cu2+ hydrolysis reaction
|
1 Table S1 Hydrolysis formation constants of Zn2+ and Cu2 in NaCl
Table S1 Hydrolysis formation constants of Zn2+ and Cu2 in NaCl logβ a) Species M I = 0 15b) T = 298 15 K I = 0 5b) T = 298 15 K I = 1b) T = 298 15 K I |
What is the equation for the hydrolysis reaction?
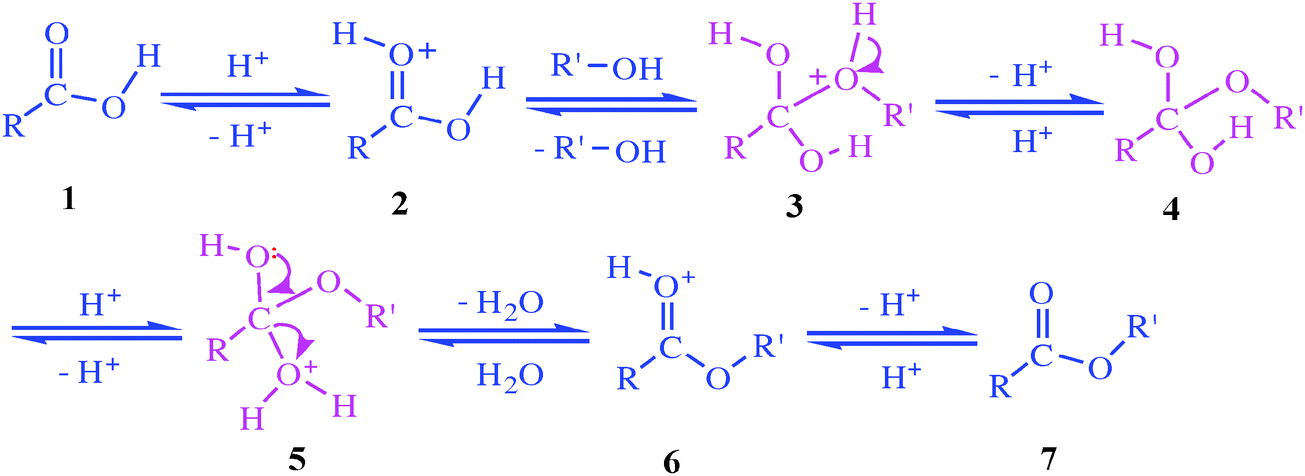
Thus, if a compound is represented by the formula AB in which A and B are atoms or groups and water is represented by the formula HOH, the hydrolysis reaction may be represented by the reversible chemical equation AB + HOH ⇌ AH + BOH.
Copper sulphate hydrolyse in water to give cupric hydroxide which is weak base and sulphuric acid which is strong acid.
So hydrogen ion is more than hydroxyl ion in water.As a result the solution is acidic.
Copper sulphate is a salt of a strong acid, H2SO4, and a weak base, Cu(OH)2.
What is the hydrolysis reaction of CuCl2?
ABSTRACT: Hydrolysis of CuCl2 is the water splitting step of the Cu−Cl thermochemical cycle, where CuCl2 reacts with steam to produce Cu2OCl2 and HCl.
In the present work, this gas−solid reaction was investigated to understand the mechanism and kinetics.
Does copper undergo hydrolysis?
Copper ions Cu2+ from copper chloride CuCl2 undergoes hydrolysis when dissolved in water to produce a slightly acidic solution.
Hydrolysis alters the pH of the solution.
|
Catalytic Hydrolysis of Esters of 2-Hydroxypyridine Derivatives for
20 feb 2008 of catalytic amounts of Cu2+; 55 turnovers of hydrolysis are observed for the best substrate acetic acid 2-hydroxypyridine ester. |
|
Reduction of Cu2+ in mine waters by hydrolysis of ferrous sheet
descending Cu2+-bearing meteoric waters with smectite fillings in thin fractures. of the zoned copper mineral sequences is the hydrolysis of smectite in ... |
|
Catalytic Hydrolysis of Esters of 2-Hydroxypyridine Derivatives for
20 feb 2008 of catalytic amounts of Cu2+; 55 turnovers of hydrolysis are observed for the best substrate acetic acid 2-hydroxypyridine ester. |
|
Hydrolysis of Cu(II)1
24 mar 1976 Potentiometric titration of Cu2+ [Cu = 1.5 x 1O-s M |
|
Unraveling the Hydrolysis of Z2Cu2+ to ZCu2+(OH)? and Its
3 sept 2021 Here we propose a scheme involving the hydrolysis of ... reaction (transient CO oxidation) |
|
Phosphonate Ester Hydrolysis Catalyzed by Two Lanthanum Ions
model compound and investigated in terms of catalysis of hydrolysis by two metal ions in concert. The divalent metal ions Zn2+ |
|
A highly selective fluorescent probe for the sensing of Cu2+ based
A highly selective OFF–ON fluorescent probe is developed for the sensing of Cu2+ based on the hydrolysis of a quinoline-. 2-carboxylate moiety. |
|
Computational Study of Copper (II) Complexation and Hydrolysis in
Using two full hydration shells around the Cu2+ ion. (18 ligands) gives Gibbs free energies of aqueous reactions that are in excellent agreement with experiment |
|
Rate Enhancing Multi-site Interactions in Ester Hydrolysis Catalyzed
Summary The Cu2+ complexes of sutfactant imidazole ligands having a hydroxyl and a cationic or an anionic polar head groups |
|
1 Table S1 Hydrolysis formation constants of Zn2+ and Cu2 in NaCl
Mesmer R. E. |
|
Reduction of Cu2+ in mine waters by hydrolysis - GeoScienceWorld
In an early stage of reaction, the pH-increase resulting from smectite hydrolysis, together with a sufficiently high pco,, produces the malachite (Cu2(OH)2CO3) |
|
Copper(II) ion hydrolysis in aqueous solution
than 10 -sm and with pH less than 8 5, the hydrolysis of copper(II) ions can be characterized by the following reaction: Cu 2+ + nH20 ~ Cu(OH) 2-~ +nH + |
|
Adsorption and desorption of copper(II) - CORE
adsorption of Cu2 ions on the adsorbent was found to be dependent on the initial time and pH, the The hydrolysis of SCAM-1 in the aqueous solu- tion can be |
|
Formation of nanoparticles in water-in-oil microemulsions controlled
play a key role in the radiolytic reduction of Cu2+ of Cu2+ in microemulsion and clarify the mechanism, which will favor ated through the hydrolysis of Cu + |
|
1 Table S1 Hydrolysis formation constants of Zn2+ and Cu2 in NaCl
Table S1 Hydrolysis formation constants of Zn2+ and Cu2 in NaCl logβ a) Species M I = 0 15b) T = 298 15 K I = 0 5b) T = 298 15 K I = 1b) T = 298 15 K |