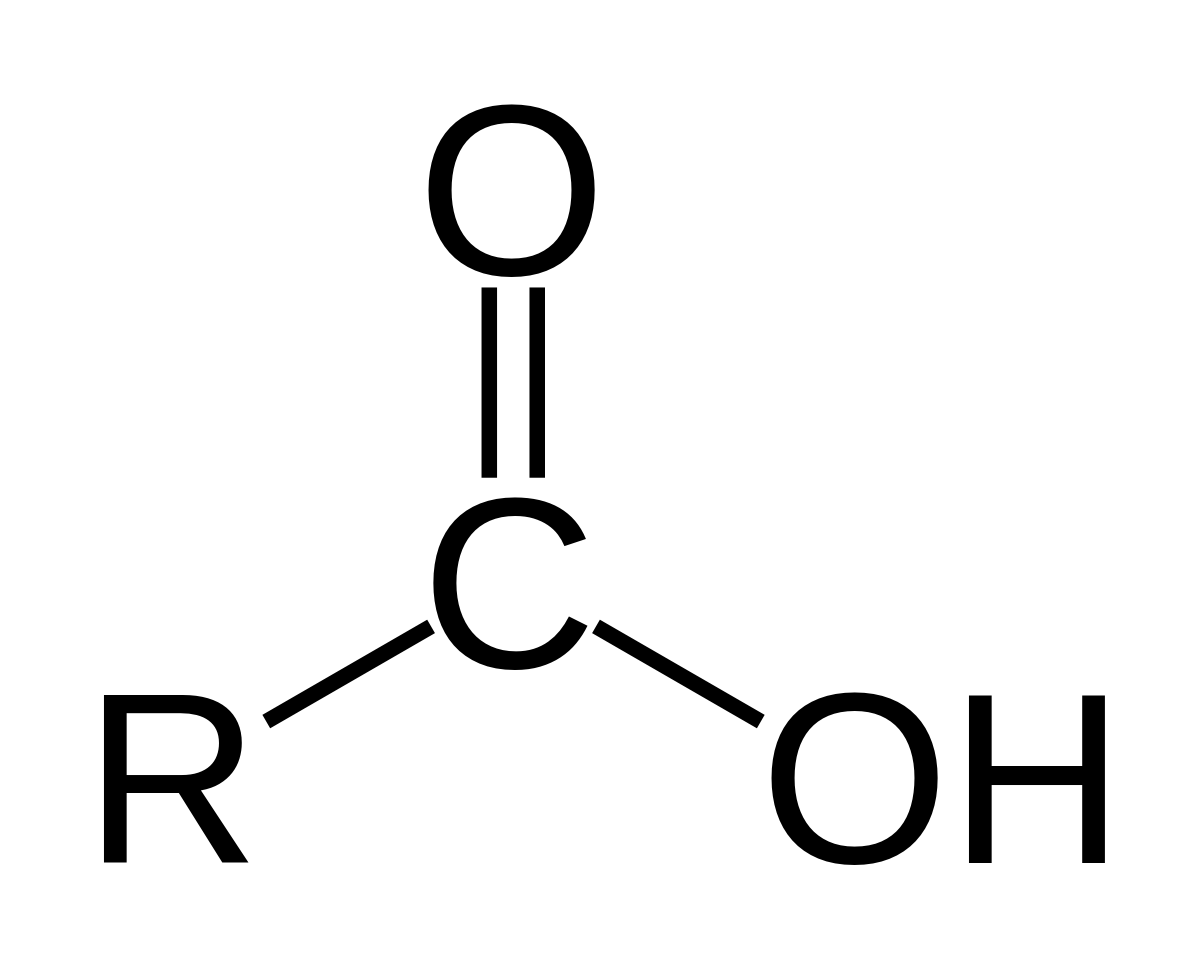
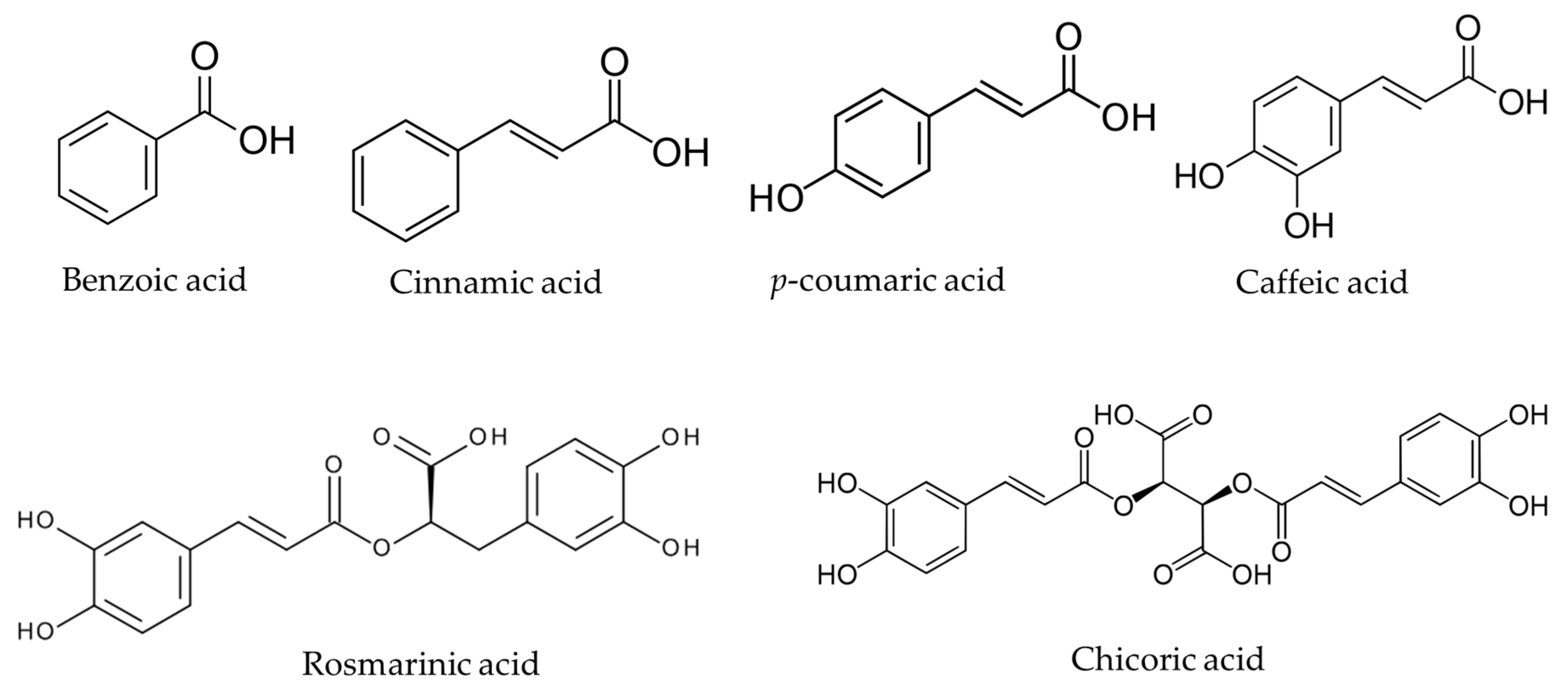
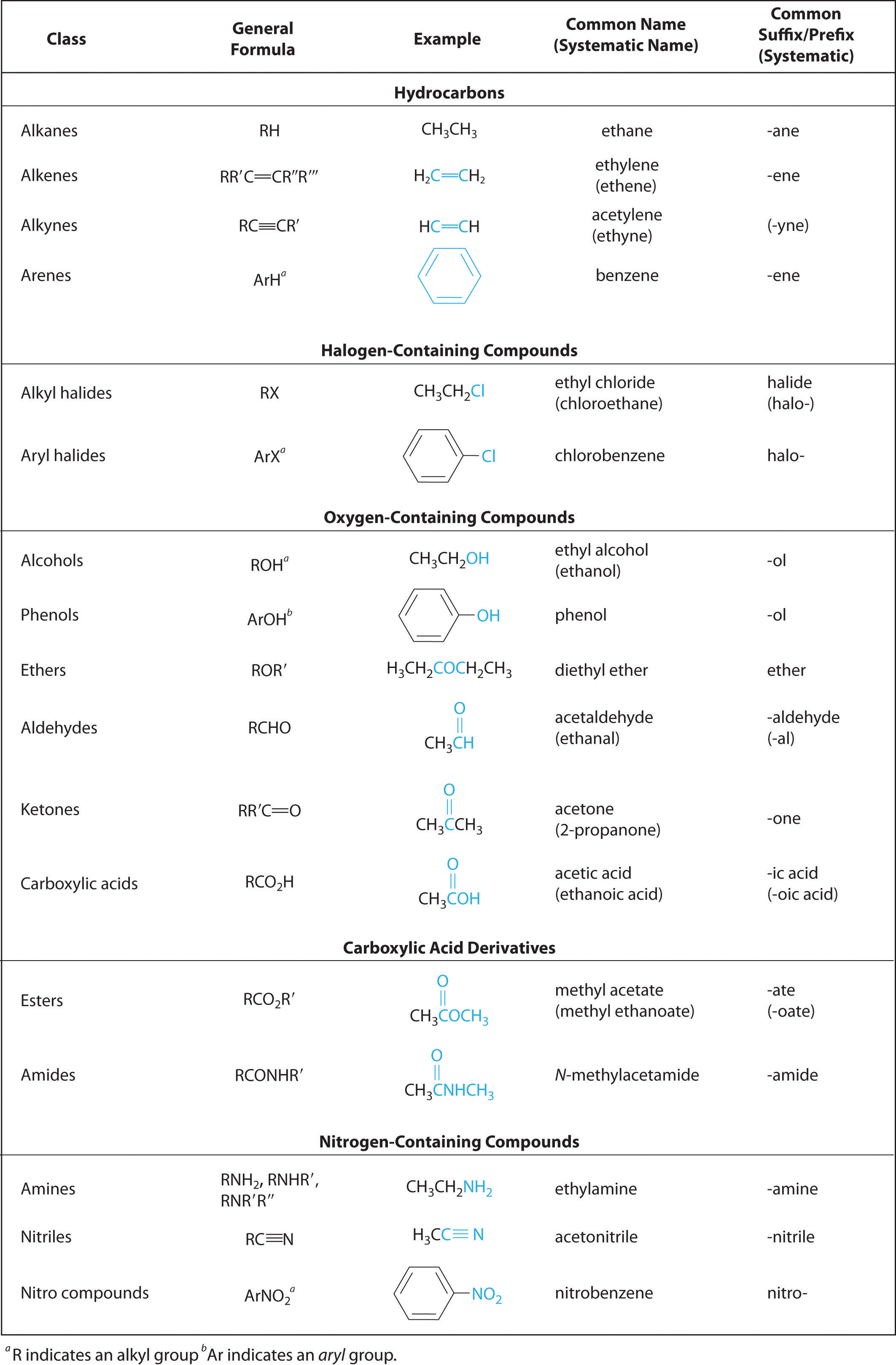
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group (−C(=O)−OH) attached to an R-group.
The general formula of a carboxylic acid is often written as R−COOH or R−CO 2H, sometimes as R−C(O)OH with R referring to the alkyl, alkenyl, aryl, or other group.
What is the classification of ester?
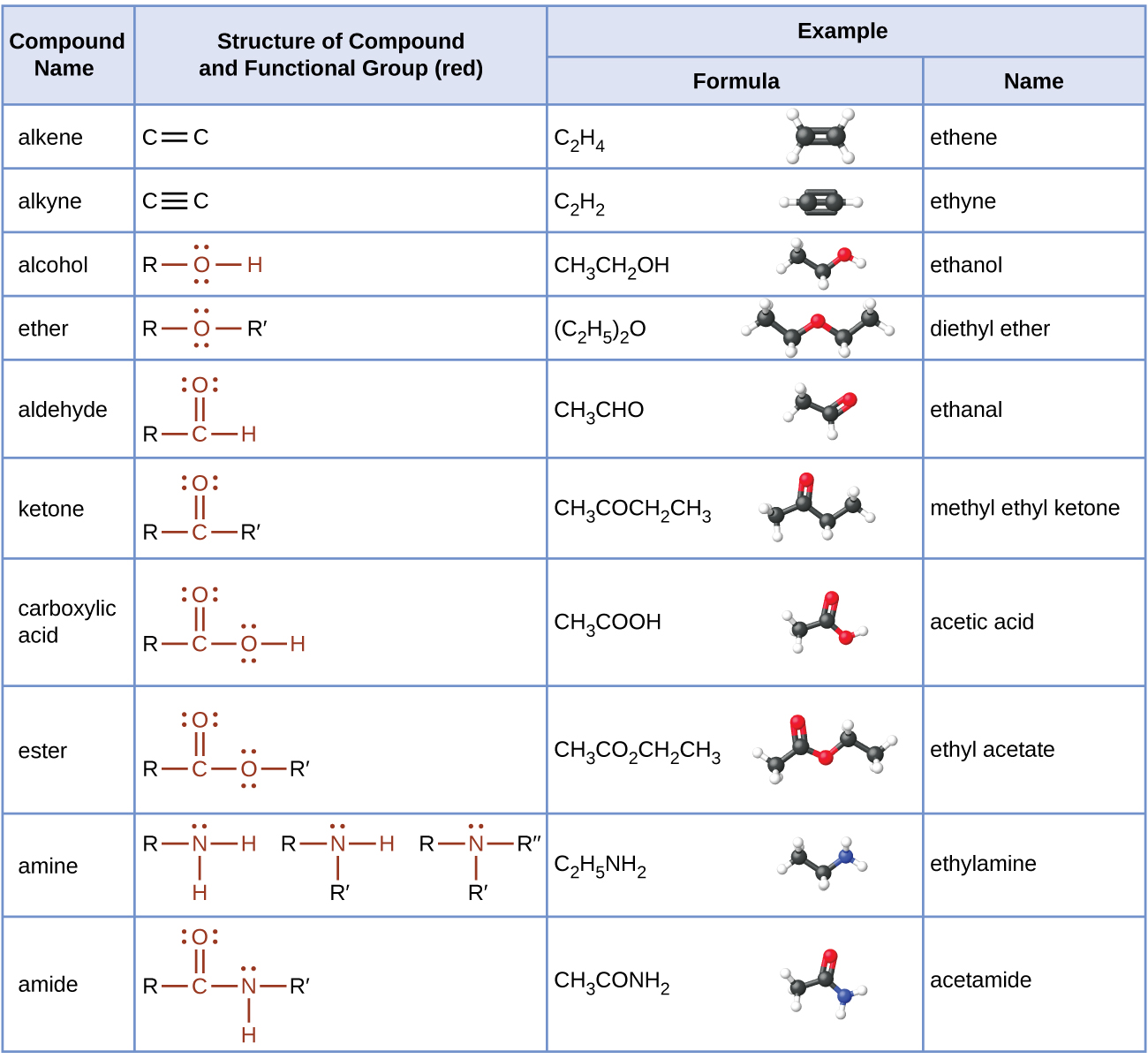
ester, any of a class of organic compounds that react with water to produce alcohols and organic or inorganic acids.
Esters derived from carboxylic acids are the most common.
What is the structural formula of ester?
The ester structural formula is, R-CO-OR, where, R, is an alkyl group, the, C=O, is the carbonyl group, and, -OR, is the alkoxy group.
The angles between the bonds are equal to 120 degrees.
The structure of esters can be drawn by following the, R-CO-OR, format.
What is the formula for carboxylic and ester?
In general, carboxylic acids are represented by the formula RCOOH, where R is a hydrocarbon group.
Esters are represented by the formula RCOOR', where R and R' are hydrocarbon groups.
|
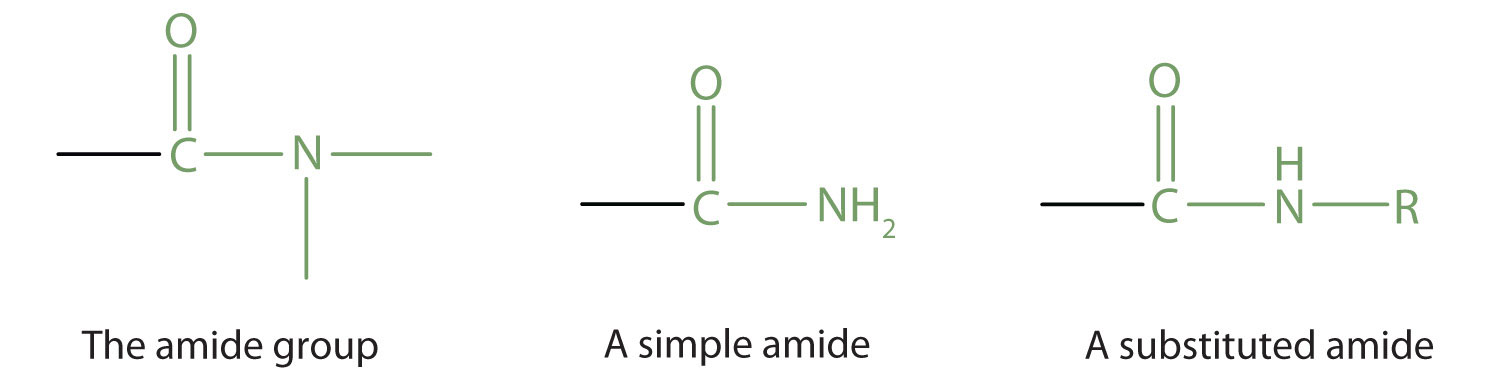
Chapter 6 Amines and Amides
Amides are named by changing the -oic acid ending of the corresponding carboxylic acid to -amide. If alkyl groups are attached to the nitrogen they are named |
|
Chapter 5 Carboxylic Acids and Esters
Which member of each of the following pairs of compounds would you expect to have a higher solubility in water? – 2-butanone or propanoic acid. – hexanoic acid |
|
Chapter 1 Organic Compounds: Alkanes Organic chemistry
Draw all possible structures having the formulas. C4H10 C5H12 |
|
Untitled
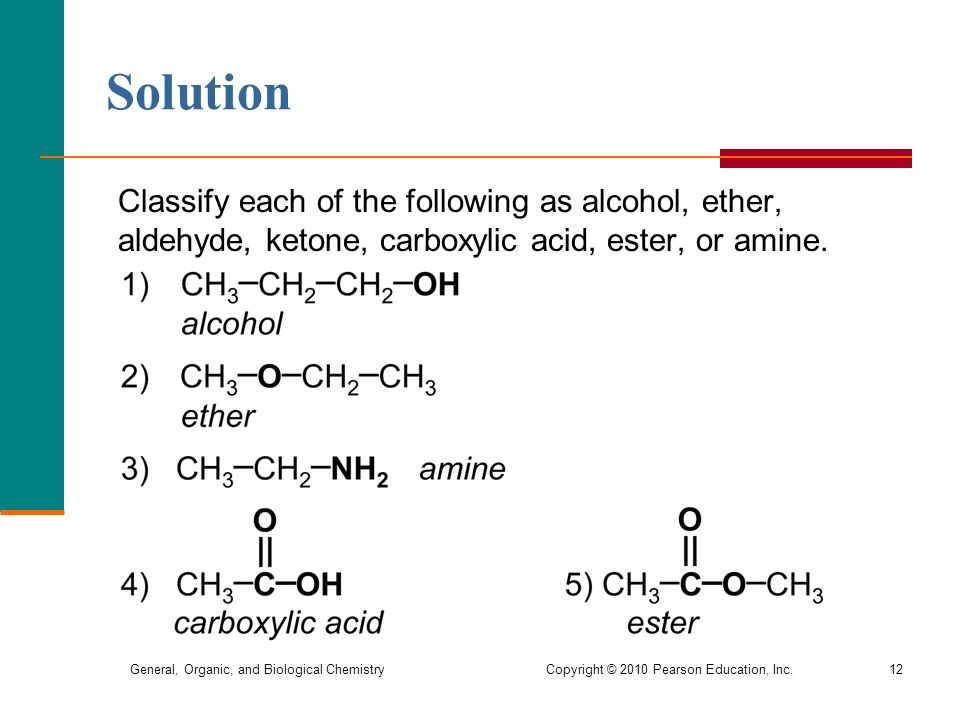
Classify each of the organic compounds below as an alcohol organic acid |
|
Untitled
following structural formula belong? A) ethers. B) cones. 14. CH3. CH3N. CH3. C) esters. D) amines. Which structure represents an amide? о. A). CH3COCH2CH3. |
|
Functional Groups Key
Functional Group Classification ketone ether amide carboxylic acid ester alcohol ester. 5. Draw the monomers that would be used to prepare the following |
|
Practice Tests Answer Keys Organic Chemistry I
Predict the major organic product (1 major structure is all that is needed in each case) for each Classify each chiral carbon as R or S. (Some structures may ... |
|
Chapter 8 - Alkenes Alkynes and Aromatic Compounds
Each fatty acid can have different Classify each compound as saturated or unsaturated. 1. Page 44. 44. 2. 3. Give the molecular formula for each compound. |
|
Classification and Nomenclature of Amines
Amides are named by changing the -oic acid ending of the corresponding carboxylic acid to -amide. If alkyl groups are attached to the nitrogen they are named |
|
Chapter 3 Alcohols Phenols
https://www.angelo.edu/faculty/kboudrea/index_2353/Chapter_03_2SPP.pdf |
|
Chapter 6 Amines and Amides
Examples: Classifying Amines Draw structural formulas for the following molecules: ... of the corresponding carboxylic acid to -amide. If. |
|
Untitled
4 avr. 2020 Classify each of the organic compounds below as an alcohol organic acid |
|
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters and learn how to Draw structural formulas for the following molecules:. |
|
Practice Tests Answer Keys Organic Chemistry I
For each of the following pairs of resonance structures circle the one that would make a Carboxylic Acid. Ketone. Alcohol. Alkene. Amine. Ester. Ester. |
|
Chapter 1 Organic Compounds: Alkanes Organic chemistry
Draw all possible structures having the formulas. C4H10 C5H12 |
|
QSAR Toolbox
also serves as complete reference manual for all QSAR Toolbox features ethers |
|
Final Report Part A: Polymers
6 déc. 2012 4.3.6 Hazards from Polymers Monomers and All Substances . ... Oxy carboxylic acid (HO-Y-COOH |
|
1. Identify the functional groups on the following organic molecules.
12 avr. 2018 a. Carboxylic Acid ???? b. Halide c. Ester d. Amine e. Ketone f. Aldehyde ... Which is the structural formula for propanone. (acetone)?. |
|
Structural Isomers – Just how many structures can you make from a
condensed line formula structural or constitutional isomers for our saturated formula of C5H12. ... aldehyde ketone carboxylic acid ester. 1o amide. |
|
Nitrosamines EMEA-H-A5(3)-1490 - Assessment Report
25 juin 2020 N-nitrosamine-carboxylic acid. N-nitrosamine-benzyl ester. Figure 2.2.2.2-4 Valsartan specific N-Nitroso compounds (potentially) present in ... |
|
Chapter 13 Carboxylic Acids, Esters, Amines, and Amides
A carboxylic acid contains a carboxyl group, which Give the IUPAC names of each compound Draw the structure of the following compounds Amines are classified as primary, secondary, or tertiary Write the structural formula for |
|
Carboxylic Acids, Amines, and Amides
Given the structure of a carboxylic acid, carboxylate ion, ester, amide, or amine molecule, be able to give the systemic names and vice versa 2 Know and Identify chiral carbon atoms in structural formulas Given the Draw each molecule NOTE: When They are classified a primary, secondary, and tertiary according to |
|
Chapter 9 Lecture Notes: Carboxylic Acids, Amines, and Amides
Given the structure of a carboxylic acid, carboxylate ion, ester, amide, or amine molecule, be Identify chiral carbon atoms in structural formulas (C=O), and a ______ group (-OH) that are connected to each other and the hydrocarbon part as group instead of an alkyl group (R) it is still classified as carboxylic acid |
|
Chapter 17 An Introduction to Organic Chemistry, Biochemistry, and
Exercise 17 1 - Organic Compounds: Identify each of these structures as a alkane b amine c ether d ester e ketone f carboxylic acid g amide Exercise 17 2 - Condensed Formulas: Write condensed formulas to represent the Lewis Classify each of the following as organic or inorganic (not organic) compounds |
|
Organic_overheadspdf
Draw the Lewis electron-dot structure for an atom of carbon: C How many Classify each of the organic compounds below as an alcohol, organic acid, aldehyde, ketone, ether, ester, amine, or amide and then draw its structural formula 1 |
|
Chapter 17: Amines and Amides
hydrogen: Alkanes, alkenes, alkynes, and aromatic hydrocarbon are all carbon- hydrogen carboxylic acids, and esters In this chapter we extend our discussion to 17 2 Structure and Classification of Amines General formula CnH2n+3N |
|
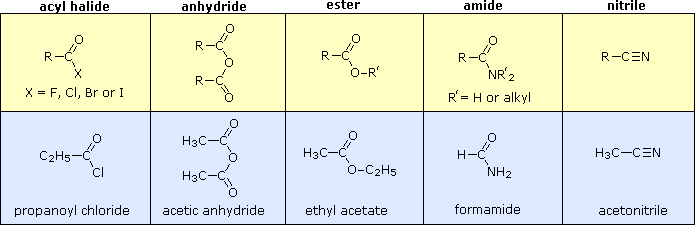
Esters, Amides, and Related Molecules - Organic Chemistry
general formula R-C(=O)-Z (Figure 15 02) of acid chlorides into carboxylic acids, esters, amides, or anhydrides (Figure 15 09) to all of the other compounds with the structure R-C(=O)-Z (Figure 15 09 and Table 15 01) We classify it |
|
Chapter 23: Substituted Hydrocarbons and Their Reactions
You will classify reactions of Carbonyl Ketone Carbonyl Carboxylic acid Carboxyl Ester Ester Amide Amido ——O * — C — H carboxylic acids have the general formula Classify each of the following structures as one of the types of |
|
Mutipage Intro org chem
Type of compound General structure cture Classify each of the following compounds as an alkane, Family Carboxylic Acid Ester Aldehyde Ketone Amide Amine Functional / to produce the full formula, add hydrogen atoms to every |
|
Lab 14: Qualitative Organic Analysis - California State University
Both strong and weak acids (Carboxylic acids and phenols) will be deprotonated by NaOH Also, for each classification test that you Water Soluble Amides Alkaline hydrolysis C-2 Tests for the presence of amides esters Elemental The chemical reaction(s) you are attempting (with skeletal structures R groups are |