amine reactions pdf
|
Chapter 17: Amines and Amides
Reactions of amines All amines whether soluble or insoluble in water react quantitatively with strong acids to form water-soluble salts 17 9 Heterocyclic Amines A heterocyclic amine is an organic compound in which nitrogen atoms of amine groups are part of either an aromatic or a nonaromatic ring system |
|
Organic Chemistry II / CHEM 252 Chapter 20 – Amines
Reactions of Amines 31 • Reactions of Amines with Nitrous Acid • Nitrous acid (HONO) is prepared in situ by reaction of sodium nitrite with a strong aqueous acid • Primary amines undergo diazotization with nitrous acid – The unstable diazonium salts decompose to form carbocations |
|
Ch 06 Amines and Amides
• Learn to recognize the amine and amide functional groups • Learn the IUPAC system for naming amines and amides • Learn the important physical properties of the amines and amides • Learn the major chemical reactions of amines and amides and learn how to predict the products of amide synthesis and hydrolysis reactions |
How do amines react with a nucleophile?
Step 1: Reaction of a nucleophile with an electrophile to form a new covalent bond. • All amines, whether soluble or insoluble in water, react quantitatively with strong acids to form water‐soluble salts. • Step 2: Take a proton away. Converts the amine salt to a free amine.
How amine reacts with alkyl halide?
• Reaction of an amine with an alkyl halide can be used to form a new covalent bond. Step 1: Reaction of a nucleophile with an electrophile to form a new covalent bond. • All amines, whether soluble or insoluble in water, react quantitatively with strong acids to form water‐soluble salts.
Why is amine a basic molecule?
Its basicity is due to the resonance delocalization of the positive charge over the three nitrogen atoms. • Reaction of an amine with an alkyl halide can be used to form a new covalent bond. Step 1: Reaction of a nucleophile with an electrophile to form a new covalent bond.

Amine Synthesis Reactions Organic Chemistry

Amine Synthesis Reactions

Imine and Enamine Formation Reactions With Reductive Amination
|
Reactions of Amines
From 1º Alkyl Halides: Alkylation of Ammonia (Section 19-12 19-21A) (See reaction 3). R. Br excess NH3. R. NH2. • Access: 1º Amines only. |
|
Chapter 6 Amines and Amides
Learn the major chemical reactions of amines and amides and learn how to predict the products of amide synthesis and hydrolysis reactions. |
|
Amine Transaminases in Multi-Step One-Pot Reactions
Front Cover: Graphic interpretation of how amine transaminases (ATAs) can be combined with other enzymes and metal catalysts in one reaction pot to perform |
|
19.11 REACTIONS OF ALDEHYDES AND KETONES WITH AMINES
REACTIONS OF ALDEHYDES AND KETONES WITH AMINES. A. Reaction with Primary Amines and. Other Monosubstituted Derivatives of Ammonia. A primary amine is an |
|
Assessing the Potential for the Reactions of Epoxides with Amines
18-09-2015 amine?epoxide reactions to occur at a wider range of SOA pH ... (PDF). ? AUTHOR INFORMATION. Corresponding Author. |
|
Chapter 19: Amines
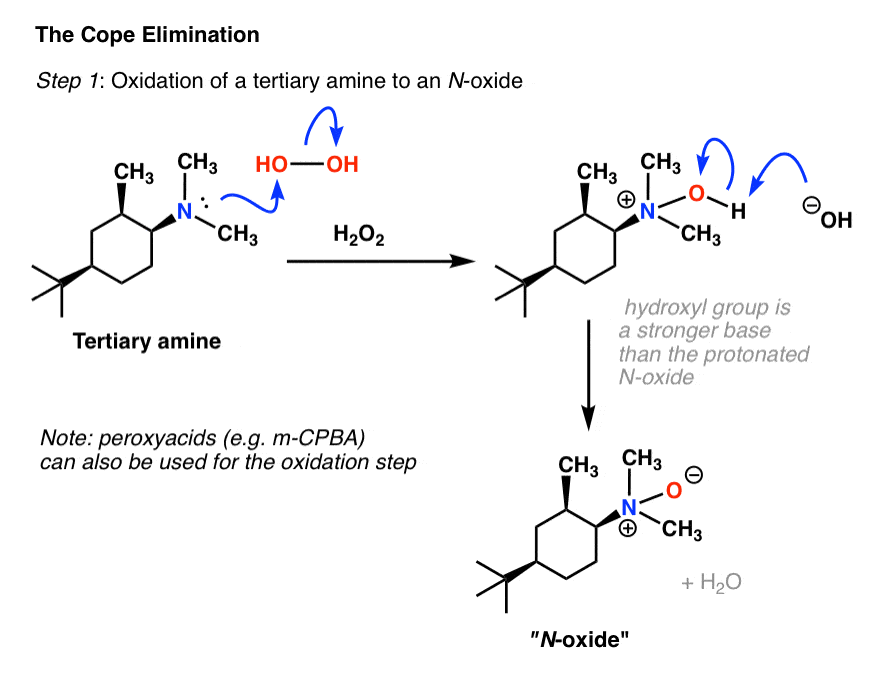
Reaction of quaternary ammonium salt in the presence of a base gives alkenes following the. Hofmann elimination mechanism. The least substituted alkene is |
|
Sch_206-amines.pdf
The unshared electron pair in cyclohexylamine is localized on nitrogen less strongly held |
|
Reactions of Amines
From 1º Alkyl Halides: Alkylation of Ammonia (Section 19-12 19-21A) (See reaction 3). R. Br excess NH3. R. NH2. • Access: 1º Amines only. |
|
Study on Reaction y Mechanisms of Amine Absorbent and Aldehydes
08-09-2015 Qualitative analysis of reaction products. • Results and Discussions. ? Reaction mechanism of amines and aldehydes. • Primary amine. |
|
INSTRUCTIONS - NHS and Sulfo-NHS
NHS is not required for carbodiimide reactions their use greatly enhances Reaction of Sulfo-NHS-activated molecules with primary amines is most. |
|
Reactions of Amines
From 1º Alkyl Halides: Alkylation of Ammonia (Section 19-12, 19-21A) (See reaction 3) R Br excess NH3 R NH2 • Access: 1º Amines only |
|
Organic Chemistry II / CHEM 252 Chapter 20 – Amines
Department of Chemistry University of Massachusetts by designating the organic groups separately in front of the word amine • In systematic nomenclature |
|
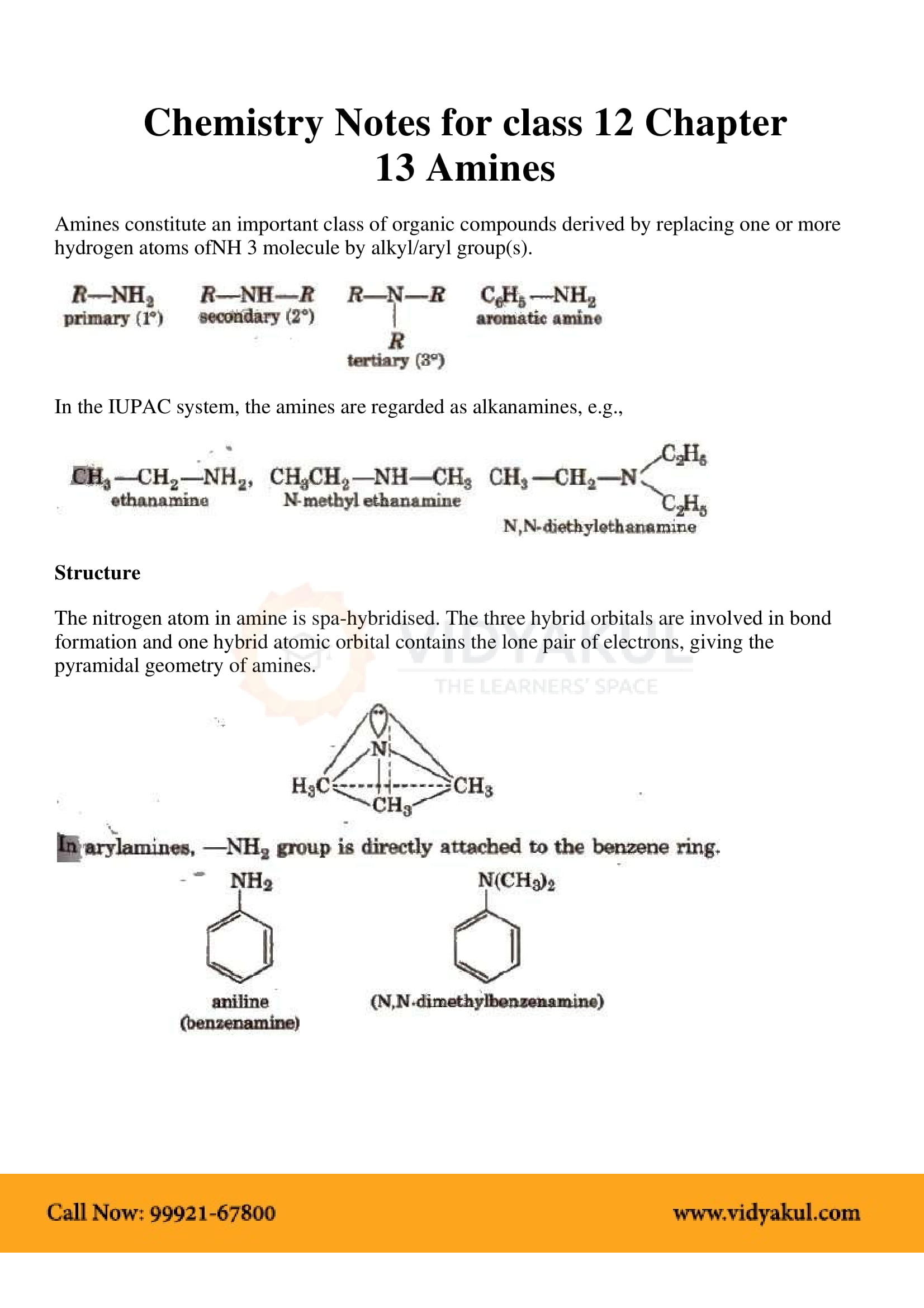
Amines Amines - NCERT
In this degradation reaction, migration of an alkyl or aryl group takes place from carbonyl carbon of the amide to the nitrogen atom The amine so formed contains |
|
Amines
Synthesis of Amines Nucleophilic Substitution Reactions: Direct Alkylation Method The nucleophilic substitution of alkyl halides with ammonia is a general |
|
Amines - UNF3S
comportement basique comportement nucléophile réaction avec les acides comportement acide des H portés par le N (amines I et II) réaction avec les bases |
|
Les amines - Remedeorg
Le produit obtenu avec une amine secondaire est effectivement soluble en milieu acide et basique et cette réaction permet bien de connaître la classe de l'amine |
|
AMINES
The unshared pair of electrons on the nitrogen atom of an amine dominates the chemistry of amines and is responsible for the basicity (sharing their lone pair of |
|
Chapter 17: Amines and Amides
aromatic amines and the simplest aromatic amine is the aniline are important in medicinal, agricultural, food, and industrial chemistry, as well as in the |
|
Chapter 9 Lecture Notes: Carboxylic Acids, Amines, and Amides
Given the structure of a carboxylic acid, carboxylate ion, ester, amide, or amine Predict the products for the reactions of amines with water or with strong acids |
|
AMINES
29 mar 2020 · When reaction is carried out with ammonia , the product is primary amine, with primary amine the product is secondary amine and with secondary |



![PDF] An Insight of the Reactions of Amines with PDF] An Insight of the Reactions of Amines with](https://www.vedantu.com/content-images/revision-notes/cbse-class-12-chemistry-notes-chapter-13-amines/6.png)


















![Reaction of butadiyne with amines - [PDF Document] Reaction of butadiyne with amines - [PDF Document]](http://dosingpdf.com/images/pdf/syncozymes.com1-1_3.png)
![Deracemisation of [alpha]-chiral primary amines by a one-pot two Deracemisation of [alpha]-chiral primary amines by a one-pot two](https://demo.vdocuments.mx/img/742x1000/reader018/reader/2020012309/5b34ced57f8b9a7e4b8c8257/r-1.jpg?t\u003d1604668026)









![A Summary of the Reactions of Aldehydes With Amines - [PDF Document] A Summary of the Reactions of Aldehydes With Amines - [PDF Document]](https://upload.wikimedia.org/wikipedia/commons/thumb/7/7a/Carbylamine_mechanism.png/400px-Carbylamine_mechanism.png)

![PDF) RuCl(η 5-C 5H 5)(PPh 3) 2] as catalyst in the reaction of PDF) RuCl(η 5-C 5H 5)(PPh 3) 2] as catalyst in the reaction of](https://d2cbg94ubxgsnp.cloudfront.net/Pictures/480xany/1/9/1/511191_full_text__41929_2020_564_onlinepdf_12_730420.png)