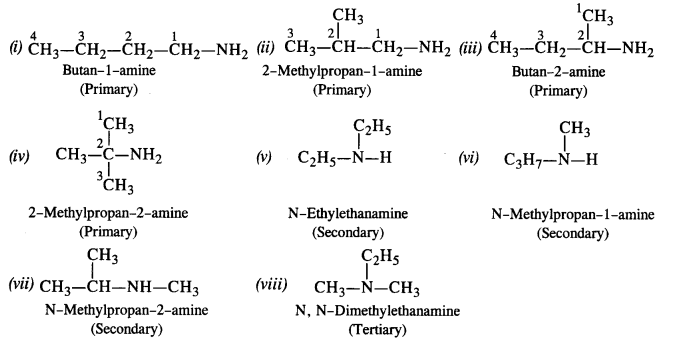
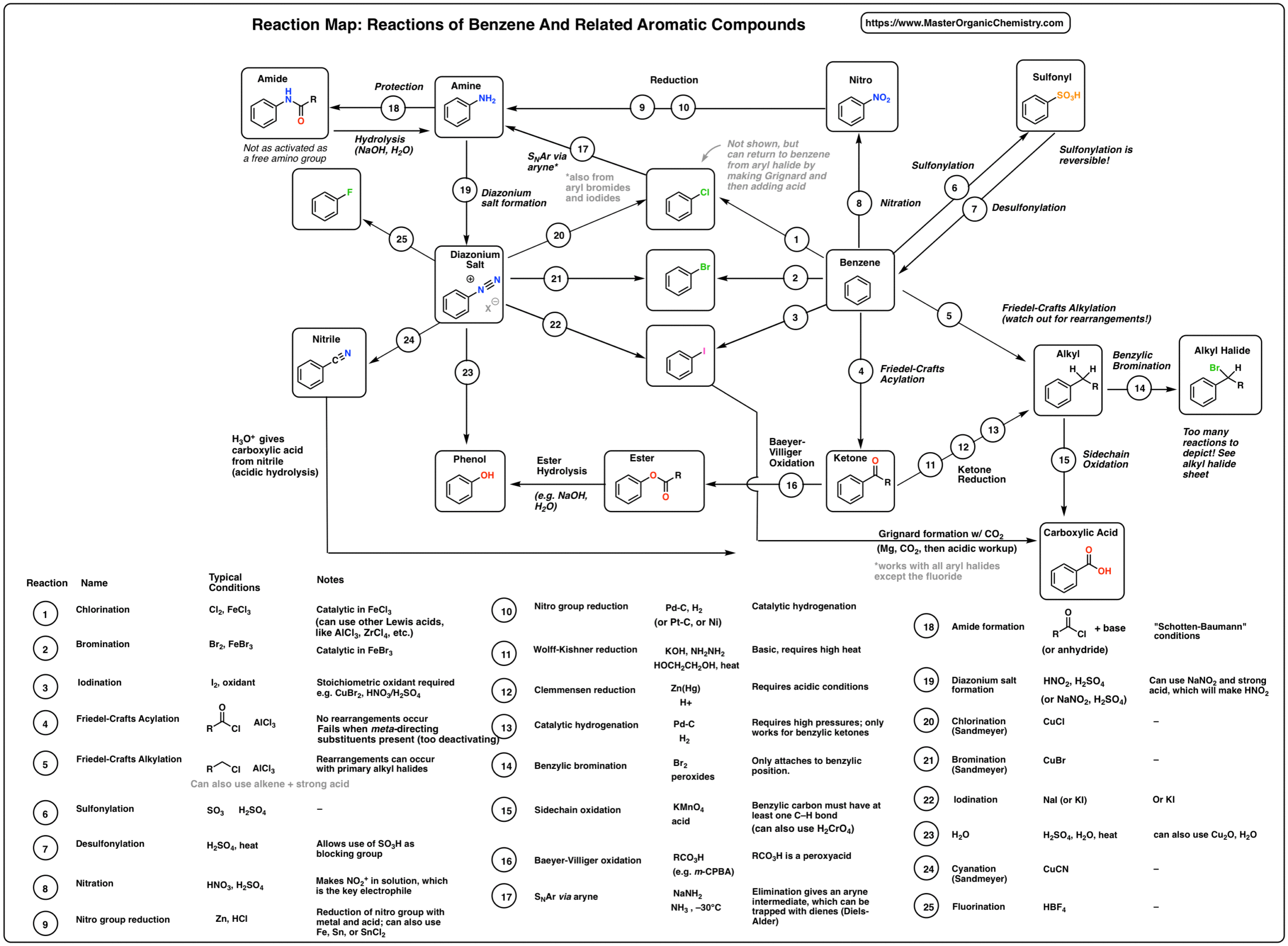
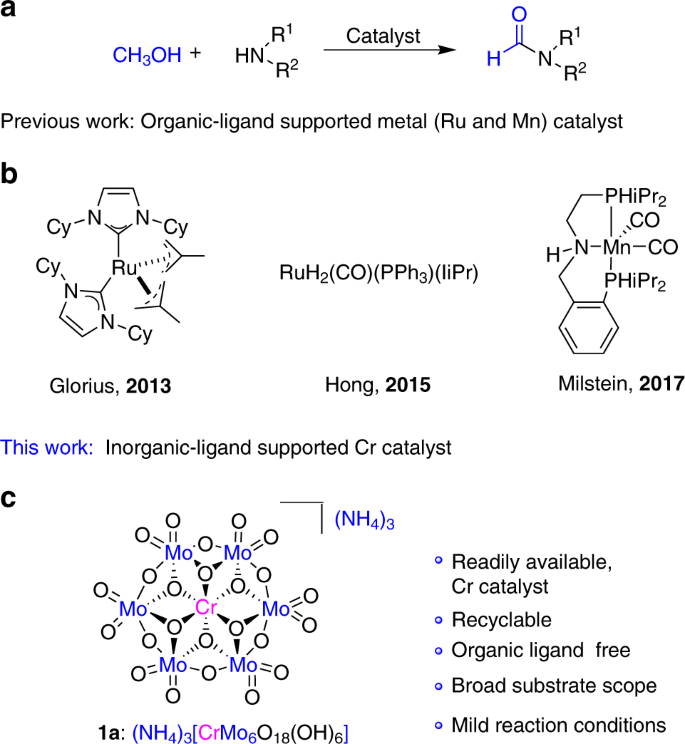
amine hcl reaction
How do aliphatic amines react with nitrous acid?
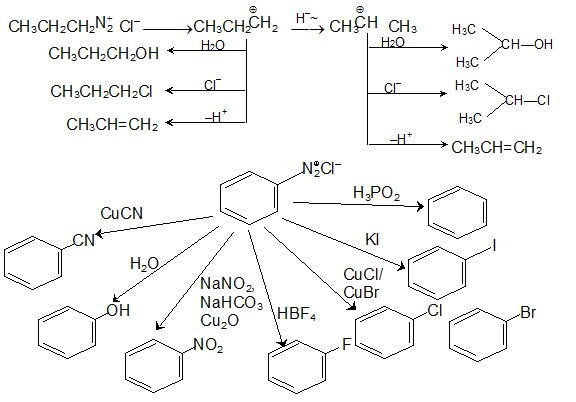
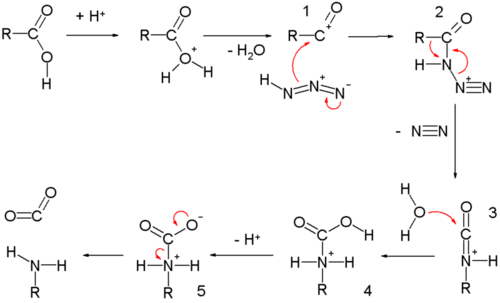
Usually it is produced indirectly in a mixture of NaNO 2 and a strong acid such as HCl or H 2 SO 4 in dilute concentration, so that the H + ions will associate with the NO 2– ions in solution. Primary aliphatic amines with nitrous acid give very unstable diazonium salts which spontaneously decompose by losing N 2 to form a carbenium ion.
How do amines react with alkyl halides?
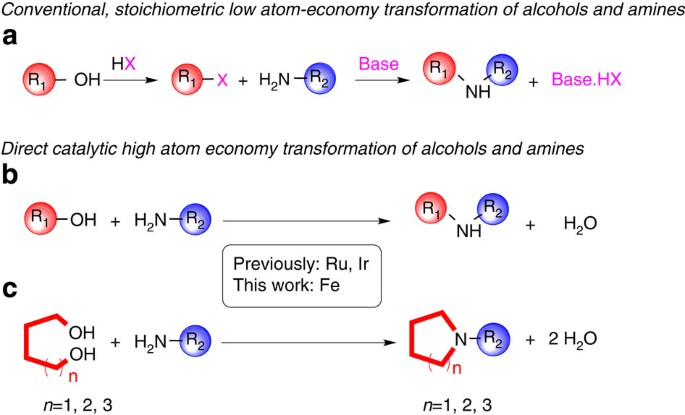
In contrast, amines react with alkyl halides directly to give N-alkylated products. Since this reaction produces HBr as a co-product, hydrobromide salts of the alkylated amine or unreacted starting amine (in equilibrium) will also be formed.

Amine Synthesis Reactions Organic Chemistry

Amine Synthesis Reactions

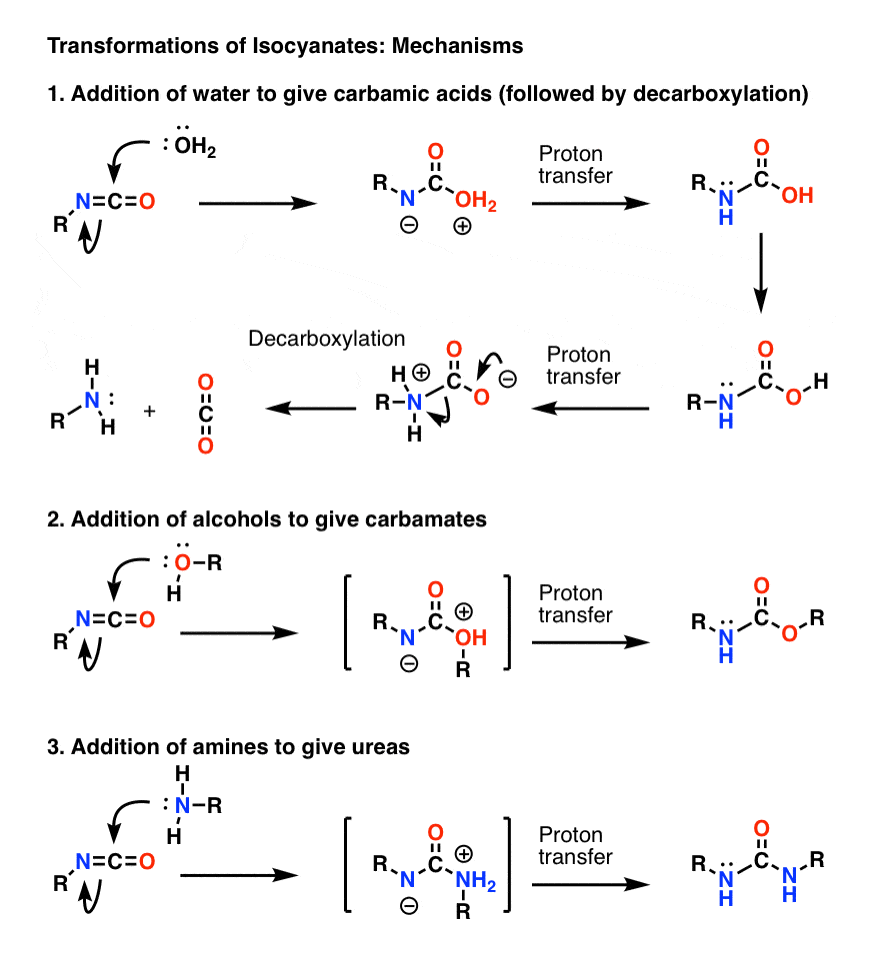
Hofmann Rearrangement and Curtius Reaction Mechanism
|
Reactions of Amines
But 1º 2º |
|
Maillard Reaction of Lactose and Fluoxetine Hydrochloride a
than formulations with starch as the diluent due to the Maillard reaction between the drug a secondary amine hydrochloride |
|
Highly Efficient Oxidative Amidation of Aldehydes with Amine
success partly due to the competing oxidation reaction of the amine. We rationalized that the corresponding amine hydrochloride salt. |
|
Manufacturing Process
15 nov. 2019 Group-2. 1. BIS (2-Chloro Ethyl Amine) Hydrochloride. Manufacturing Process: Di-Ethanol Amine React with Thionyl Chloride. |
|
Maillard reaction of lactose and fluoxetine hydrochloride a
15 déc. 1997 than formulations with starch as the diluent due to the Maillard reaction between the drug a secondary amine hydrochloride |
|
Chapter 6 Amines and Amides
Learn the major chemical reactions of amines and amides and learn how to predict the Complete the following reactions: CH3NH2 + HCl. NHCH3. + HCl. |
|
The Reaction of Thiophene with Formaldehyde and Salts of
ported that hydroxylamine hydrochloride gave salts would undergo a type of Mannich reaction ... from a hydrochloric acid solution of amine IV. |
|
Amine hydrochloride salts: a problem in polyurethane synthesis
HCl(s) can occur. Reaction in a closed system shows no amine production while reaction in an open system permits within solubility limits |
|
Chapter 23 The Chemistry of Amines
react with the amine to give a mixture of diastereomeric salts. In 1 M HCl solution cocaine is protonated on its amino nitrogen. |
|
LES AMINES.pdf
Il s'agit de la réaction des amines avec les acides carboxyliques et ses dérivés d'une solution aqueuse d'HCl sur un nitrite de sodium ( Na+ NO2. |
|
Reactions of Amines
Reaction with Ketones or Aldehydes (Section 18-16,17 and 19-10) R' R O aldehyde Fe, HCl NH2 • Access: 1º Amines only (especially aromatic amines) |
|
Les Amines - AC Nancy Metz
Synthèse de Gabriel alkylation réductive Réact d'Eschweil Réduction Page 2 sur 31 Les Amines carbamoyle qui perd facilement HCl On obtient ainsi les |
|
CHAPTER 7 AMINES
A molecule with two amine groups can react with two molecules of HCl or This neutralization reaction can occur in amino acids within the same molecule: |
|
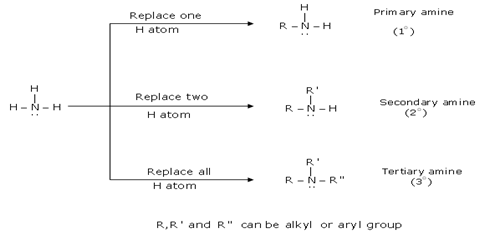
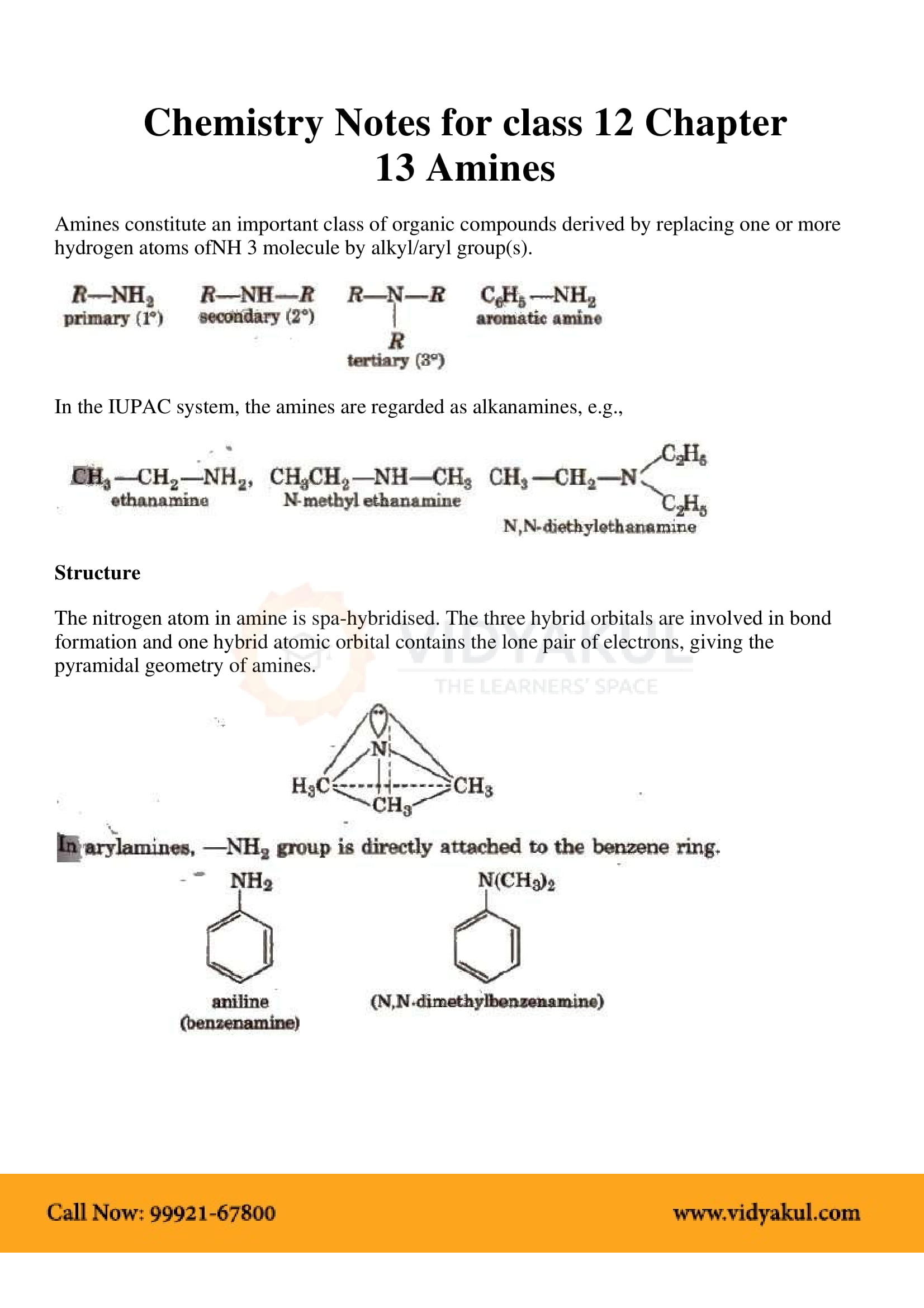
Have an NH2, amine group Amines are derivatives of ammonia
Nitrous acid is unstable so must be made when needed NaNO2 + HCl → HNO2 Sodium nitrite Hydrochloric acid Nitrous acid b) Diazotisation reaction: • |
|
610 Amines - chemrevise
primary amine can react in the same nucleophilic way in a successive series of reactions forming As the reaction is carried out in HCl the salt C6H5NH3 |
|
Reaction of glucose with some amines - CORE
hydrochloric acid (50) Ho attempt has been made to review the literature of this most ccaanon amino sugar except for a ccmparison of the stability of the amine |
|
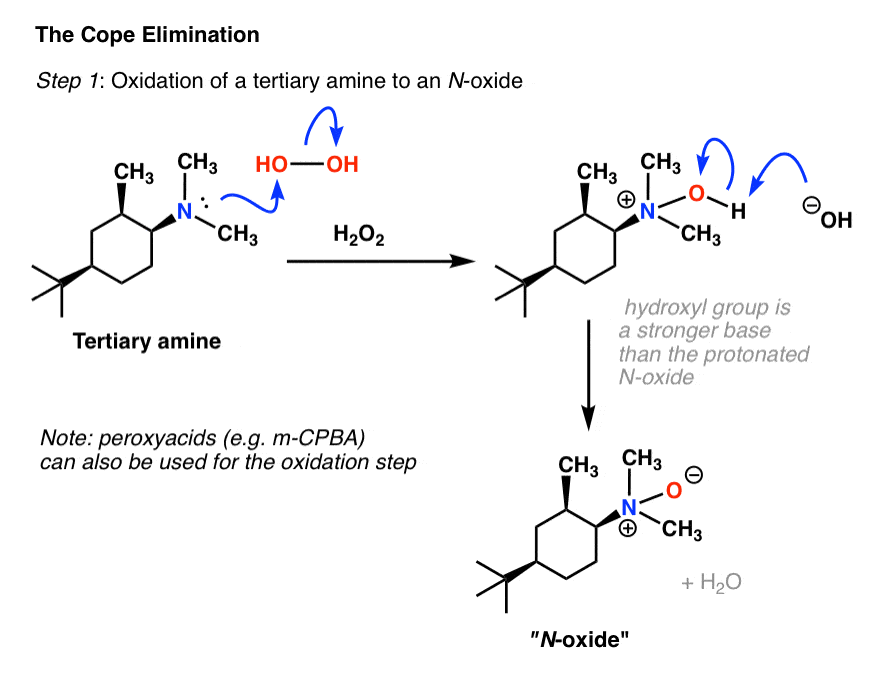
AMINES
For tertiary amines the reaction with Dragendorff reagent Secondary amines do not react in this way aromatic amines in hydrochloric acid solution |






















![Deracemisation of [alpha]-chiral primary amines by a one-pot two Deracemisation of [alpha]-chiral primary amines by a one-pot two](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/dcb0f096-c77e-4683-b703-d8ed65d2cea9/mcontent.gif)