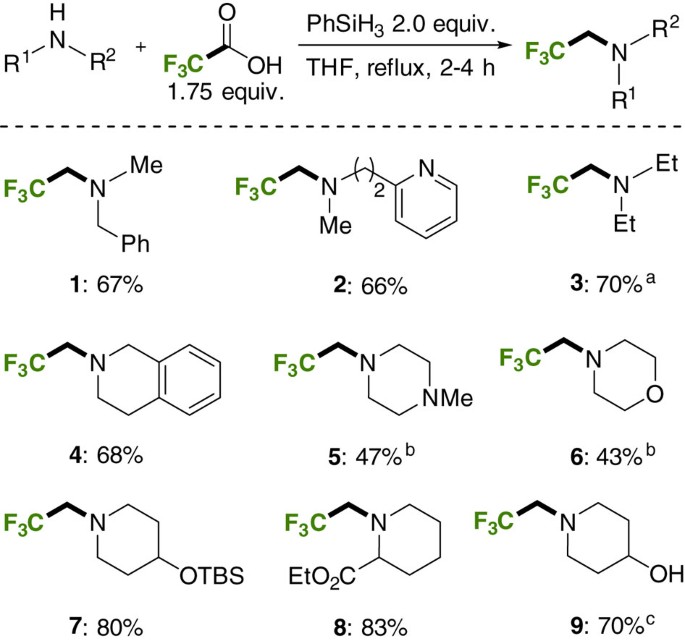
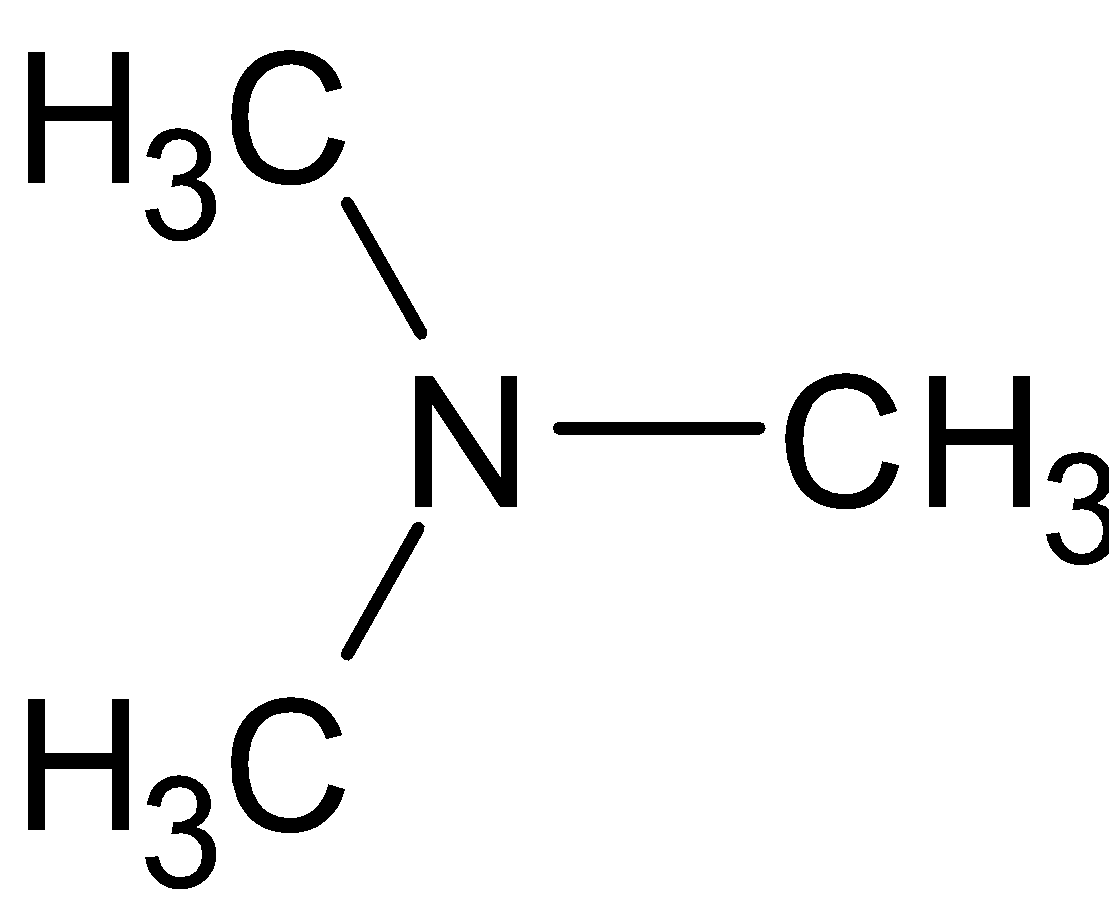
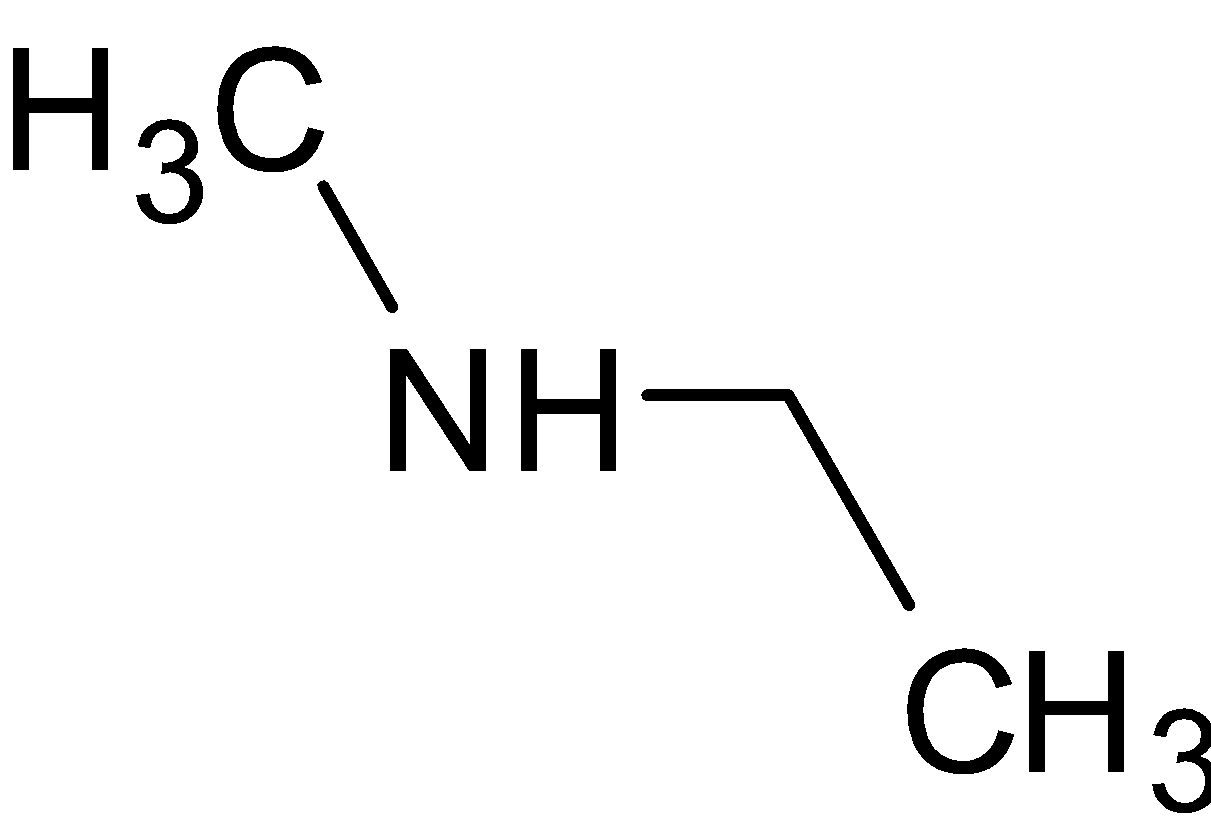
amine salt + naoh
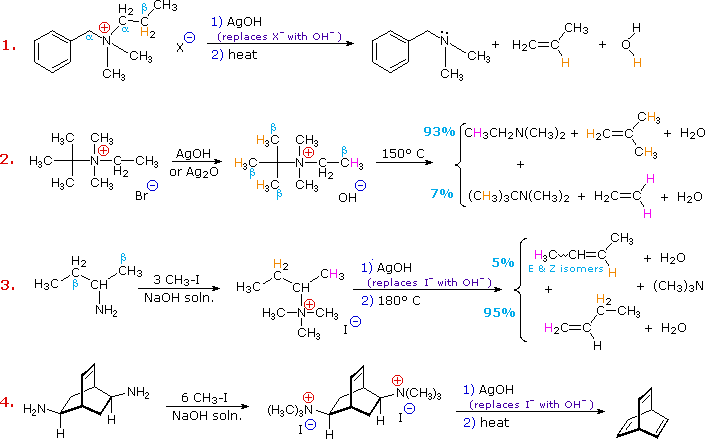
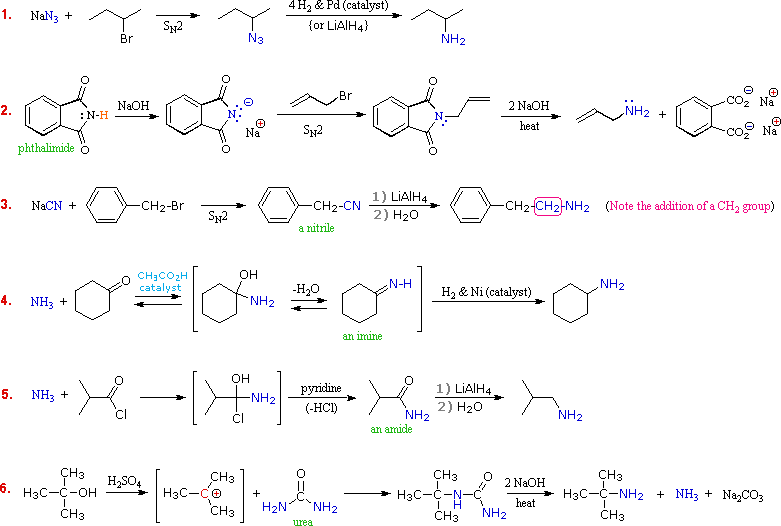
Can 3O amines be alkylated to form quaternary 4O ammonium salts?
Even 3º-amines may be alkylated to form quaternary (4º) ammonium salts. When tetraalkyl ammonium salts are desired, as shown in the following example, Hünig's base (N,N-diisopropylethylamine) may be used to scavenge the HI produced in the three S N 2 reactions. Steric hindrance prevents this 3º-amine (Hünig's base) from being methylated.
How amines are soluble in water?
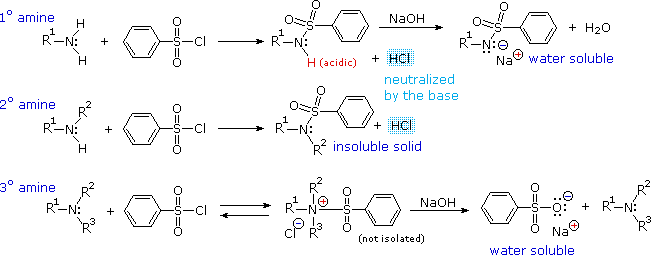
For amines one can take advantage of their basicity by forming the protonated salt (RNH 2+ Cl − ), which is soluble in water. The salt will extract into the aqueous phase leaving behind neutral compounds in the non-aqueous phase. The aqueous layer is then treated with a base (NaOH) to regenerate the amine and NaCl.
Is amine salt polar or polar?
As a result, the amine salt functional group is highly polar, with a positive charge on the nitrogen that is balanced by the negative charge from the anion of the acid. In the case of hydrochloric acid, this is the chloride ion, Cl –. Recall that, in a classical acid-base reaction, one of the products is called a salt (5).

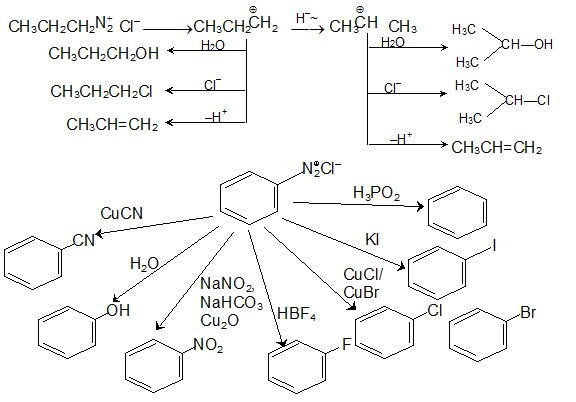
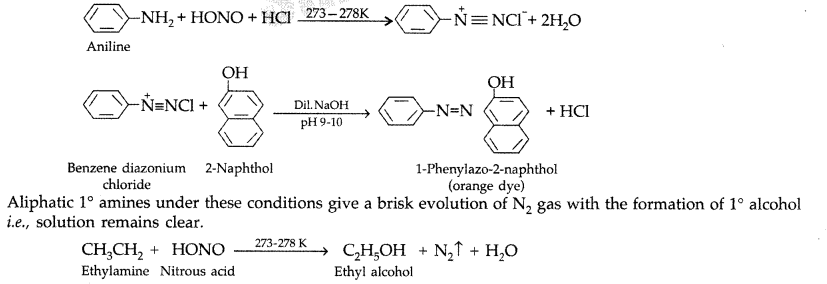
(L-20) Amine reaction with HNO2 Diazonium Salt Formation with Mechanism by Arvind Arora

Amine and HCl

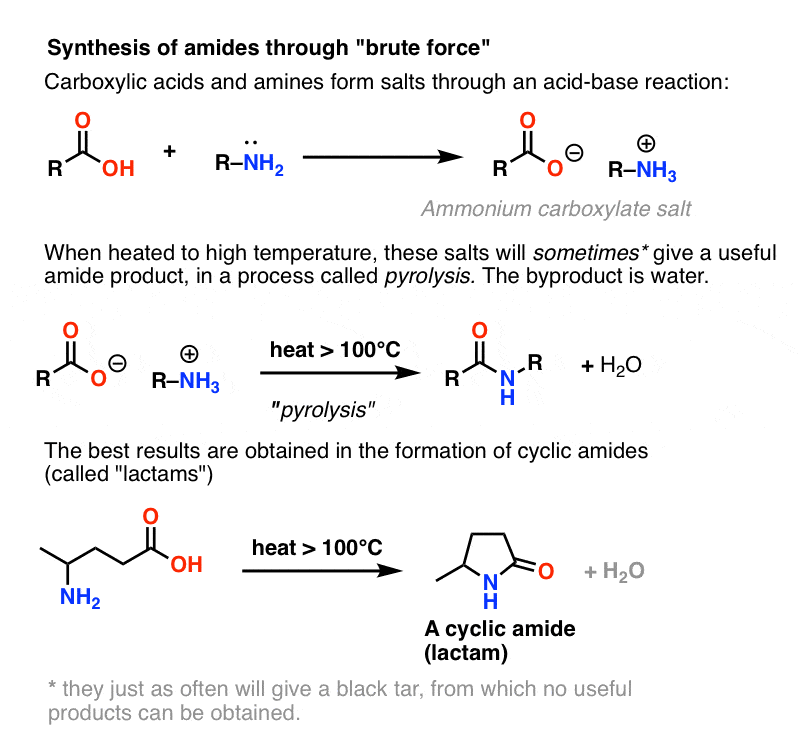
Amines 4. Reactions with acids to form Salts.
|
Reactions of Amines
H-X (proton acid). NaOH amine base ammonium salt. (acidic) Amines are completely converted to ammonium salts by acids. • Ammonium salts are completely ... |
|
Chapter 6 Amines and Amides
Conversion of an alkylammonium salt back to an amine. R—NH3. +. Cl. -. + NaOH -? R— |
|
THE STRENGTH OF TRIMETHYLAMINE OXIDE AND TRIMETHYL
Meisenheimer2 describes the amine oxides as “very weak bases much weaker than ammonia” and the alkoxyl ammonium salts as “colorless |
|
Dissolving Carboxylic Acids and Primary Amines on the Overhead
9 mars 2010 soluble in water are dissolved by the addition of aqueous NaOH. (or HCl). ... A small volume of a carboxylic acid (or amine) along with a. |
|
Adopted from pdflib image sample (C)
AMINE SALT SOLUTIONS CONTAINING 10 MOLAR NaOH. SOLUBILITY OF QUARTZ AT ROOM TEMPERATURE. ADSORPTION OF CHLORIDE IONS FROM AMINE SALT SOLUTIONS. |
|
AMINE BUFFERS FOR p H CONTROL
Buffer solutions consisting of ammonium salts tion of sodium hydroxide that is added to increase the pH will dilute the solu-. |
|
Chapter 5 Carboxylic Acids and Esters
Carboxylate Salts. • Carboxylic acids react with strong bases such as sodium hydroxide (NaOH) and potassium hydroxide. (KOH) to produce carboxylate salts. |
|
MCPA (257) First draft prepared by Dr. Yibing He Department of
second base hydrolysis over 2 nights at 70 ºC using 3N NaOH:methanol (1:1 solution |
|
N Goalby chemrevise.org 1 20 Nitrogen compounds Primary Amines
Addition of NaOH to an ammonium salt will convert it back to the amine. The ionic salts formed in this reaction means that the compounds are soluble in the |
|
Reactions of Amines
NaOH amine base ammonium salt (acidic) • Mechanism: Required (protonation) • Reverse Amines are completely converted to ammonium salts by acids |
|
THE REACTION OF QUATERNARY AMMONIUM - SMARTech
butyl-quaternary ammonium salts the rate of reaction and the methane to butane ratio with an excess of solid NaOH and the amine separated The amine was |
|
Paper Proposed for Presentation at the Laurence Reid Gas
plant data for amine systems contaminated with sodium and heat stable salt Some operators employ addition of a stronger base (typically NaOH or KOH) to |
|
Amines
Primary amine reacts with benzoyl chloride to give the acylated product c) Carbylamine is mixed with nitrous acid, an equilibrium is established among the tertiary amine, its salt, Dissolves in NaOH due to acidic H-attached to Nitrogen) |
|
Identifying an Unknown Compound by Solubility, Functional Group
Water-insoluble compounds that are soluble in 5 NaOH are then tested with 5 sodium hydrogen water-soluble amine salts, as shown in Equation 4 R N |
|
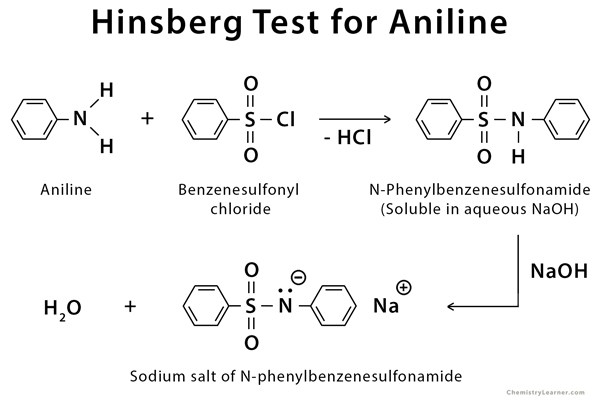
Amines Amines - NCERT
and tertiary amines, and finally quaternary ammonium salt 13 4 Preparation Amine salts on treatment with a base like NaOH, regenerate the parent amine |