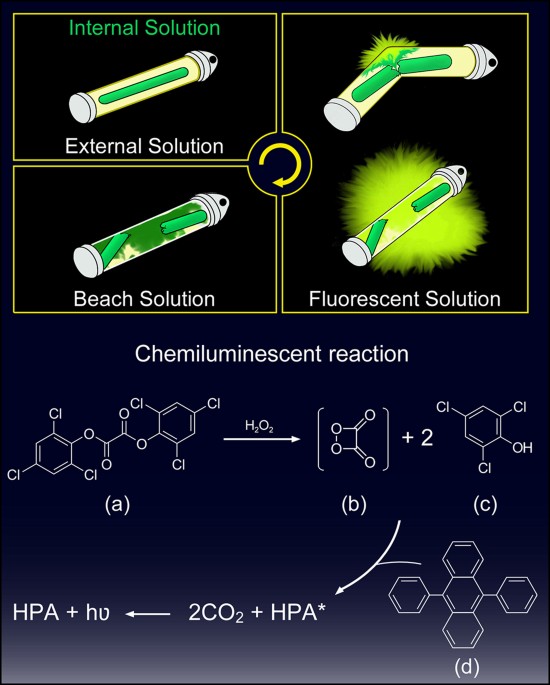
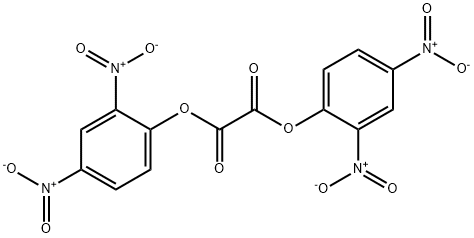
diphenyl oxalate chemiluminescence
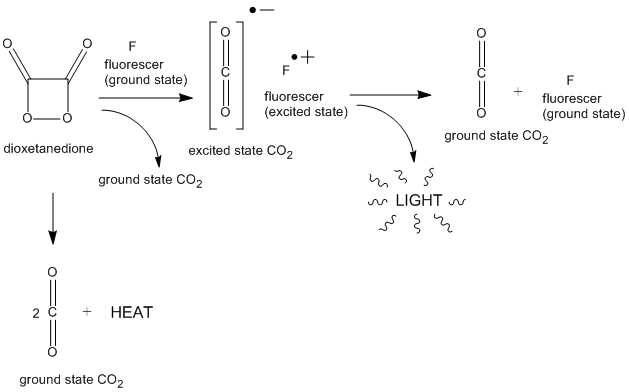
Glow sticks light up when oxalate esters react with hydrogen peroxide to form a high-energy intermediate (possibly 1,2-dioxetanedione).
How do you synthesize diphenyl oxalate?
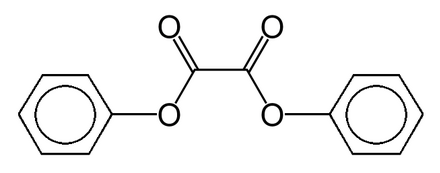
Diphenyl oxalate (trademark name Cyalume) is a liquid ester whose oxidation products are responsible for the chemiluminescence in a glowstick.
It can be synthesized by fully esterifying phenol with oxalic acid.
What is the mechanism of Cyalume?
Cyalume light sticks are based on the reaction of bis(2,4,5-trichlorophenyl-6-carbopentoxyphenyl)oxalate (CPPO) with hydrogen peroxide.
The hydrogen peroxide causes the CPPO to decompose to carbon dioxide, releasing energy that excites the dye, which then de-excites by releasing a photon.
|
CHEMILUMINESCENT MATERIALS - DTIC
Efforts to prepare improved chemiluminescent oxalic acid derivatives and improved fluorescers are described The effects of oxalate structure fluorescer |
|
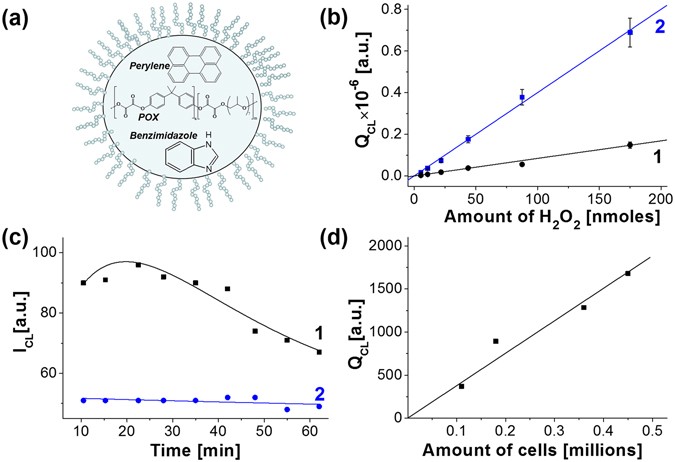
Chemiluminescent PEG-PCL micelles for imaging hydrogen peroxide
Key words: hydrogen peroxide; chemiluminescence; PEG- PCL; micelles; imaging Diphenyl oxalate was purchased from TCI America (Port- land OR) |
|
Effect of some surfactants on the chemiluminescent reactions of bis
Key words: TCPO 2-NPO surfactant chemiluminescence hydrogen peroxide Résumé : On a étudié les réactions chimiluminescentes de l'oxalate de bis(24 |
|
Chemiluminescence Reagents
Cypridina luciferin analogs achieve chemiluminescence On the other hand oxalates are oxidized by hydrogen peroxide 4-(45-Diphenyl- |
|
United States Patent Office
chemiluminescent reaction employing oxalyl chloride may ditions can unexpectedly provide chemiluminescence diphenyl oxalate II ---------- |
|
Chemiluminescence: Synthesis of Cyalume
Chemiluminescence: Synthesis of Cyalume and Making it Glow Intro Chemiluminescence is the process Bis(246-trichlorophenyl)oxalate (A cyalume) |
|
Chemiluminescence Whats inside of a light stick anyway? - MIT
poured out into Solution 1 consists of Cyalume a Phenyl Oxalate Ester plus a fluorescent dye molecule When the H2O2 in Solution 2 is poured into Solution 1 a |
|
Lightsticks - Chemiluminescence - NC State University
two mixtures react to produce a chemiluminescent reaction The overall reaction involves forming phenol and CO2 from the phenyl oxalate ester and H2O2 (as |
|
Chemiluminescence Whats inside of a light stick anyway? - MIT
poured out into Solution 1 consists of Cyalume a Phenyl Oxalate Ester plus a fluorescent dye molecule When the H2O2 in Solution 2 is poured into Solution 1 a |
|
1 Cyalume Experiment
Chemiluminescence: Synthesis of Cyalume and Making it Glow Intro Chemiluminescence is the Bis(2,4,6-trichlorophenyl)oxalate (A cyalume) mw = 449 |
|
Chemiluminescence
One of the most sensitive detection methods available is chemiluminescence Diphenyl oxalate (Cyalume®) is not by itself chemiluminescent, but after oxida- |
|
Chemiluminescence - ScienceDirectcom
of oxalate esters (often referred to as peroxyoxalate chemiluminescence) M substituted phenyl oxalate, 0 05 M H2 02 , 6 x 10 4 M fluorescer (9,10-diphenyl- |
|
Chemiluminescence and the Synthesis of 9 - Mount Holyoke College
chemiluminescence as an analytical tool, particularly in biomedical research A common chemistry in glow sticks involves the oxidation of diphenyl oxalate into 1 |







![PDF] Detection of hydrogen peroxide with chemiluminescent micelles PDF] Detection of hydrogen peroxide with chemiluminescent micelles](https://pubs.rsc.org/image/article/2015/pp/c4pp00311j/c4pp00311j-s2_hi-res.gif)