concentration of resulting solution calculator
C1V1=C2V2 is used to calculate an unknown quantity where two solutions/mixtures are proportional … C1V1 = Concentration/amount (start) and Volume (start) C2V2 = Concentration/amount (final) and Volume (final) 1.
What will be the concentration of the resulting solution?
Divide the mass of the solute by the total volume of the solution.
Write out the equation C = m/V, where m is the mass of the solute and V is the total volume of the solution.
Plug in the values you found for the mass and volume, and divide them to find the concentration of your solution.
What is the molar concentration of the resulting solution?
To get the molarity, you divide the moles of solute by the litres of solution.
For example, a 0.25 mol/L NaOH solution contains 0.25 mol of sodium hydroxide in every litre of solution.
To calculate the molarity of a solution, you need to know the number of moles of solute and the total volume of the solution.
How do you find the resulting concentration of a solution?
Step 1: Identify the mass of the solute.
Step 2: Identify the volume of solution.
Step 3: Divide the mass of the solute by the volume of solution to find the concentration of the solution.
|
Pharmaceutical calculation
16 juil. 2021 Example Calculations of the Dilution and Concentration of Liquids ... calculate percent strength of the resulting solution? Convert mg to g. |
|
2 Amount and concentration: making and diluting solutions
The concentration of the resulting solution is 1M /10 = 0.1M where 10 is the dilution factor. [Although it is really quite obvious you can convince |
|
Calhoun Community College
WHAT IS THE FINAL CONCENTRATION OF THE SOLUTION PREPARED FOR IM THIRD WHAT IS THE RESULTING TOTAL VOLUME OF THIS ... SOLUTION CALCULATION ANSWER. |
|
Laboratory Math II: Solutions and Dilutions
We talk about solutions in terms of concentrations how much of each substance |
|
Reconstitution of Solutions
Terms. • Solution. – Resulting mixture of solute plus solvent What is the dosage concentration after reconstitution? ... Solution Calculation Example. |
|
Dilutions Occasionally a solution is too concentrated to be used as it
dilution factor and so on until the final concentration is known. Example: A 5M solution of HCl is diluted 1/5. The resulting solution is diluted 1/10. |
|
Sample Exercise 17.1 Calculating the pH When a Common Ion is
Calculate the fluoride ion concentration and pH of a solution that is 0.20 M Equilibrium Calculation: We now turn our attention to the equilibrium that ... |
|
Making Dilutions Worksheet Key.pdf
Challenge Question: Exactly 16.0 mL of a solution A is diluted to 300 mL resulting in a new solution. B that has 0.50 M concentration. If the solution was made |
|
Guidance For The Use Of The Synthetic Precipitation Leaching
APPENDIX C Calculation of Leachate Concentrations under Field Conditions Using the resulting solution is known as the leachate. Method 1312 directs the ... |
|
2 Amount and concentration: making and diluting solutions
you perform these calculations quickly, reliably and accurately The concentration of the resulting solution is 1M /10 = 0 1M where 10 is the dilution factor |
|
Dilution and Concentration
dilution and concentration uncomplicates these problems Many problems and petrolatum Pharmaceutical Calculations for the Pharmacy Technician would be the percentage concentration w/v of the resultant solution after a 5-g packet of |
|
Dilutions Occasionally a solution is too concentrated to be used as it
The resulting solution is diluted 1/10 Determine The calculations used to determine the concentration of each solution in a series may be used in reverse to |
|
Laboratory Math II: Solutions and Dilutions - NIH Office of Intramural
What units you will be reporting the concentration in □ If molarity or normality, the molecular or formula weight of the substance (solute) ▫ The desired volume |
|
Concentration of Solutions and Molarity - Denton ISD
b) Three moles of sodium chloride dissolved in water produce 6 mol of particles because each formula unit of NaCl dissociates into two ions 3 moles Na+ and 3 |
|
Making Dilutions Worksheet Keypdf
Remember that you can change the concentration of a solution by adding more are questions regarding molarity and the others involve the dilutions formula above 1 16 0 mL of a solution A is diluted to 300 mL, resulting in a new solution |
|
Reconstitution of Solutions
Cengage Learning ALL RIGHTS RESERVED Terms • Solution – Resulting mixture of solute plus solvent 2 What is the dosage concentration after reconstitution? • 100 mg per mL Solution Calculation Example • Physician orders |
|
Concentrations and Dilutions INTRODUCTION
solutions INTRODUCTION Concentrations of many pharmaceutical preparations are expressed as total quantity *To accurately perform concentration calculations, the proportion What is the resulting ratio strength when you dilute 12 mL |









/how-to-calculate-normality-609580final2-0d5efa5a961f4fa0a7efc780921faee1.png)
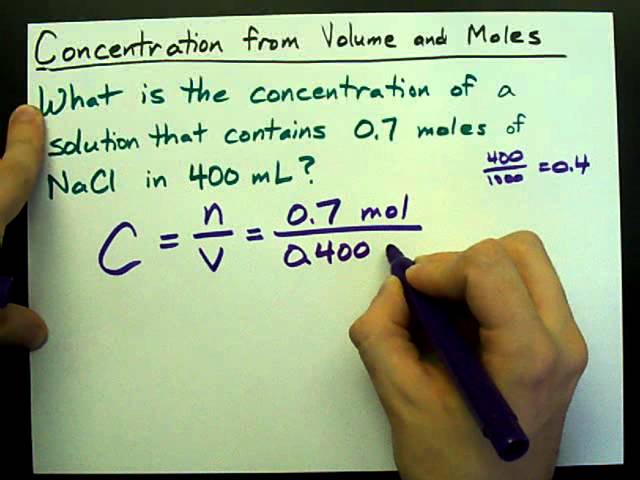





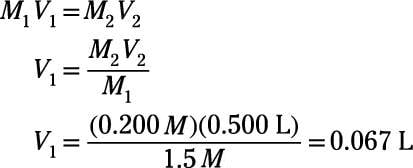






/606823-calculate-molarity-of-a-solution-FINAL-5b7d7e15c9e77c0050355d4e.png)

