easl hepatitis b guidelines 2015
|
EASL 2017 Clinical Practice Guidelines on the management of
Natural history and assessment of patients with chronic HBV infection based upon HBV and liver disease markers *Persistently or intermittently °°HBV DNA |
|
EASL Clinical Practice Guidelines: Management of chronic hepatitis
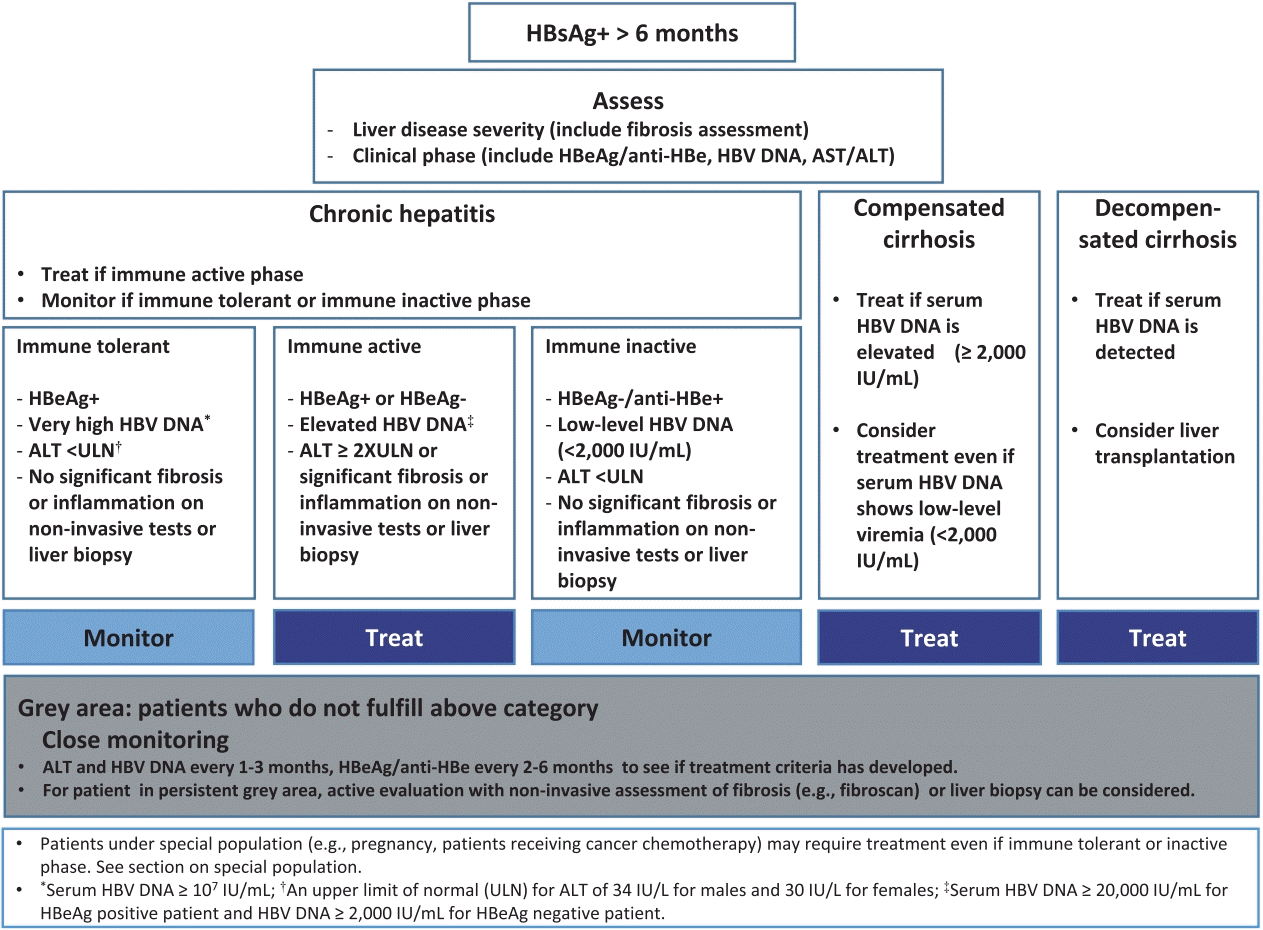
Long-term therapy with NAs HBV DNA reduction to undetectable levels by real-time PCR (i e below 10–15 IU/ml) should ideally be achieved to avoid resis- tance |
|
EASL HBV guidelines 2017
1) Goals of therapy 2) Endpoints of therapy 3) Indications for treatment 4) Monitoring of patients currently not treated 5) Treatment strategies |
|
Guidelines for the Prevention and Treatment of Chronic Hepatitis B
2 août 2023 · EASL 2017 Clinical Practice Guidelines on the Asian-Pa- cific clinical practice guidelines on the management of hepatitis B: a 2015 update |
|
The WHO guidelines for chronic hepatitis B fail to detect half
28 mar 2019 · Background Aims:In 2015 the World Health Organization (WHO) issued guidelines for the management of chronic hepati- tis B (CHB) in low- and |
What are the guidelines for Hep B in 2023?
CDC recommends screening all adults aged 18 years and older for hepatitis B at least once in their lifetime using a triple panel test.
To ensure increased access to testing, anyone who requests HBV testing should receive it regardless of disclosure of risk.How do you classify hepatitis B EASL?
Chronic HBV infection can be classified into five phases: (I) HBeAg-positive chronic infection, (II) HBeAg-positive chronic hepatitis, (III) HBeAg-negative chronic infection, (IV) HBeAg-negative chronic hepatitis and (V) HBsAg-negative phase.
What are the latest treatment guidelines for hepatitis B?
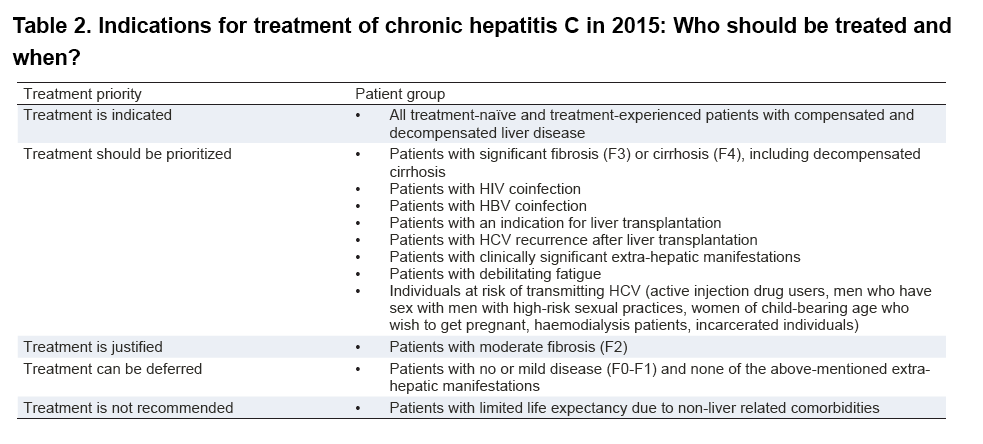
The recommendations in these guidelines promote the use of simple, non-invasive diagnostic tests to assess the stage of tiver disease and eligibility for treatment; prioritize treatment for those with most advanced liver disease and at greatest risk of mortality and recommend the preferred use of nucleos(t) ide with a
- Today, in a significant step toward viral hepatitis elimination, the Centers for Disease Control and Prevention (CDC) published CDC Recommendations for Hepatitis B Screening and Testing — United States, 2023.
The updated recommendations advise hepatitis B screening for all adults at least once in their lifetime.
|
EASL 2017 Clinical Practice Guidelines on the management of
Hepatitis B virus (HBV) infection remains a global public health problem with changing epidemiology due to several factors. |
|
EASL-ALEH Clinical Practice Guidelines: Non-invasive tests for
Journal of Hepatology 2015 vol. Received 9 April 2015; accepted 9 April 2015 ... hepatitis B and C with AUROC ranging from 0.75 to 0.84 for sig-. |
|
EASL Recommendations on Treatment of Hepatitis C 2018
The quality of the evidence in the rec- ommendations has been classified into one of three levels: high. (A) moderate (B) or low (C). The GRADE system offers |
|
ANNUAL REPORT 2015
EASL Recommendations on Treatment of Hepatitis C 2015. • EASL-ALEH Clinical Practice Guidelines: Non-invasive tests for evaluation of liver disease severity |
|
The WHO guidelines for chronic hepatitis B fail to detect half of the
28.03.2019 Although the sensitivity of the WHO 2015 criteria was low compared with the EASL 2017 guidelines the specificity was found to be high. This ... |
|
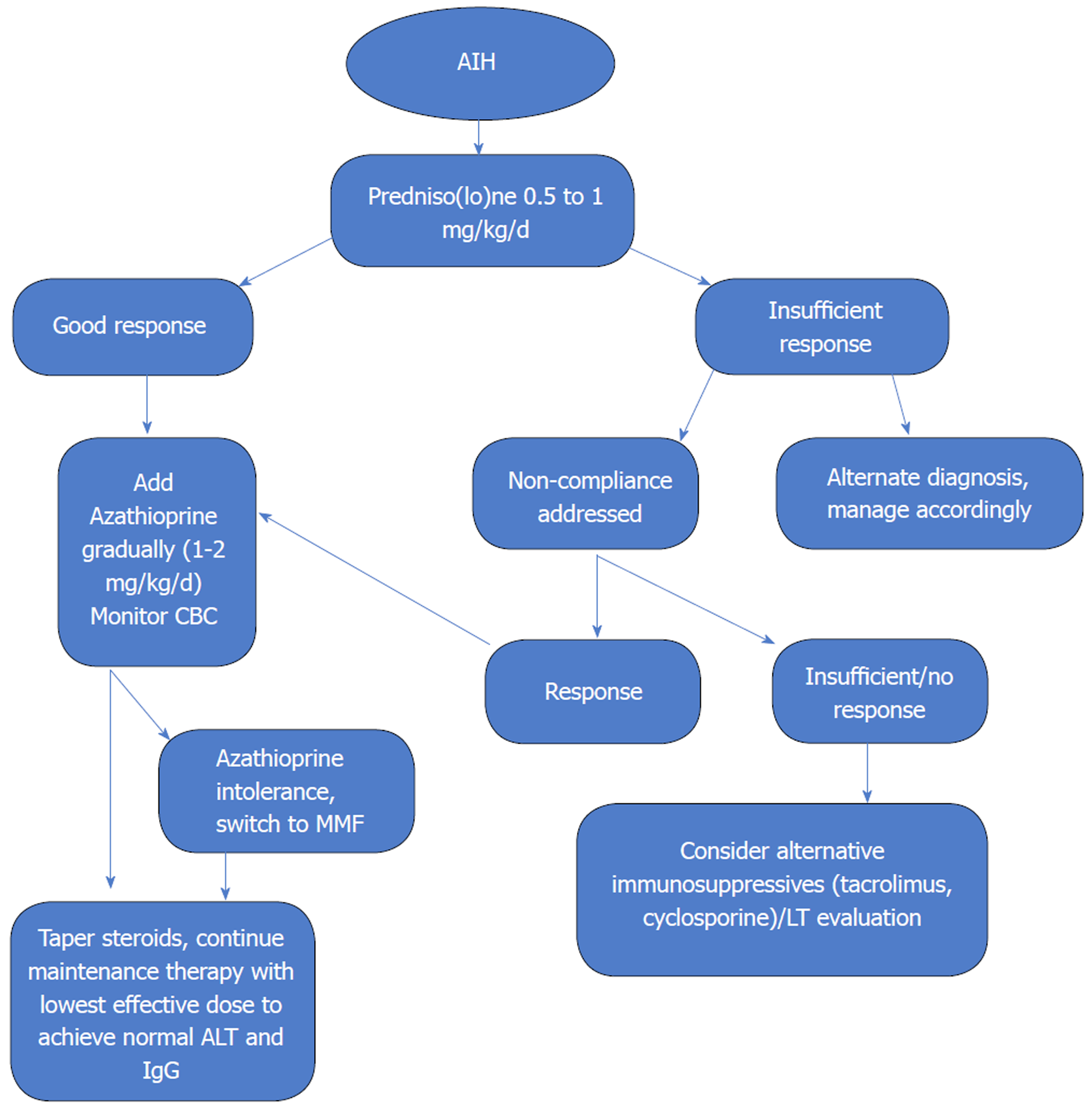
EASL Clinical Practice Guidelines: Autoimmune hepatitisq
Vaccination against hepatitis A virus and HBV should be given. Clinical Practice Guidelines. 994. Journal of Hepatology 2015 vol. 63 971–1004. Page 25. to all |
|
EASL Clinical Practice Guidelines for the management of patients
guidance on the management of decompensated cirrhosis with the only purpose of improving hepatitis B virus (HBV) infection treatment with antiviral. |
|
EASL Clinical Practice Guidelines: Drug-induced liver injury
including chronic hepatitis B virus (HBV) or HCV infection as well as alcoholic liver disease and elevated liver enzymes prior to initiation of therapy were |
|
S3-Leitlinie der Deutschen Gesellschaft für Gastroenterologie
887 000 Menschen die im Jahr 2015 an einer Hepatitis B verstor- ben sind [1]. Leitlinienadaption EASL Clinical Practice Guideline HBV 2017 |
|
Guidelines - EASL Recommendations on Treatment of Hepatitis C
Also histol- ogy may be required in cases of known or suspected mixed aeti- ologies (e.g. HCV infection with HBV coinfection |
|
EASL 2017 Clinical Practice Guidelines on the management of
EASL 2017 Clinical Practice Guidelines on the management of hepatitis B virus infection q European Association for the Study of the Liver |
|
Hepatitis B EASL Guidelines - EASL-The Home of Hepatology
This EASL Guideline on Hepatitis B presents updated recommendations and knowledge for the optimal management of HBV infection |
|
Management of chronic hepatitis B virus infection - EASL
The objective of this manuscript is to update the recommendations for the opti- mal management of chronic HBV infection The CPGs do not fully address |
|
[PPT] Chronic HBV infection Chronic hepatitis B - EASL
These slides give a comprehensive overview of the EASL clinical practice guidelines on the management of hepatitis B infection |
|
The WHO guidelines for chronic hepatitis B fail to detect half - EASL
28 mar 2019 · Background Aims:In 2015 the World Health Organization (WHO) issued guidelines for the management of chronic hepati- tis B (CHB) in low- and |
|
Comparison of clinical practice guidelines for the management of
28 août 2020 · The EASL recommends the treatment of patients with HBV DNA >20000 IU/mL and ALT >2× ULN regardless of the degree of fibrosis whereas all |
|
EASL 2017 Clinical Practice Guidelines on the management of
During treatment liver function tests should be performed every 3–4 months during the first year and every six months thereafter Serum HBV DNA should be |
|
Autoimmune Hepatitis EASL Guidelines 2015 - FILFOIE
Histological demonstration of hepatitis is a prerequisite for the diagnosis of AIH » (EASL Practice Guidelines J Hepatol 2015) |
|
Hepatitis B: Who to treat? A critical review of international guidelines
20 fév 2020 · EASL only recommends antiviral prophylaxis if they are at high risk of HBV reactivation (EASL evidence level II-2 grade 1 recommendation) WHO |
|
EASL Clinical Practice Guidelines: Autoimmune hepatitisq - SNFGE
Vaccination against hepatitis A virus and HBV should be given Clinical Practice Guidelines 994 Journal of Hepatology 2015 vol 63 \ 971–1004 Page 25 to all |
What is EASL guidelines?
The EASL Clinical Practice Guidelines (CPGs) on the management of hepatic encephalopathy (HE) present evidence-based answers to a set of relevant questions (where possible, formulated in PICO [patient/population, intervention, comparison and outcomes] format) on the definition, diagnosis, differential diagnosis andWhat are the phases of hepatitis B EASL?
Chronic HBV infection can be classified into five phases: (I) HBeAg-positive chronic infec- tion, (II) HBeAg-positive chronic hepatitis, (III) HBeAg-negative chronic infection, (IV) HBeAg-negative chronic hepatitis and (V) HBsAg-negative phase.What is the Aasld guidelines for hepatitis B vaccination?
Vaccination. The following recommendations are from the 2018 AASLD guidance update : Administer HBV vaccines as a 3-dose series at 0, 1, and 6 months (± hepatitis A vaccine). Alternatively, for adults, combination hepatitis A and B vaccine (Twinrix) uses a 4-dose regimen at 0, 7, 21-30 days, and 12 months.Treatment for chronic hepatitis B may include:
Antiviral medications. Several antiviral medicines — including entecavir (Baraclude), tenofovir (Viread), lamivudine (Epivir), adefovir (Hepsera) and telbivudine — can help fight the virus and slow its ability to damage your liver. Interferon injections. Liver transplant.
| EASL 2017 Clinical Practice Guidelines on the management of |
| Hepatitis B Virus (HBV) Hepatitis B Virus |
| Guideline on the clinical investigation of hepatitis B |
| United States 2015 - Centers for Disease Control and Prevention |
| EASL Clinical Practice Guidelines: Management of chronic |
| Le d-ib td-hu va-top mxw-100p>Hep B Antiviral Treatment Info - Website For HCPs |
|
EASL Clinical Practice Guidelines: Management of chronic hepatitis
ALT levels should remain persistently within the nor- mal range according to traditional cut-off values (approximately 40IU/ml) [14] and HBV DNA should be below |
|
Non-invasive tests for evaluation of liver disease severity - EASL
EASL-ALEH Clinical Practice Guidelines: Non-invasive tests 0 93 81 95 n a Afdhal et al [129] HCV/HBV 2015 560** 66 7 8 4 0 73 58 75 70 14 8 12 8 |
|
EASL 2017 Clinical Practice Guidelines on the - ASSCAT
Hepatitis B virus (HBV) infection remains a global public health problem with changing Keywords: Hepatitis B; EASL guidelines; Treatment; Interferon; Entecavir; Tenofovir; TAF; HBsAg Clin Infect Dis 2015;61:584–592 [9] Ott JJ, Horn J, |
|
Mon titre - FILFOIE
Autoimmune Hepatitis EASL Guidelines 2015 of hepatitis is a prerequisite for the diagnosis of AIH » (EASL, Practice Guidelines, J Hepatol 2015) (Histologie: |






![PDF] Clinical Practice Guidelines EASL 2017 Clinical Practice PDF] Clinical Practice Guidelines EASL 2017 Clinical Practice](https://ars.els-cdn.com/content/image/1-s2.0-S0168827818302150-gr3.jpg)