colligative properties definition chemistry
The word colligative has been derived from the Latin word 'colligatus' which literally means 'bound together'.
It is also known that in any solution, the solute and solvent particles are bonded to each other.
What are colligative properties and melting point?
Because the change in vapor pressure is a colligative property, which depends only on the relative number of solute and solvent particles, the changes in the boiling point and the melting point of the solvent are also colligative properties.
What is a colligative property in chemistry?
Colligative properties are the physical changes that result from adding solute to a solvent.
Colligative Properties depend on how many solute particles are present as well as the solvent amount, but they do NOT depend on the type of solute particles, although do depend on the type of solvent.23 sept. 2022
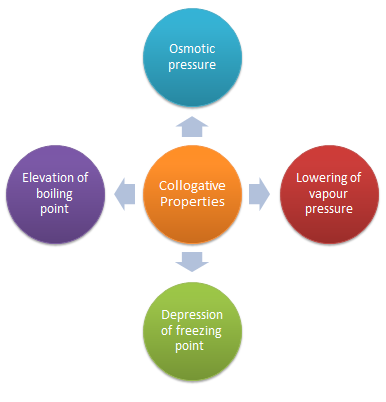
What are the 4 colligative properties?
These colligative properties include vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure.
This small set of properties is of central importance to many natural phenomena and technological applications, as will be described in this module.
|
Colligative properties of solutions
Medical Chemistry. Viktoriia Tsuber PhD. Page 2. Definition. Colligative properties depend only on Definition 1: Osmotic pressure is equal to. |
|
Properties Of Solutions Chemistry
27 août 2022 Inorganic Chemistry . 5. Coordination. Compounds . 14 Marks . ... Colligative properties – definition and examples and its use in determination ... |
|
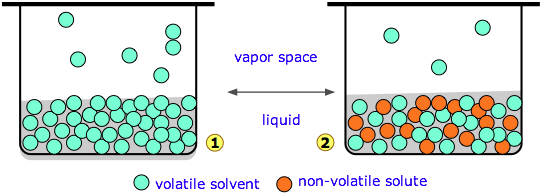
Solutions
The properties of solutions which depend on the number of solute particles and are independent of their chemical identity are called colligative properties. |
|
Colligative Properties Of Solutions Include All The Following Except
It covers topics including soil chemical environment soil minerals |
|
Solutions
pressure and colligative properties. We will begin with a solution is defined as: ... mass percentage is commonly used in industrial chemical. |
|
Colligative Properties Of Solutions Include All The Following Except
300 new definitions added for the latest terms in health care drugs and medical and nursing terminology. Tietz Fundamentals of Clinical Chemistry and Molecular |
|
Colligative Properties
colligative properties to measure the molecular weight of polymers. A well known result from introductory chemistry is that the boiling point elevation ... |
|
Colligative Properties Of Solutions Include All The Following Except
6 sept. 2022 Tietz Fundamentals of Clinical Chemistry and Molecular Diagnostics - E-Book. Carl A. Burtis. 2014-08-14 A condensed easier-to-understand ... |
|
Colligative Properties Of Solutions Include All The Following Except
been optimized for a one-semester introductory course in physical chemistry for students of biosciences. Mosby's Dictionary of Medicine Nursing and Health |
|
The Science of a Sundae: Using the Principle of Colligative
In each unit we use these familiar food items to illustrate how colligative properties (or simply |
|
Colligative Properties of Foods - Encyclopedia of Life Support Systems
colligative properties (i e , freezing point depression, elevation of boiling point, and A combination of Equations (2) and (3) to define the chemical potential of |
|
Colligative Properties
Historically, colligative properties have been one means A well known result from introductory chemistry is that the boiling point elevation is propor- tional to |
|
Experiment 1: Colligative Properties - ULM
Background: Colligative properties are properties of a solvent, such as freezing point depression and boiling point elevation, which depend on the concentration of solute particles dissolved in the solvent |
|
Colligative properties - General Chemistry, Boston University
Increased vapor entropy corresponds to lower vapor pressure, that is, fewer particles in a given volume and so a greater number of arrangements W Examples: |
|
Phase Equilibria and Colligative Properties
The chemical potential of a system is zero at equilibrium To maintain used to develop a broad range of equations, the equations for colligative properties The Gibbs free 9 2 An osmotic pressure defined by the height difference of liquid |
|
Lecture 4: Colligative Properties
By definition a colligative property is a solution property (a property of mixtures) for If colligative properties depend on the amount of the solute in the solvent, then the you don't learn about unless you take upper division physical chemistry |
|
AP* Chemistry PROPERTIES OF SOLUTIONS
Colligative Properties— properties that depend on the number of dissolved particles; A solution does not have a sharply defined freezing point, a solvent does |
|
Chapter 13 Properties of Solutions
The differences between the definitions of molarity (M) and molality (m) and between their Some solutions form by physical processes and some by chemical processes Colligative properties depend on number of solute particles |
|
Solutions - NCERT
describe colligative properties of pressure and colligative properties Chemistry Type of Solution Solute Solvent Common Examples Gaseous Solutions |