amines and diazonium salts
What is diazonium salt?
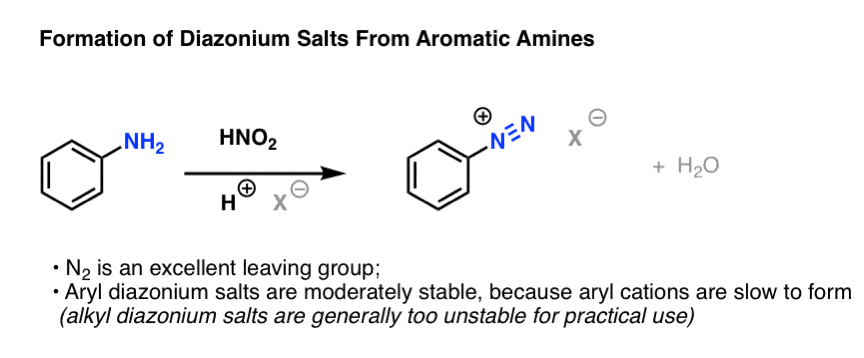
diazonium salt, any of a class of organic compounds that have the molecular structure in which R is an atomic grouping formed by removal of a hydrogen atom from an organic compound. Diazonium salts are usually prepared by the reaction (diazotization) of primary amines with nitrous acid; their most striking property is their instability.
How do nitrous acid reactions of 1o-aryl amines produce diazonium cations?
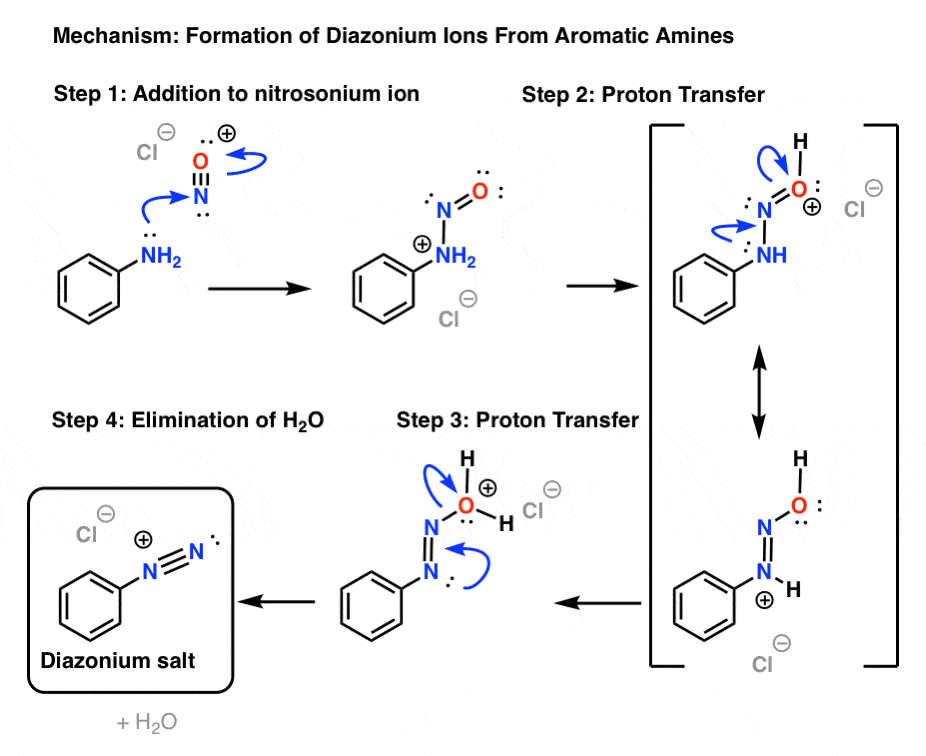
Nitrous acid reactions of 1º-aryl amines generate relatively stable diazonium species that serve as intermediates for a variety of aromatic substitution reactions. Diazonium cations may be described by resonance contributors, as in the bracketed formulas shown below. The left-hand contributor is dominant because it has greater bonding.
Why do diazonium ions lose nitrogen faster than aliphatic 1o amines?
Loss of nitrogen is slower than in aliphatic 1º-amines because the C-N bond is stronger, and aryl carbocations are comparatively unstable. Aqueous solutions of these diazonium ions have sufficient stability at 0º to 10 ºC that they may be used as intermediates in a variety of nucleophilic substitution reactions.
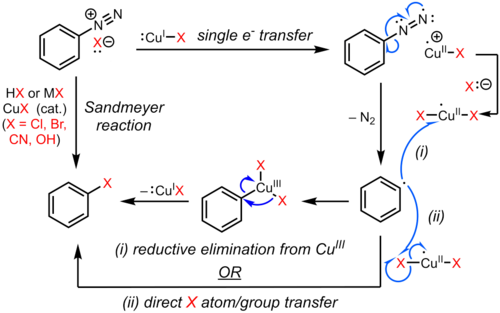
What happens if diazonium group is replaced by a copper salt?
The diazonium group can be replaced by numerous atoms or groups of atoms, often with the aid of copper or a copper salt; these reactions make possible the preparation of a wide variety of aromatic derivatives. Chemical reduction of aromatic diazonium salts leads to formation of hydrazine derivatives.

DIAZONIUM SALT in One Shot

AMINES in One Shot

Amines 12 Diazonium Salt ( Full Concept ) Preparation Physical and Chemical Properties
|
DIAZONIUM SALTS
29 ????? 2020 Diazonium compounds or diazonium salts are a group of organic compounds ... primary amine to its diazonium salt is known as diazotization. |
|
Amines Amines
Quaternary ammonium salts are used as surfactants. Diazonium salts are intermediates in the preparation of a variety of aromatic compounds including dyes. In |
|
Amines Amines
describe the method of prepara- tion of diazonium salts and their importance in the synthesis of a series of aromatic compounds including azo dyes. Objectives. |
|
Interaction of diazoimidazoles and their diazonium salts with primary
1 ??? 2002 The reactions of diazo imidazoles with primary amines are ... Unexpectedly the reaction of diazonium salt 1a with methyl- amine resulted in ... |
|
Unit: 4 : Amines
Methods of Formation of Primary Amines(alkylaryl) One way to transform diazonium salts is by treating them with various compounds of copper. |
|
Raman Characterization of Phenyl-Derivatives: From Primary Amine
Aryl-diazonium salts are widely used in organic chemistry as reactants for different syntheses. For instance the process of nitrogen elimination from diazonium. |
|
Diazotisation of aromatic amines and solvolysis of diazonium salts in
Keywords: aromatic amines diazotisation |
|
UNIT II A. Phenols* B. Aromatic Amines* C. Aromatic Acids* 10
Quaternary ammonium salts are used as surfactants. Diazonium salts are intermediates in the preparation of a variety of aromatic compounds including dyes and |
|
Kinetics and Mechanism of the Coupling of Diazonium Salts with
equilibrium with the diazonium ion. The present work4 consists of a kinetic study of the coupling of diazonium salts with aromatic amines under conditions |
|
The reduction of diazonium salts derived from aromatic primary
The reduction of diazonium salts derived from aromatic primary amines has well been known to be a convenient method for the replacement of the amino group |
|
11 Amines, Amino Acids and Diazonium Salts
Aromatic primary amines are not very soluble in water, but react with cold nitrous acid to give a soluble diazonium salt which is stable below 5°C nitrous acid |
|
From Primary Amine to Diazonium Salts - CORE
Aryl-diazonium salts are widely used in organic chemistry as reactants for different syntheses For instance, the process of nitrogen elimination from diazonium |
|
DIAZONIUM SALTS
29 mar 2020 · Synthesis of diazonium salts Treatment of primary amine with nitrous acid results in the formation of diazonium salt, a compound of the type |
|
Amines Amines - NCERT
Diazonium salts are intermediates in the preparation of a variety of aromatic compounds including dyes In this Unit, you will learn about amines and diazonium |
|
Lecture 16 Aromatic Diazonium Salts - NPTEL
C diazotization of primary aromatic amine occurs NH2 NaNO2 HCl,< 5 oC N2 Cl Mechanism |
|
Sulfonium Salts, Aliphatic and Aromatic Diazonium - ResearchGate
acid on primary amines Almost any aromatic amine can be converted to a diazonium salt in the above way Although the stability of the aromatic diazonium salts |
|
NITROGEN CONTAINING COMPOUNDS Amines - Patna Science
Primary amines react with nitrous acid (NaNO2/HCl at O°C) to form diazonium salts Alkyldiazonium salts are unstable and decompose to give a mixture of alcohol |