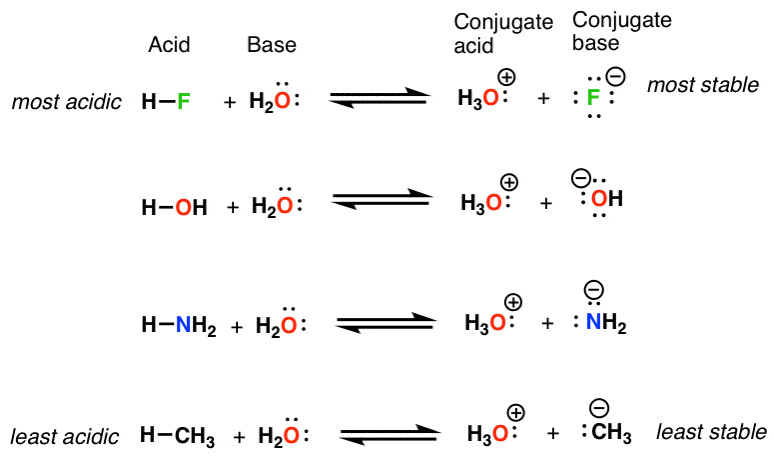
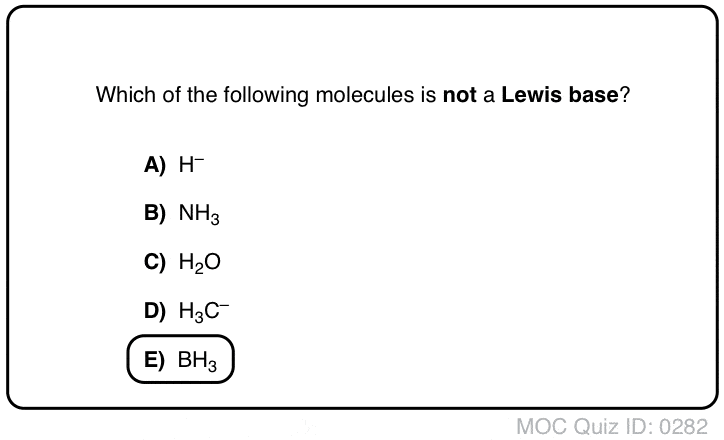
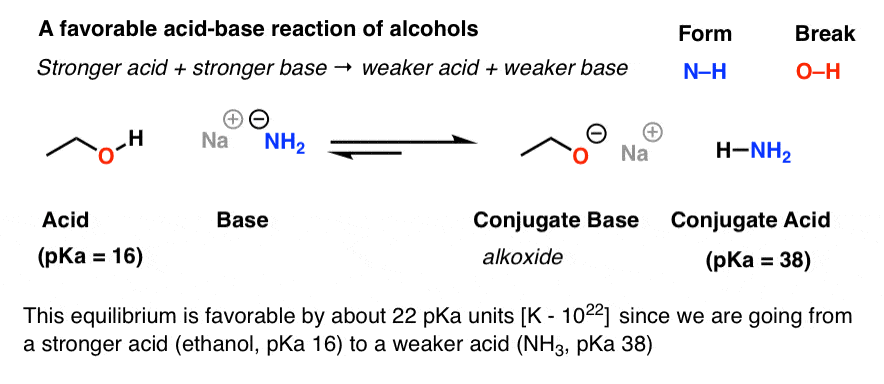conjugate acids and bases practice problems
|
Practice Problems 1 Draw the structure of the conjugate acid of
Draw the structure of the conjugate base of each of the following ions or compounds 3 Classify each compound as a Lewis acid or Lewis base in the reactants |
|
Conjugate Pairs Practice Questions
Conjugate Pairs Practice Questions 1 Identify the acid base conjugate acid and conjugate base for each of the following a) HClO4(aq) + H2O(l) ⇌ H3O+(aq) |
Is it accurate to say that 2.0 M solution of H2SO4 which contains two acidic protons per molecule is 4.0 M in H+?
It is not accurate to say that a 2.
0) M solution of H2SO4, which contains two acidic protons per molecule, is 4.
0) M in H+ because a 2.
0) M solution of H2SO4 is equivalent to 4.
0) N in H+.18 jui. 2021How do you find the conjugate acids and bases?
The formula of the conjugate base is the formula of the acid less one hydrogen.
The reacting base becomes its conjugate acid.
The formula of the conjugate acid is the formula of the base plus one hydrogen ion.Is h3o +/ OH a conjugate acid base pair?
Answer and Explanation:
The species that accept the hydrogen ion are conjugate acids and the species from which the proton is removed are the conjugate bases.
The conjugate base for H 3 O + is water which acts as a weak base.
Hence, H 3 O + and are not a conjugate acid-base pair.
|
Test2 ch17a Acid-Base Practice Problems
the more concentrated the conjugate base. c. the more concentrated the acid. 11. Ammonia (NH3) acts as a weak base in aqueous solution. What is the |
|
Sample Exercise 16.1 Identifying Conjugate Acids and Bases
Plan: We approach this problem as we would previous equilibrium problems. We begin by writing the chemical equation for the equilibrium and tabulating the known |
|
Chapter 11 - Acids and Bases - Practice Problems - Section 11.1
by one H. Conjugate acid-base pairs: H2SO4/HSO4 andH3O+ /H?O. El. Practice Problems. |
|
Chapter 11 – Acids and Bases – Practice Problems Section 11.1
When H2O acts as a base it gains one H+ |
|
Practice Problems -? Answers 1. Draw the structure of the conjugate
Draw the structure of the conjugate acid of each of the following ions or Classify each compound as a Lewis acid or Lewis base in the reactants. |
|
1. For each of the species below identify the most acidic proton and
PRACTICE PROBLEMS FOR BRONSTED-LOWRY ACID-BASE CHEMISTRY structure of the corresponding conjugate base. ... corresponding conjugate acid. |
|
Organic Chemistry Jasperse Acid-Base Practice Problems
Acid-Base Practice Problems. A. Identify each chemical as either an “acid” or a “base” in the following reactions and identify “conjugate” relationships. |
|
Untitled
Ch.14 - Acids Bases |
|
Chapter 17 Acid-Base Equilibrium Systems Solutions to Practice
24?/08?/2007 Problem. Identify the conjugate acid-base pairs in the following reactions: ... Write two chemical equations to show the ionization of. |
|
Ch 7 Practice Problems
Ch 7 Practice Problems Identify the Brønsted acids and bases in the following equation (A ... Which of the following is a conjugate acid-base pair? |
|
Ch 17a Acid-Base Practice Problems
the more concentrated the conjugate base c the more concentrated the acid 11 Ammonia (NH3) acts as a weak base in aqueous solution What is the |
|
Sample Exercise 161 Identifying Conjugate Acids and Bases
conjugate base of the acid H2O Practice Exercise Solution Analyze and Plan: We are asked to write two equations representing reactions between HSO3 |
|
Chapter 11 – Acids and Bases – Practice Problems Section 111
When H2O acts as a base, it gains one H+, which forms its conjugate acid, H3O+ Practice Problems 8 Write the formula for the conjugate base for each of the |
|
Acids and Bases Acids and Bases Conjugate Pair Question
A common equilibrium situation involves acids and bases ▫Many cell conjugate acids H3O+ H2O Examples ▫ Calculate the pH of: ▫ 0 001 M HNO3 [H ] 0 001 M H 3 0 Example: H2CO3 (carbonic acid) has 2 protons that can be |
|
Acid Base Practice Problems 1 Write the formula for the conjugate
When solving problems identification of the chemicals is very important Is the chemical a strong acid (ionizes 100 ), a weak acid (partial ionization and has a |
|
Ch14 Acids Bases Practice Problems - keypdf
A B b) The conjugate acid of NH3 is NH4* The conjugate base of HCO3 is H2CO3 HzC3 + H2O d) The conjugate acid of H2O is H307 e |
|
Practice Problems on Acid-‐Base Chemistry -‐ Answers 1 For each
Practice Problems on Acid-‐Base Chemistry -‐ Answers 1 For each of Draw the structures of the conjugate acid and conjugate base that result from each acid |
|
Ch 7 Practice Problems - UCSB CLAS
Which of the following does not represent a conjugate acid-base pair? A) HF and F- following equations does HSO4 The conjugate base of a weak acid is |
|
Chapter 5 Page 265, Practice Problems 511 1 a) H2SO4 acid b
Page 268, Practice Problems 5 1 2 + NH4 + acid base base acid conjugate pairs: HNO2/ NO2 - and NH3/NH4 + Page 270, 5 1 Review Questions 1 |
|
Bronsted-lowry_acids_and_bases___answerspdf
Worksheet – Bronsted-Lowry Acids and Bases Name Period Date Identity the A base accepts a proton to form a conjugate acid 1 ) HCO3 + NH3 + CO, |