d and f block elements iit jee notes pdf
|
D and f Block Elements IIT JEE Notes PDF
d and f block elements IIT JEE notes pdf helps students to have a quick revision before the exam These notes also help students to save a lot of time right |
How do I make short notes in IIT?
Always make notes in the Language you're more comfortable with. 2.
Never copy the text exact from book.
First read the content and then write it in your own words so it will easy to understand quickly on revision time.D block elements are the elements that can be found from the third group to the twelfth group of the modern periodic table.
The valence electrons of these elements fall under the d orbital.
D block elements are also referred to as transition elements or transition metals.
Should I make notes for IIT?
If you are one of these young aspirants preparing for the IIT JEE exam, then you should know taking notes to play an important role in the candidate's preparation for the IIT JEE exam.
It helps students to understand the major topics and chapters with ease.
So, what are you waiting for?
What are the D and f-block short notes?
In the d- blocks, the last electron enters into the d-orbital of the penultimate shell.
In the f-block elements, the electron enters into the f-orbital of anti-penultimate shell.d-block elements show a variable oxidation state, while most f-blocks show +3 oxidation state.
|
Unit 4: Chemistry of d-block elements
(a) size of atoms & ions. (b) reactivity. (c) catalytic activity. (d) oxidation state. (e) complex formation ability. (f) colour. (g) magnetic properties. |
|
5 Edition
d and f-block Elements. Topics Covered. CHEMISTRY. FOR JEE MAIN & ADVANCED IIT-JEE and AIEEE. 9000+ Problems ... Tips & Tricks Facts |
|
CLASSIFICATION OF ELEMENTS AND PERIODICITY IN
classify elements into s p |
|
S-BLOCK ELEMENTS
Hydrogen is a unique element it has only one electron in the 1s-orbital and no electrons in the (a) These hydrides are formed by d- and f-block metals |
|
Revised Syllabus to be followed from JEE (Advanced) 2023
Revised Syllabus to be followed from JEE (Advanced) 2023 Orbital overlap and covalent bond; Hybridisation involving s p and d ... f-Block Elements. |
|
Sri Chaitanya IIT Academy.India.
16 Transition elements (d- & f-block elements) Jee Main 2020(Sep)Che_Analysis. Analysis prepared by. Mr. M.Uma Shankar. National IIT Coordinator ... |
|
PERIODIC TABLE & PERIODICITY
Those elements which have partially filled d-orbitals in neutral state or in any stable oxidation state are called transition elements. (d) f-Block elements. |
|
Untitled
15-Aug-2021 D & F BLOCK ELEMENTS (NEET). BY BALADRISIR ... Dropper + 12 - IIT JEE (Target 2021) ... Plus learners will get access to in-app notes. |
|
Untitled
15-Aug-2021 D & F BLOCK ELEMENTS (NEET). BY BALADRISIR ... Dropper + 12 - IIT JEE (Target 2021) ... Plus learners will get access to in-app notes. |
|
Untitled
Subscribe to Unacademy Iconic for IIT JEE and get (ii) f>d >p> s ... (iii) For transition elements the 3d-orbitals are filled with electrons after. |
|
5 d & f BLOCK ELEMENTS - Mahesh Tutorials Science
A transition element may also be defined as the element which partially filled d- orbital in their ground state or most stable oxidation state Cu (Z = 29) = 1s2 2s2 |
|
CHEMISTRY OF D-BLOCK ELEMENTS
(a) size of atoms ions (b) reactivity (c) catalytic activity (d) oxidation state (e) complex formation ability (f) colour (g) magnetic properties |
|
For jee main & advanced - TopperLearning
d and f-block Elements Topics Covered CHEMISTRY IIT-JEE and AIEEE 9000+ Problems based on Tips Tricks, Facts, Notes, Misconceptions, Key Take |
|
Organic chemistry - Resonance
d-Block Elements their compounds 95 18 Qualitative Analysis 101 ∆H° resonance = ∆H°f, experimental – ∆H°f, calclulated = ∆H°c, calclulated – ∆H° c, |
|
Periodic Properties - Bansal Classes
Transition elements have very small volumes and therefore these are present at the bottoms of the curve Exercise 1 s, p, d and f ), depending upon the subshell to which the last electron enters (IIT JEE Previous Year's Questions) Q 1 |
|
Periodic table - This PDF is the Sample PDF taken from our
d-orbitals are called as transition elements Gold in its +3 oxidation state has electron configuration [Xe]54, 5d86s0 (D) 16th group elements like oxygen and |
|
Aman Dhattarwal - Link to all the Computer Science PDF
Dhattarwal Notes For Physics of Unacademy Plus PDF Aman Dhattarwal Wiki block elements iit jee notes pdf , d and f block elements notes for neet, d and f |








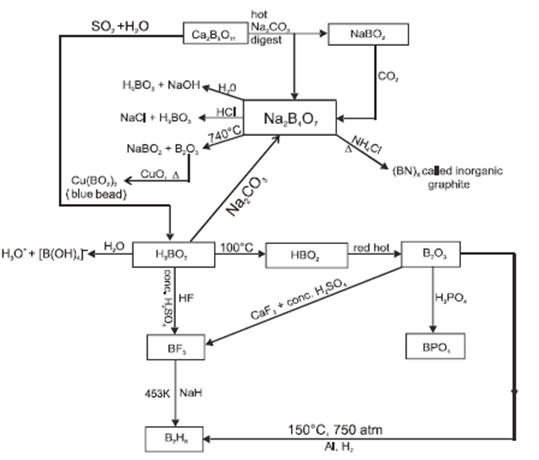





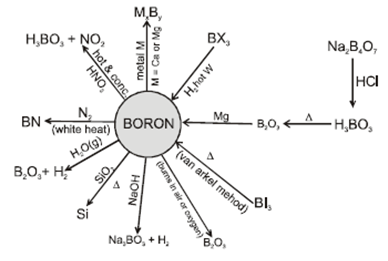








![PDF] Inorganic Chemistry Handwritten Notes For IIT JEE PDF] Inorganic Chemistry Handwritten Notes For IIT JEE](https://www.vedantu.com/content-images/revision-notes/cbse-class-12-chemistry-notes-chapter-8-the-d-and-f-block-elements/18.png)


