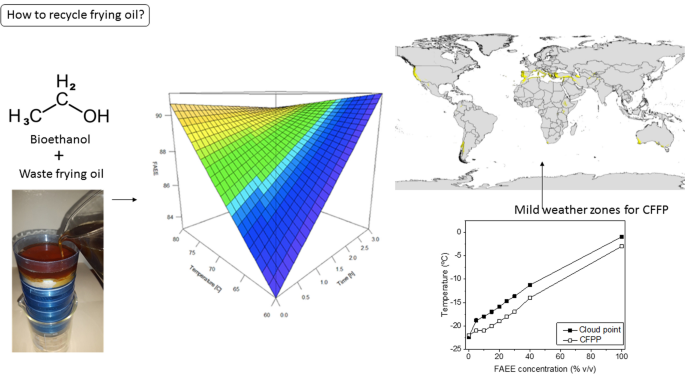
does esters react with sodium metal
Hence, the ester enolate anion resonance structure IV plays a smaller role and the enolate anion is less stabilized in the ester compared to the ketone.
Consequently, the ester is less acidic than the ketone.
Do aldehydes react with sodium metal?
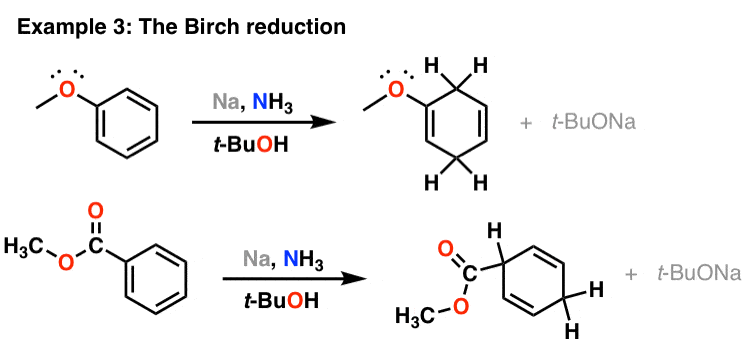
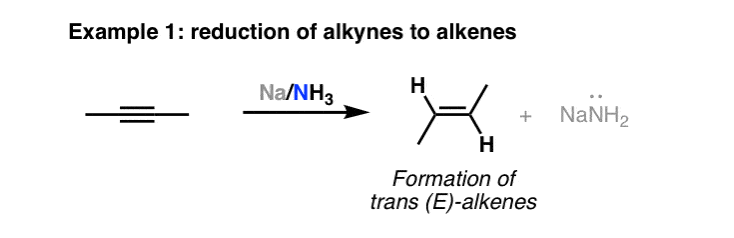
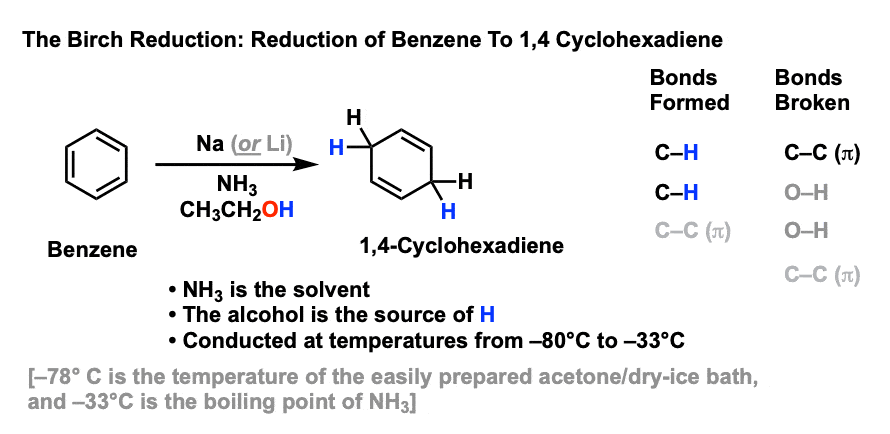
The reaction is called a Na/NH3 reduction.
The aldehyde is typically dissolved in the solvent, and a small piece of sodium metal is added to the mixture.
The sodium metal reacts with the solvent to form a complex called a sodamide, which can then reduce the aldehyde to an alcohol.
|
THE REACTION BETWEEN METALLIC SODIUM AND AROMATIC
Esters of aromatic acids react likewise |
|
The Reactions of Sodium Naphthalenide with Carbonyl Compounds
8 janv. 1972 Reaction Between Sodium and Esters in Liquid Ammonia ... When naphthalene is treated with sodium metal in solvents such as. |
|
Revisiting the mechanisms of low-temperature base-catalysed ester
The conversion of triglycerides into biodiesel is called a transesterification and the divalent metal catalysed reaction between dimethyl terephthalate (DMT) |
|
THE ACETOACETIC ESTER CONDENSATION. III. THE ROLE OF
reacts with the ester it is the metal itself which is directly responsible for fully removed reacts with sodium just as readily as does ordinary ethyl. |
|
N Goalby chemrevise.org 1 Carboxylic Acids
Phenols will react with sodium metal and sodium hydroxide but are not strong enough an acid to react carboxylic acids |
|
Chapter 5 Carboxylic Acids and Esters
Esters may be broken apart under basic conditions by sodium hydroxide (lye) or potassium hydroxide to form carboxylate salts and alcohols. • This reaction is |
|
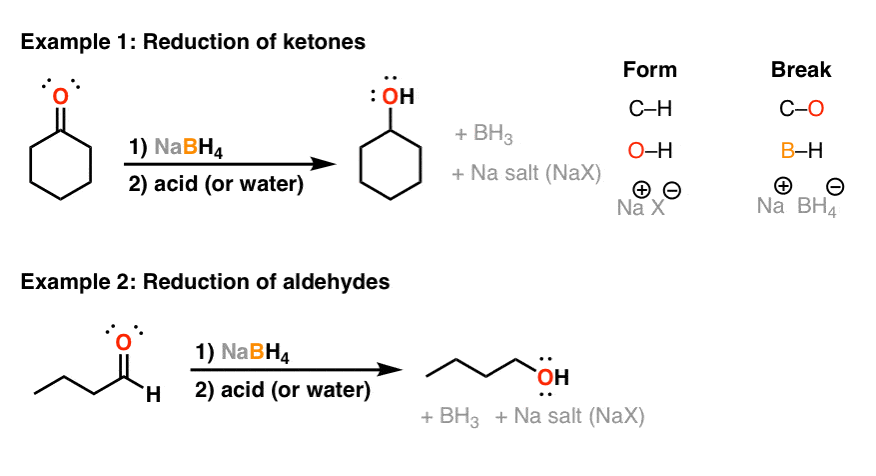
Reducing-agents.pdf
It will reduce aldehydes ketones |
|
Direct synthesis of some significant metal alkoxide bvyu emi1jg a
Reaction between sodium metal and absolute ethanol in that the fractionation of more volatile ester is much easier as there is a. |
|
SYNTHESIS METHOD FOR BORONIC ACID ESTER COMPOUND
25 Nov 2020 |
|
A new method for the reduction of esters
H. J. PRINS. For the reduction of esters only one method is available that of. BOUVEAULT and BLANC with sodium and ethyl alcohol or amyl alcohol. |
|
Reducing Agents
It can reduce acid chlorides (and some esters) quickly, but is slow to react with aldehydes; therefore LiAl(OtBu)3H provides a convenient way to synthesize |
|
Revisiting the mechanisms of low-temperature, base-catalysed ester
metal alcoholates that are active at low temperature ester It is the logical assumption therefore, that the sodium has combined with the triglyceride fat in some form, and Subsequently, this glycerolate anion can react with triglycerides and |
|
Metal ion complex catalysis of amino acid ester hydrolysis - CORE
Metal ion catalysis of the hydrolysis of a-amino acid esters was discovered in the reactions written for the base hydrolysis of esters is 2 one in which the was then converted to the sodium salt by addition of NagSO^ and precipitation of the |
|
Process of transformation of radioactive waste of metal sodium into
Qnce the reaction is complete, xylene is distillated and the alcoholate is in solution in an 1 1 - Rappel de quelques caractéristiques physiques du sodium métal minés ainsi que les esters en raison de leur facilité d'hydrolyse en milieu |
|
Chemical Properties and Derivatives of Glycerol - Science The
goods and food fields, the esters of glycerine, particu- larly the partial hydroxyl hydrogens are replaceable by metals to form reactive than another, there is generally some reaction or sodium dichromate and chromic acid oxidize glycerol |

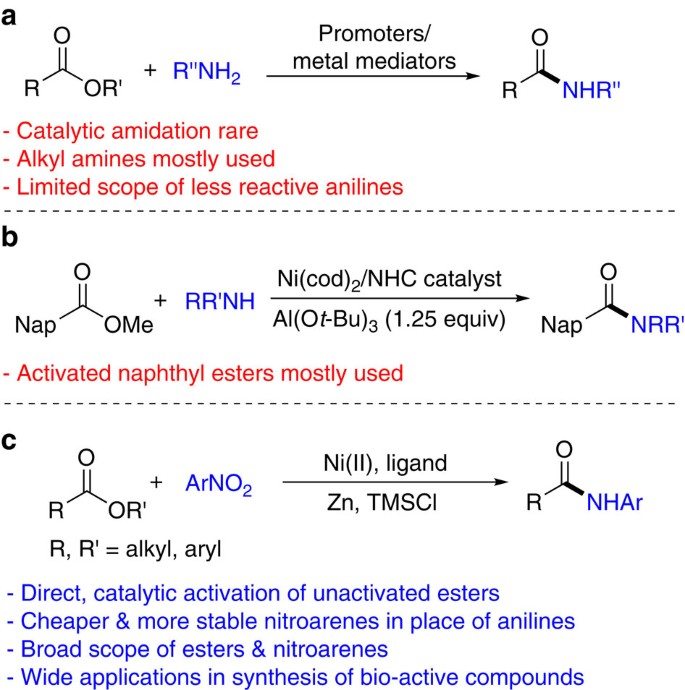














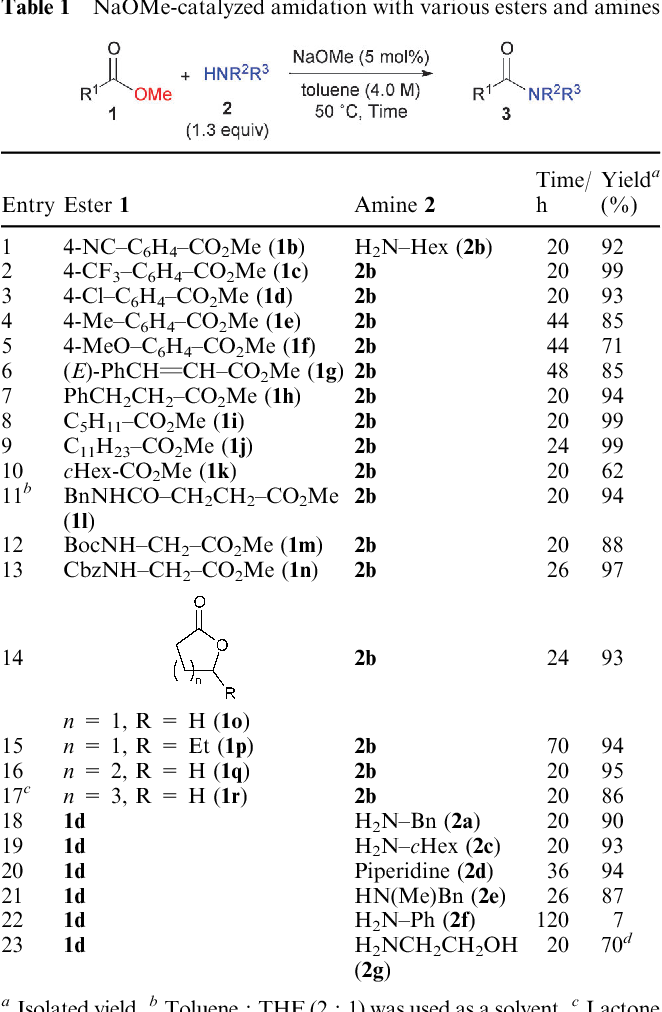
![PDF] Sodium methoxide: a simple but highly efficient catalyst for PDF] Sodium methoxide: a simple but highly efficient catalyst for](https://i1.rgstatic.net/publication/263338999_Sodium_dodecyl_sulphate_A_very_useful_surfactant_for_Scientific_Invetigations/links/0c96053bc20659e5f9000000/largepreview.png)