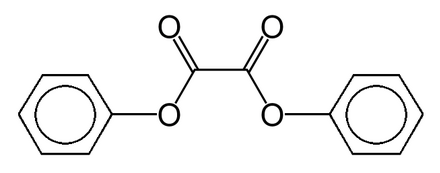
diphenyl oxalate and hydrogen peroxide
Diphenyl oxalate is commonly used in the production of glow sticks, which are novelty items that emit colorful light.
The chemical reaction between diphenyl oxalate and hydrogen peroxide produces energy that excites a fluorescent dye, causing it to emit light.
How do you make diphenyl oxalate?
The process for synthesizing diphenyl oxalate involves heating a mixture of oxalic acid, phenol, and sulfuric acid in a solvent.
What happens when hydrogen peroxide reacts with phenyl oxalate ester?
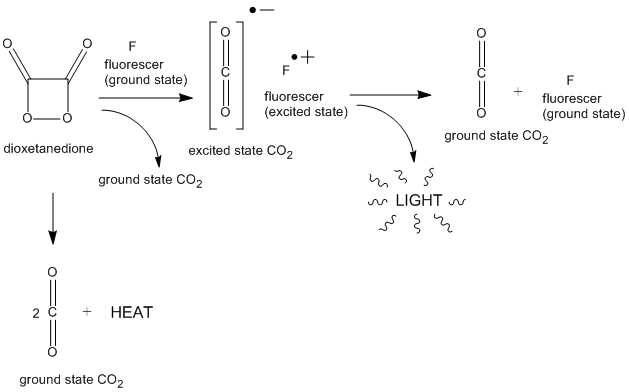
Glow sticks light up when oxalate esters react with hydrogen peroxide to form a high-energy intermediate (possibly 1,2-dioxetanedione).
This intermediate reacts with dye, which moves to an excited state (indicated with *) and then releases light as it relaxes.19 oct. 2021
What chemicals react with hydrogen peroxide?
Hydrogen peroxide can be derivatised to form compounds capable of acting as a source of active oxygen by reacting with many compounds such as borates, pyrophosphates, carbonates, sulphates, silicates and organic compounds such as carboxylates and amides.
|
Destruction of oxalate by reaction with hydrogen peroxide
of oxidation of oxalate by hydrogen peroxide in acid solution at the reElux temperature was adequate for process application; reaction half-times at 100°C |
|
Chemiluminescent PEG-PCL micelles for imaging hydrogen peroxide
trast agents that can detect hydrogen peroxide in vitro and in vivo. Diphenyl oxalate was purchased from TCI America (Port- land OR). |
|
Survey and health assessment of glow sticks
oxalate ester and peroxide (activator) typically hydrogen peroxide. An information retrieval identified 18 known diphenyl oxalate esters that are used ... |
|
Chemiluminescence from reactions of electronegatively substituted
Abstract: Reactions of a series of oxalate esters with hydrogen peroxide and fluorescent compounds were examined for chemiluminescent light emission. |
|
Determination of Hydrogen Peroxide by Flow Injection Analysis with
ANALYSIS WITH ARYL OXALATE-SULFORHODAMINE 101. CHEMILUMINESCENCE determination of hydrogen peroxide by aryl oxalate ... Perylenel# 4 1 |
|
Laser-Induced Fluorescence of Lightsticks
1999. 1. 1. in a lightstick involves the oxidation of diphenyl oxalate ester by hydrogen peroxide: O. C. C. O. O. O. H2O2. |
|
CHEMILUMINESCENT MATERIALS
oxalate esters with hydrogen peroxide in the presence of such fluorescent compounds as 910-diphenylanthracene (DPA) or rubrene have been shown. |
|
Detection of Hydrogen Peroxide in vitro and in vivo Using
2011. 5. 10. Diphenyl peroxalate-encapsulated micelles could detect hydrogen peroxide ... oxalate was known to accelerate addition of hydrogen per-. |
|
Determination of absolute chemiluminescence quantum yields for
oxalate (PCPO) hydrogen peroxide (H202) and 9: 10 diphenyl anthracene (DPA) have been determined. A fully corrected chemiluminescence monitoring |
|
CHEM 341 Problem Set 20 Halloween Edition When confronted with
Light sticks work by the slow reaction of hydrogen peroxide with H. O. O. O. O. (dioxetane). (diphenyl oxalate). (hydrogen peroxide). |
|
Chemiluminescence Whats inside of a light stick anyway? - MIT
vial Solution 2 is H2O2 The chemicals inside of the plastic light stick that were poured out into Solution 1 consists of Cyalume a Phenyl Oxalate Ester plus a |
|
1 Cyalume Experiment
hydrogen peroxide When you bend the light stick, the hydrogen peroxide vial breaks, the hydrogen peroxide Bis(2,4,6-trichlorophenyl)oxalate (A cyalume) |
|
Chemiluminescence Whats inside of a light stick anyway? - MIT
vial Solution 2 is H2O2 The chemicals inside of the plastic light stick that were poured out into Solution 1 consists of Cyalume a Phenyl Oxalate Ester plus a |
|
Red Orange Yellow Green Blue How do glow sticks produce light?
releasing hydrogen peroxide solution This then reacts with a diphenyl oxalate, producing 1,2-dioxetanedione; this product is unstable, decomposes to carbon |
|
Chemiluminescence
Diphenyl oxalate (Cyalume®) is not by itself chemiluminescent, but after oxida- O2 + fluorophore* Cyalume unstable peroxide h*ν firefly luciferin oxyluciferin |
|
Career: Environmental Chemist The Chemistry of - Nautilus Live
dissecting the glow stick, use store-bought hydrogen peroxide to fill a shallow glass ampule are a mixture of a dye and diphenyl oxalate, a chemical derived |










![PDF] Detection of hydrogen peroxide with chemiluminescent micelles PDF] Detection of hydrogen peroxide with chemiluminescent micelles](https://sciencenotes.org/wp-content/uploads/2020/10/glow-stick-experiment.jpg)




