BAXTER CONFIDENTIAL INTERNAL USE ONLY
|
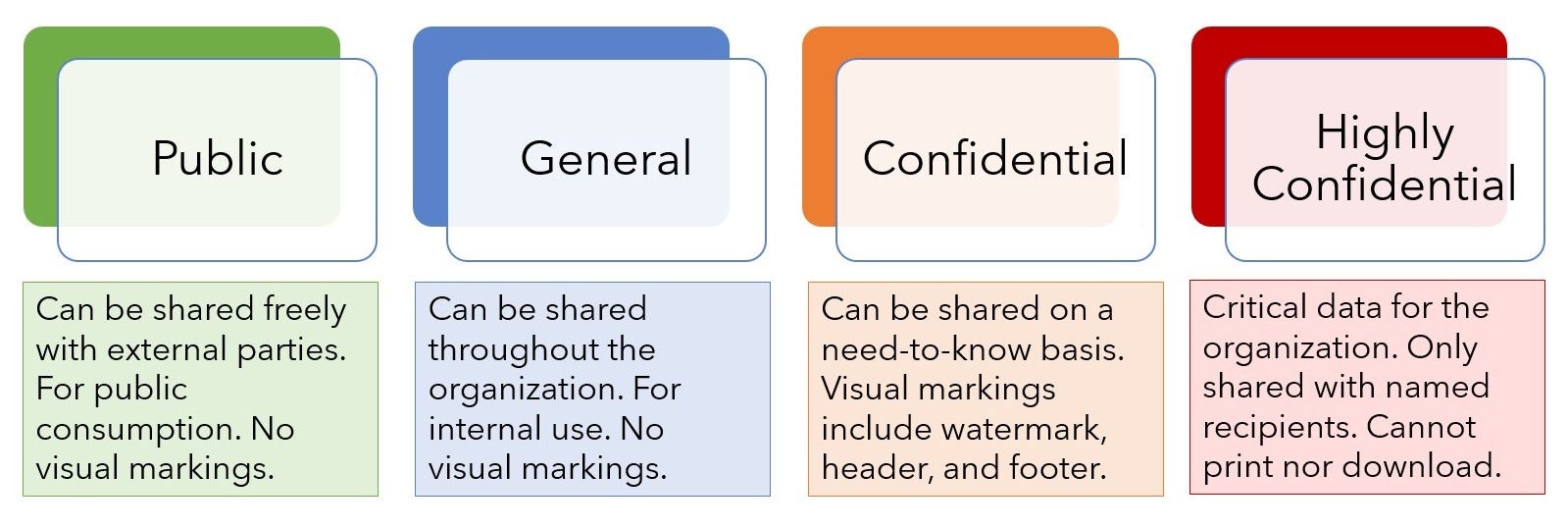
BAXTER CONFIDENTIAL
The efect of ALTAPORE SHAPE on patients with the following conditions: documented renal disease pregnancy and nursing and radiation bone therapy The efect of mixing ALTAPORE SHAPE with substances other than autologous blood or autologous bone The e ect of ALTAPORE SHAPE in pediatric patients |
|
BAXTER CONFIDENTIAL
ALTAPORE MIS System is supplied prepackaged in a sterile 7 5 ml cartridge and contains ALTAPORE microgranules sized 1–2 mm 80-85 total porosity suspended in an absorbable aqueous gel carrier ALTAPORE does not set in-situ following implantation |
|
BAXTER CONFIDENTIAL
Dosage is for SINGLE USE ONLY Do not attempt to re-sterilize or re-use Instructions for use Step 1 Open both outer (non-sterile) and inner (sterile) packaging in the sterile field Step 2 If desired ALTAPORE may be mixed with Bone Marrow Aspirate (BMA) or autologous bone Mixing equipment must be sterilized prior to use |
|
BAXTER CONFIDENTIAL
• This medicine has been prescribed for you only Do not pass it on to others It may harm them even if their signs of illness are the same as yours • If you get any side effects talk to your doctor or pharmacist or nurse This includes any possible side effects not listed in this leaflet See section 4 |
|
BAXTER CONFIDENTIAL
TRIOMEL may only be administered via a tube (catheter) into a large vein in your chest (central vein) BAXTER CONFIDENTIAL - INTERNAL USE ONLY Part Number: BE-30-03-354 Date: 10 SEP 2020 |
|
BAXTER CONFIDENTIAL
BAXTER CONFIDENTIAL - INTERNAL USE ONLY Part Number: BE-35-04-110 Date: 27OCT20 Proofread No : P04 Designer: RK Page: 1 Colour Reference:P185U For single use only Read the package leaflet before use Keep out of the sight and reach of children Do not freeze Store in the overpouch Do not infuse before |
|
WHEN IT S
Baxter employees may not possess use or unlawfully sell drugs on Baxter property or perform their work under the influence of alcohol or the unlawful use of drugs Baxter may require employees to take drug and/or alcohol tests to ensure a drug and alcohol-free workplace MINIMUM HIRING AGE Regular full-time employees must be at least 18 years old |
|
Supplier Notice of Change - Section A. Change Request
OWNER CODE: GQC. BAXTER CONFIDENTIAL - INTERNAL USE ONLY. ISSUE DATE: 31 MAR 2017. EFFECTIVE DATE: 21 AUG 2017. Baxter Tracking Number:. |
|
Section A. Change Request
Mar 5 2019 BAXTER CONFIDENTIAL - INTERNAL USE ONLY. ISSUE DATE: SEE STAMP. EFFECTIVE DATE: SEE STAMP. Suppliers must receive Baxter approval |
|
PRISMASOL and PHOXILLUM safely and effectively.
Use only with extracorporeal dialysis equipment appropriate for CRRT BAXTER CONFIDENTIAL - INTERNAL USE ONLY. Part Number: 07-25-00-0109. |
|
PRISMASOL renal replacement solution
Use only with extracorporeal dialysis equipment appropriate for CRRT BAXTER CONFIDENTIAL - INTERNAL USE ONLY. Part Number: 07-25-00-0109. |
|
Prismasol B0
substitution (or other buffer source) may increase the risk of metabolic alkalosis. • When citrate is used as an. BAXTER CONFIDENTIAL - INTERNAL USE ONLY. |
|
City of Angleton Texas Strategic Planning Session Report 2020
Sep 28 2020 CONFIDENTIAL INFORMATION – CITY OF ANGLETON. FOR INTERNAL USE ONLY. Compiled by C.J. Baxter Group |
|
Ann Cole
equates to the use of about 9 million bed days across the NHS on an annual basis3. Defining Value. 9. Baxter Confidential - Internal Use Only |
|
Package leaflet: information for the user Sodium Chloride 0.18% w/v
BAXTER CONFIDENTIAL - INTERNAL USE ONLY. Part Number: SA-30-02-285. Date: 10APR19. Designer: M.M.. Page: 1 of 8. Colour Reference: PMS 287U. Proofread No. |
|
Baxter Whistleblower Policy
[This information is only relevant for internal purposes]. Policy investigation (subject to privacy and confidentiality restrictions). |
|
1 Investigations and Audits Group Date: May 9 2019 To: All
May 9 2019 ... be privileged and confidential. It is for internal government use only and must not be ... Deerfield |
| BAXTER CONFIDENTIAL - INTERNAL USE ONLY - Part Number |
| INTERNAL USE ONLY - Part Number: 1000014990 |
| BAXTER CONFIDENTIAL - INTERNAL USE ONLY |
| TISSEEL/ARTISS Spray Set - Baxter |
| CONFIDENTIALITY UNDERTAKING - PDF4PRO |
| Baxter Whistleblower Policy |
| Title: DIRECTION INSERT CE INTERMATE SYSTEM BASIC |
| Information for the patient BREVIBLOC Premixed 10 mg/ml Solution |
| ENFit® Low Dose Tip Syringe Review |
|
M60 SET / M100 SET / M150 SET - Baxter India
使用說明 Made in France Instructions for use BAXTER CONFIDENTIAL - INTERNAL USE ONLY Part Number: 1000014921 Date: 08-JAN-2020 Proofread No |
|
112016 Made in France - Baxter India
BAXTER CONFIDENTIAL - INTERNAL USE ONLY Part Number: 1000014774 Date: 06DEC19 Proofread No : P02 Designer: R B/S C Page: 2 of 36 |
|
Package leaflet: information for the user Sodium Chloride - HPRA
shortness of breath – swelling of the ankles BAXTER CONFIDENTIAL - INTERNAL USE ONLY Part Number: TH-30-01-903 Date: 10APR19 Proofread No : 5 |
|
Section A Change Request - Baxter
5 mar 2019 · BAXTER CONFIDENTIAL - INTERNAL USE ONLY ISSUE DATE: SEE STAMP EFFECTIVE DATE: SEE STAMP Suppliers must receive Baxter |
|
AbbVie Technical Information - VIDAL
23 juil 2019 · (+Baxter EADC package No ) CB-30-02-529 using the approved 'Production Template' BAXTER CONFIDENTIAL - INTERNAL USE ONLY |
|
FLOSEAL Hemostatic Matrix, 10 mL - Baxter
Do not use FLOSEAL Matrix in patients with known allergies Rx ONLY BAXTER CONFIDENTIAL INTERNAL USE ONLY Document No : 0733787 Change |
|
DIRECTION INSERT, CE INTERMATE SYSTEM - Dufort et Lavigne
16 juil 2013 · BAXTER CONFIDENTIAL - INTERNAL USE ONLY Baxter Healthcare Corporation, Medical Products 17511 Armstrong Avenue, Irvine, |
|
PRISMASOL BO
BAXTER CONFIDENTIAL - INTERNAL USE ONLY Part Number: Use only with appropriate extra-renal replacement equipment Not for direct infusion: mix |
|
EC_Certificate_270492pdf
15 nov 2019 · Manufacturer's name: Baxter Healthcare SA Manufacturer's address: 8010 BAXTER CONFIDENTIAL - INTERNAL USE ONLY FORM NO : |