do esters react with sodium hydrogen carbonate
|
Ester Synthesis
What is the equation for the reaction between sodium bicarbonate and the residual acetic acid? Add 2 mL of saturated sodium chloride solution to the crude |
|
39 Carboxylic acids and esters
• Carboxylic acids are acidic enough to react with metals alkalis and carbonates Ethanoic acid + sodium carbonate → e Propanoic acid + lithium hydrogen |
Why is Na2CO3 used in esterification test?
Answer: Sodium carbonate solution is added to neutralise any acids and the stopper replaced. The acidic impurities and any salts formed have now been removed in the aqueous sodium carbonate solution, therefore there should be no carboxylic acid or sulfuric acid catalyst left in the ester layer.
The common name comes from the common yet essential use of the compound in baking as a leavening agent.
Typically when it reacts with an acidic material such as hydrogen phosphates, cream of tartar, juice of lemon, yoghurt, buttermilk, cocoa, and vinegar, it releases carbon dioxide.
What is the role of sodium hydrogen carbonate during esterification?
In order to isolate the ester product, the first step would be neutralizing the solution.
This can be done by adding sodium bicarbonate, which is a weak base.
Sodium carbonate reacts with the carboxylic acid as well as the acid catalyst, thus increasing the pH of the solution.
Do esters react with sodium carbonate?
CAUTION: The sodium carbonate will react vigorously with any sulfuric acid remining in the crude ester forming carbon dioxide, CO2.
Add the sodium carbonate solution dropwise to prevent the mixture from bubbling out of the test tube.
|
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters and learn sodium hydroxide (NaOH) and potassium hydroxide ... Does not cause stomach. |
|
METHYLSALICYLATE. THE ANALYTICAL SEPARATION AND
Salicylic acid or its salts and its methyl ester may be and often are |
|
Sand Binder Systems
Over-gassing leads to sodium hydrogen carbonate observed as white crystals on during the reaction between sodium silicate resin and the ester catalyst. |
|
Testsforfunctionalgroups - inorganiccompounds
Add the compound in sodium hydrogencarbonate solution slowly so that effervescence is visible clearly. These react with alcohols in the acidic medium to produce |
|
Synthesis of Esters from Acyl Chlorides and Halide Under Solid
Acyl chloride reacts with halide in the presence of sodium hydrogen carbonate and under solid-liquid phase transfer condition to afford the esters in good to. |
|
Vaccines against Drug Abuse: Morphine-Hapten Design and
Nov 14 2019 phase with glycine ethyl ester |
|
Determination of the MCPD fatty acid esters and glycidyl fatty acid
glycidyl fatty acid esters in food - Report on the collaborative trial sodium hydrogen carbonate (6.1.11) in a 50 ml volumetric flask and fill up to ... |
|
3248 Bunton and Hadwick: 665. Tracer Studies in Ester Hydrolysis
for both esterification and hydrolysis but they do not exclude the the reactions with sodium hydrogen carbonate or carbonate could be followed. |
|
24-D Interactions with Glyphosate and Sodium Bicarbonate
butoxyethylester formulation did not react with sodium bicarbonate. Nomenclature: 24-D |
|
INSTRUCTIONS - NHS and Sulfo-NHS
efficient at pH 7-8 and Sulfo-NHS-ester reactions are usually performed in using concentrated PBS or other non-amine buffer such as sodium bicarbonate. |
|
39 Carboxylic acids and esters
Carboxylic acids are acidic enough to react with metals, alkalis and carbonates Top tip: Remember the test for a carboxylic acid is fizzing with NaHCO3: The sodium carboxylate can be converted back to a carboxylic acid by the reaction |
|
Lab 12: Synthesis of an Ester - California State University, Bakersfield
In this experiment you will react a carboxylic acid and an alcohol under acidic removed during the extraction by washing with sodium bicarbonate and water |
|
7-Aldehydes, Ketones, C Acids
Carboxylic acids are polar and can form hydrogen bonds with water Aldehydes and ketones are not acidic and will not react with sodium bicarbonate |
|
CARBOXYLIC ACIDS AND THEIR DERIVATIVES 1 Carboxylic
As carboxylic acids do not give char- solubility in 5 sodium hydrogen carbonate In addition to the general reactions described, certain acids can be |
|
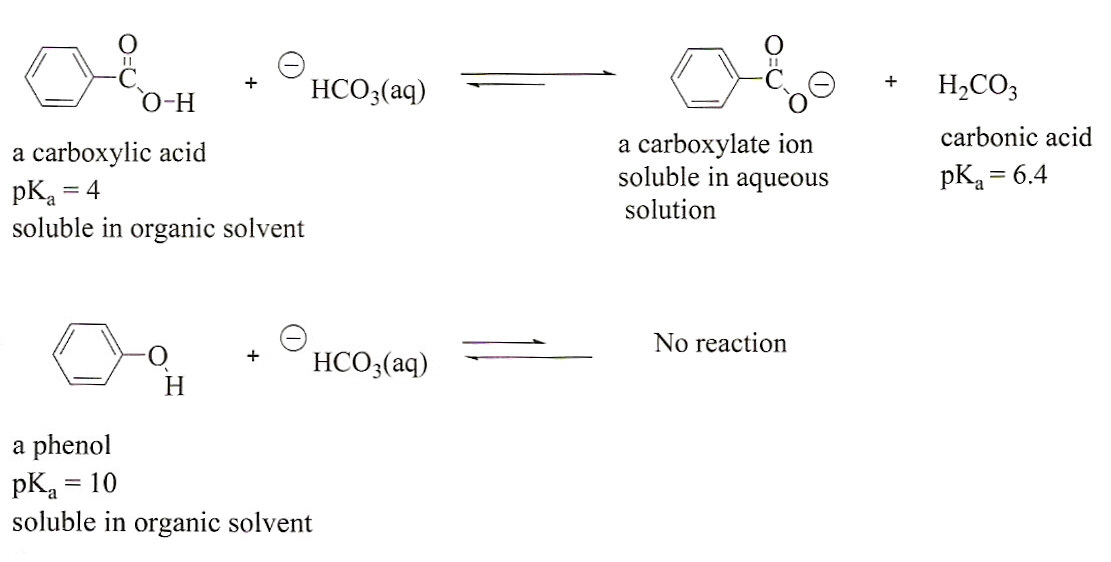
Identifying an Unknown Compound by Solubility, Functional Group
Phenols are less acidic than carboxylic acids and do not react with NaHCO3 to form water-soluble salts As a result, phenols are insoluble in 5 NaCHO3 |