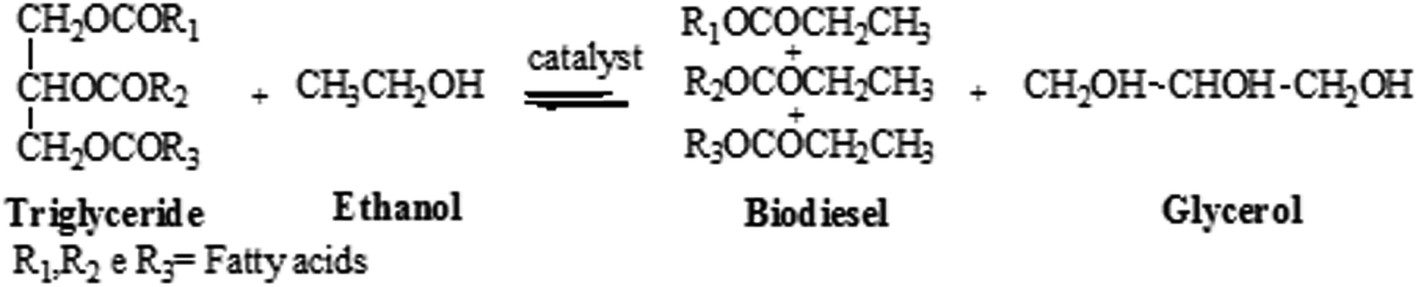do esters react with sodium hydroxide
Which of the following is formed by the reaction of an ester and NaOH?
An ester and NaOH produce an acid and an ALCOHOL in a Saponification reaction.20 mar. 2017
CAUTION: The sodium carbonate will react vigorously with any sulfuric acid remining in the crude ester forming carbon dioxide, CO2.
Add the sodium carbonate solution dropwise to prevent the mixture from bubbling out of the test tube.
What happens when sodium hydroxide reacts with ethyl ethanoate?
The reaction of ethyl ethanoate with sodium hydroxide is also called saponification reaction.
In this reaction formation of a sodium salt of the carboxylic acid takes place ( soap).
Do esters react with bases?
Esters react in the presence of an acid or a base to give back the alcohol and carboxylic acid.
|
Chapter 5 Carboxylic Acids and Esters
Esters may be broken apart under basic conditions by sodium hydroxide (lye) or potassium hydroxide to form carboxylate salts and alcohols. react with; can ... |
|
THE REACTION BETWEEN METALLIC SODIUM AND AROMATIC
ensues when aldehydes or ketones8 react with this metal. The first phase of the reaction should then consist in the addition of sodiumto the ester with the |
|
Two mild methods for synthesis of αβ–unsaturated cyanoesters
αβ–Unsaturated cyanoesters can be prepared by reaction of aldehydes and ethyl cyanoacetate catalysed by CaCl2/. Et3N or NaOH at room temperature . Keywords: α |
|
Base-catalyzed ester hydrolysis in a toluene-water system
Abstract: Hydrolysis reactions of aromatic esters in a toluene phase with sodium hydroxide in an aqueous phase have been studied in a stationary system |
|
S$D A RAPID AND SIMPLE METHOD FOR THE DETERMINATION
Sodium Hydroxide.-Aqueous solution 3.5N. Hydrochloric Acid.-One part of the Since the long-chain fatty acid esters do not with hydroxylamine in aqueous ... |
|
Optimization Study of Biodiesel Production from Refined Palm Oil
Increasing the interaction between methanol and sodium hydroxide will result in the increase in amount NaOH will react ester. Biodiesel samples were spotted ... |
|
Nomenclature of Carboxylic Acids
Esters may be broken apart under basic conditions by sodium hydroxide (lye) or potassium hydroxide to form carboxylate salts and alcohols. react with; can ... |
|
The determination of esterified fatty acids in glycerides cholesterol
values for the different esters. To accomplish this an attempt was made |
|
Testsforfunctionalgroups - inorganiccompounds
sodium hydroxide solution. Appearance of pink blue |
|
TOPIC: ORGANIC CHEMISTRY 8 – Base hydrolysis of an ester
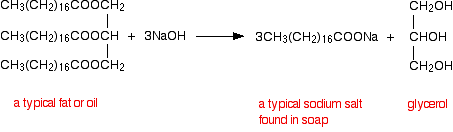
reaction can also be called SAPONIFICATION. SOAP. Soap is made when fats which are natural esters are heated with bases such as potassium hydroxide or sodium. |
|
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters and learn how to by heating lard with sodium hydroxide |
|
THE REACTION BETWEEN METALLIC SODIUM AND AROMATIC
ensues when aldehydes or ketones8 react with this metal. The first phase of the reaction should then consist in the addition of sodiumto the ester with the |
|
Transesterification of Vegetable Oils with Ethanol and
5 jui. 2009 molar ratio of 12:1 NaOH amount (1% wt/wt) |
|
N Goalby chemrevise.org 1 Carboxylic Acids
Phenols are very weakly acidic. They are weaker acids than carboxylic acids. Phenols will react with sodium metal and sodium hydroxide but are not strong. |
|
Revisiting the mechanisms of low-temperature base-catalysed ester
Subsequently this glycerolate anion can react with triglycerides and titative saponification reaction between the sodium hydroxide formed. |
|
6.1.3 revision guide carboxylic acids and esters
This reaction is reversible and does not give a good yield of the products. ii) with sodium hydroxide reagents: dilute sodium hydroxide. |
|
The determination of esterified fatty acids in glycerides cholesterol
per ester group for triglycerides cholesterol esters of long-chain fatty acids |
|
Efficient Fmoc-Protected Amino Ester Hydrolysis Using Green
27 avr. 2022 Example of a new theoretical custom amino acid that can be hydrolyzed using calcium iodide and sodium hydroxide preserving both the Fmoc and ... |
|
Preparation of Modified Acetate Fiber with Sodium Hydroxide
The hydroxyl on cellulose can react with acetic acid dehydration the ester bonds is obtained and the hydroxyl groups were closed. In order to obtain a good |
|
The Study of Chemiluminescence of Acridinium Ester in Presence of
the Chemi Luminescence (CL) reaction is acridone and that the blue light produced with out the (0.005 mol L-1 in MeOH) and 50 ?L Sodium hydroxide. |
|
39 Carboxylic acids and esters
Carboxylic acids are acidic enough to react with metals, alkalis and carbonates Usually written: RCOOH + NaOH → RCOONa + H2O Metal Carbonate + Acid The sodium carboxylate can be converted back to a carboxylic acid by the |
|
Chemical Properties and Derivatives of Glycerol - Science The
the presence of potassium or sodium hydroxide(l4), Esters can be prepared by reacting glycerine Moreover, the glycerine can be reacted with a number of |
|
Synthesis of Carboxylic Acids
Since the reaction with NaOH is always downhill, all of these reactions work R OR' O R Any “lateral” step (acid to ester or vice-versa) can be done with acid |
|
Saponification of Ethyl Acetate
Theory The rate constant k for chemical reactions is given by the Arrhenius fication of an ester by sodium hydroxide A solution containing sodium hydroxide and ethyl acetate undergoes Accordingly, a conductance bridge can be used |
|
Metal ion complex catalysis of amino acid ester hydrolysis - CORE
the reactions written for the base hydrolysis of esters is 2 hydroxide ion should be the nucleophile in the mechanisms ester by introduction of NaOH |
|
Revisiting the mechanisms of low-temperature, base-catalysed ester
Subsequently, this glycerolate anion can react with triglycerides and cause an titative saponification reaction between the sodium hydroxide formed |
|
Benzoic Acid from Ethyl Benzoate by Base Hydrolysis
esters It is a sweet smelling, colourless, liquid used in perfumery under the name Essence de Niobe; in the ethanol but the reaction does not go to completion with aqueous sodium hydroxide, the reaction going to completion, to give |