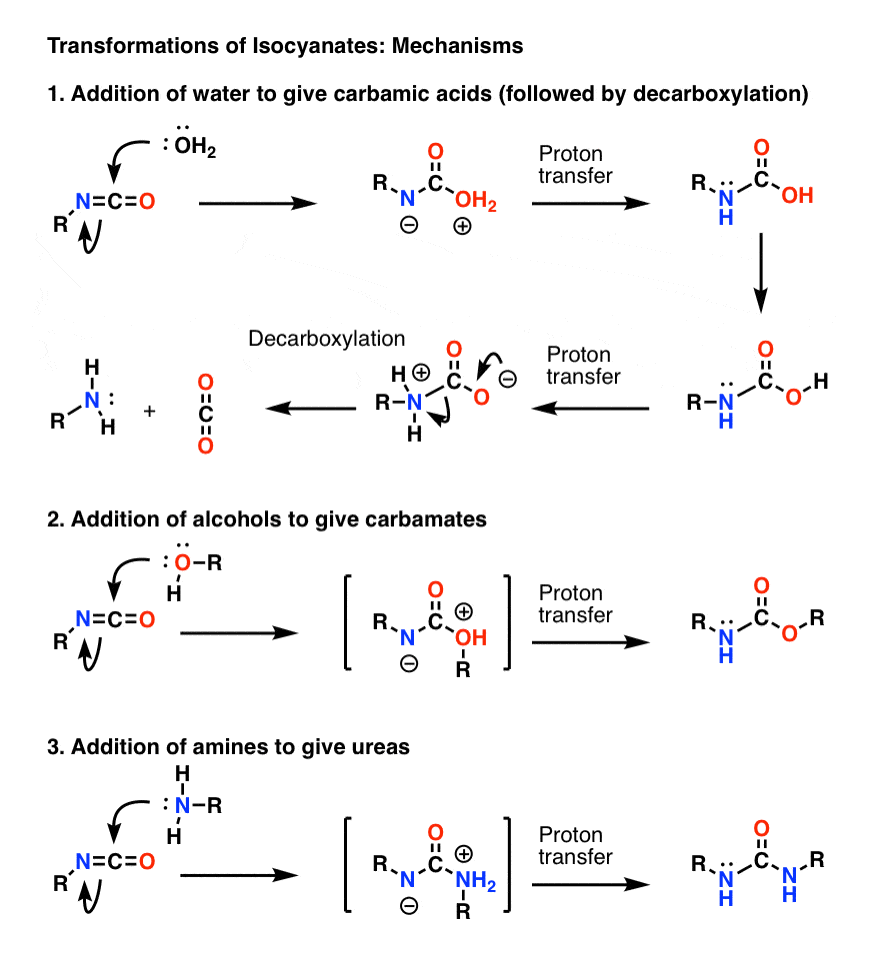
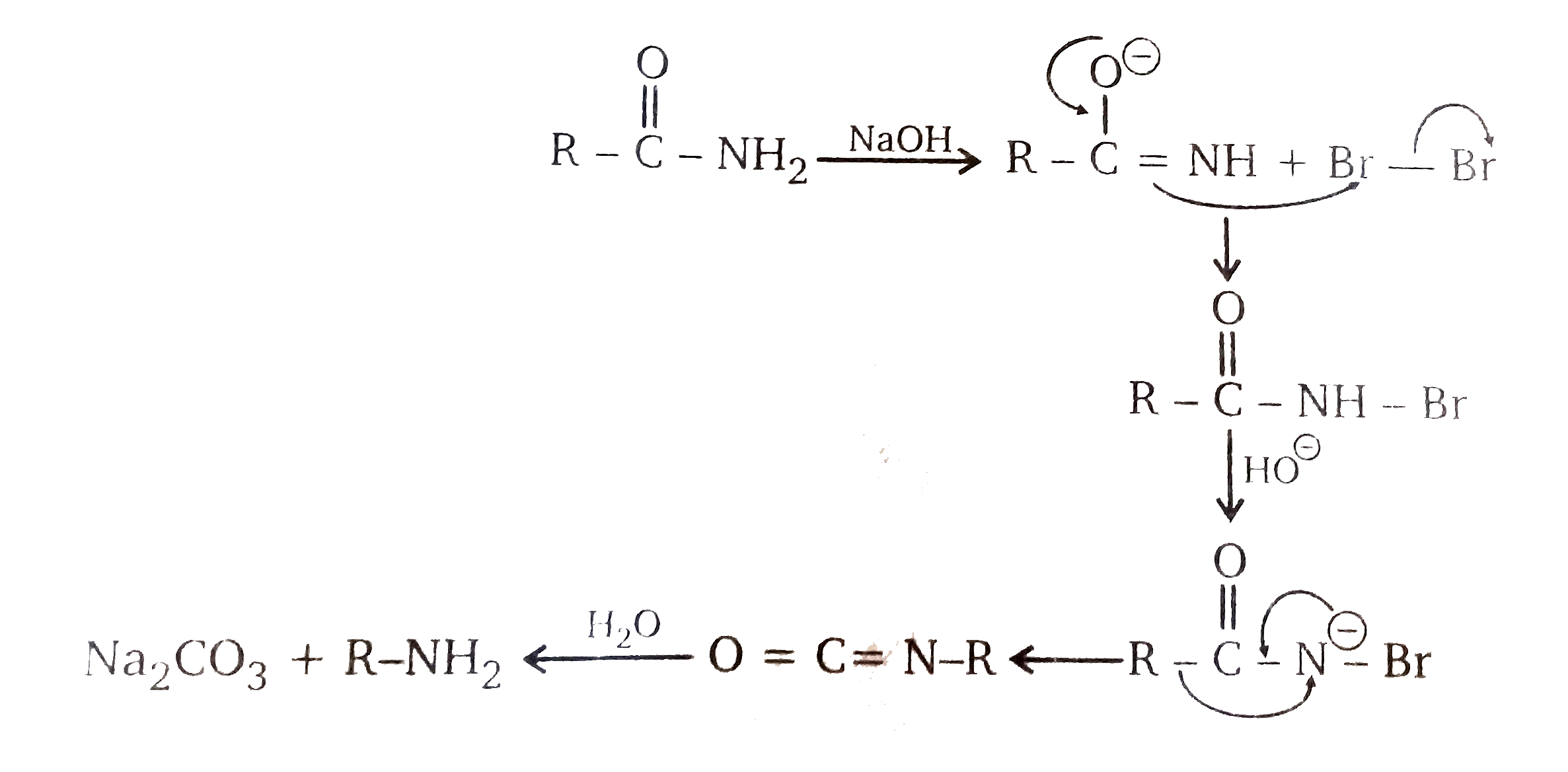amine react with naoh
Which amine reacts with loss of HCl?
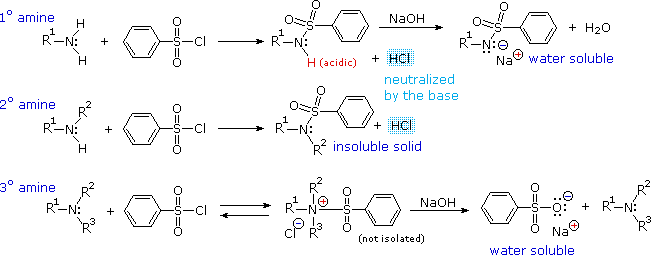
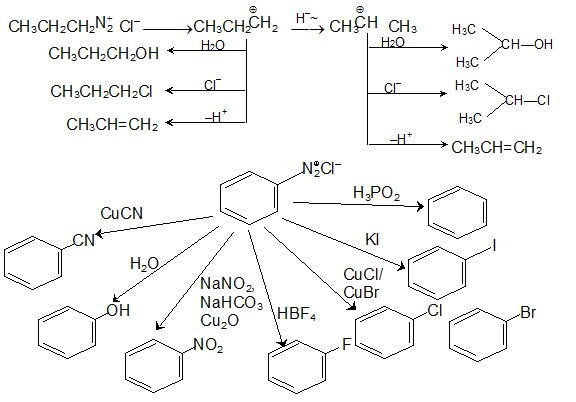
As shown in the following equations, 1º and 2º-amines react to give sulfonamide derivatives with loss of HCl, whereas 3º-amines do not give any isolable products other than the starting amine.
How do you synthesize a 1 o or 2 O amine?
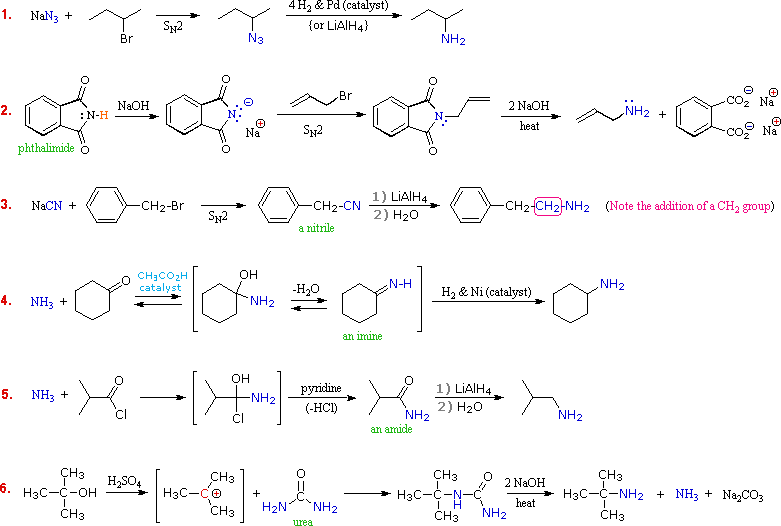
To synthesize a 1 o or 2 o amine, specific reactions are usually employed. A more efficient method starts with an S N 2 reaction between a 1 o or 2 o alkyl halide and the nucleophilic azide anion (N 3-) to produce an alkyl azide. The alkyl azide is not nucleophilic so it cannot react with additional alkyl halide to produce overalkylation.

Amine Synthesis Reactions Organic Chemistry

Amine Synthesis Reactions

Amines 4. Reactions with acids to form Salts.
|
Reactions of Amines
Reactions of Amines 1 Reaction as a proton base (Section 19-5 and 19-6) R N H H N H H R H X H-X (proton acid) NaOH amine base ammonium salt |
|
Chapter 6 Amines and Amides - Angelo State University
Examples: Basicity of Amines • Complete the following reactions: CH3NH2 + HCl NHCH3 + HCl N CH3 CH3 H CH3 + NaOH |
|
SUMMARY OF ORGANIC REACTIONS - Physics & Maths Tutor
equation: R-COOH(aq) + NaOH(aq) ? R-COO-Na+(aq) + H2O(l) b) carboxylic acids with sodium d) with primary amines (to make N-substituted amides) |
|
AMINES - Knockhardy
+Cl¯(aq) + NaOH(aq) ——> C6H5NH2(l) + NaCl(aq) + H2O(l) 2 Amines Due to their lone pair amines react as nucleophiles with • haloalkanes |
|
AMINES
An amine on reaction with benzenesulphonyl The purpose of NaOH in the reaction is : NaOH (3) primary amine isonitrile compound conc HCl |
|
Reactions of Amines
NaOH amine base ammonium salt (acidic) • Mechanism: Required (protonation) Reaction with Ketones or Aldehydes (Section 18-16,17 and 19-10) R' R O |
|
Reactions of Amines: Reductive Amination (Borch Reaction
Reactions of Amines: Reductive Amination (Borch Reaction) When ketone or aldehydes react with 1° amines an imine is formed 1) 2) neutralization, NaOH |
|
Les Amines - AC Nancy Metz
alkylation réductive Réact d'Eschweil Réduction Page 2 sur 31 Les Amines Les amines tertiaires donnent des réactions en milieu basique (NaOH) |
|
THE REACTION OF QUATERNARY AMMONIUM - SMARTech
The volatile amines from the reaction reaction the yields of amines were trimethylamine 14 2 , The addition of concentrated NaOH to the product of the |
|
Aniline par réduction du nitrobenzène I Introduction
Cette réaction est une réduction, et l'étain (Sn) jouera le rôle substitution en position méta, contrairement au groupe amine (-NH2) qui lui est contraire est tout à fait possible La réaction est la suivante : NO2 NH2 1)Sn/HCl 2)NaOH 1 |
|
Amines
Dissolves in NaOH due to acidic H-attached to Nitrogen) Secondary amine Tertiary amine : Tertiary amines do not react with Hinsberg's reagent a) Hofmann's |
|
TRICHLOROETHYLFNE IN ORGANIC SYNTHESIS: II REACTION
Abstract - In a novel reaction, trichloroethylene reacts with secondary aliphatic amines in the presence of aqueous solution of NaOH and catalytic quantity of |
|
Identifying an Unknown Compound by Solubility, Functional Group
The solubility of carboxylic acids and amines is so characteristic that solubility These functional groups typically react with H2SO4 to form new compounds |