draw the mechanism of formation of isopentyl acetate from acetic acid and isopentyl alcohol.
|
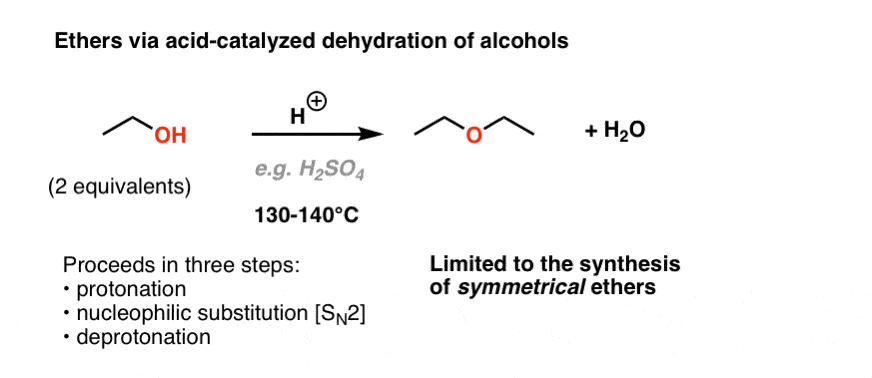
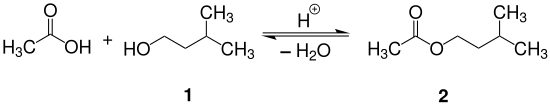
Synthesis of Isopentyl Acetate
The pheromone causes other bees to become aggressive and attack the intruder Esterification generally refers to the formation of esters from alcohol and |
What is the mechanism for isoamyl acetate?
In reaction with isoamyl alcohol, one of the acyl from acetic anhydride will bind with isoamyl alcohol and discharge one H+ to form isoamyl acetate and acetic acid.
Then, excess acyl (from acetic acid) will react with excess alcohol to form another isoamyl acetate and water.What is the mechanism of formation of isopentyl acetate?
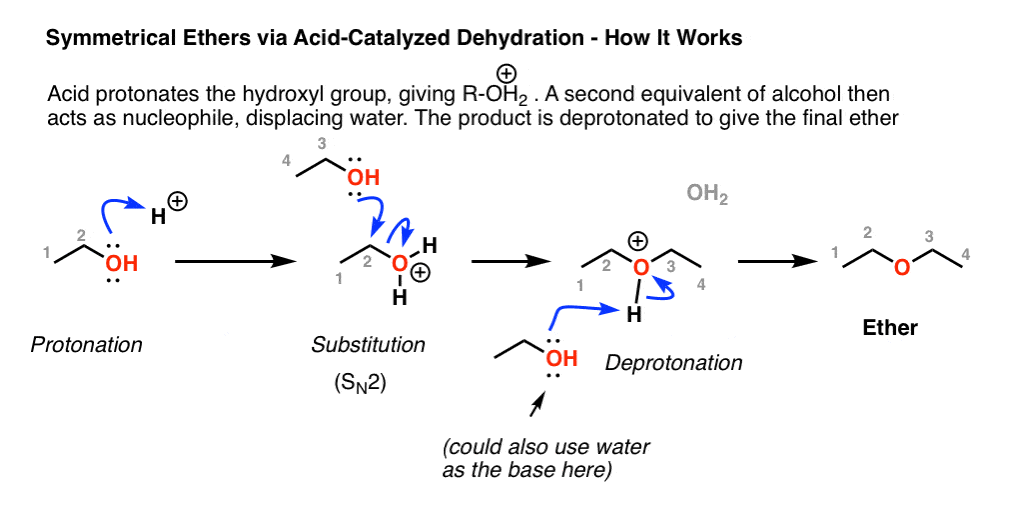
The reaction mechanism involves initial protonation of the carboxyl group, nucleophilic attack by the hydroxyl, proton transfer, and loss of water followed by loss of the catalyzing acid to produce the ester.
The process is thermodynamically controlled yielding the most stable ester product.How do you make isopentyl acetate?
.
1) Mix isopentyl alcohol (5.4 mL, via burette) and glacial acetic acid (8.5 mL, via graduated cylinder) in a round-bottom flask.
Carefully add 20 drops of conc.
H2SO4 to the mixture. .
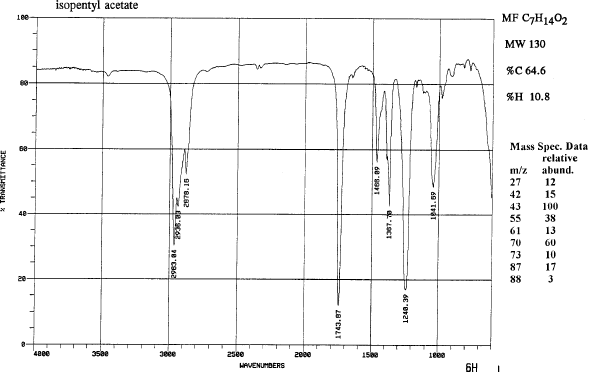
2) Put in a few boiling chips and assemble the reflux apparatus (Figure 1).Acetic acid (CH3CO2H) reacts with isopentyl alcohol (C5H12O) to yield isopentyl acetate (C7H14O2), a fragrant substance with the odor of bananas.
|
Lesson Plan: Synthesis of Isopentyl Acetate (Banana Oil)
isopentyl acetate from isopentyl alcohol and acetic acid. 2. Purpose. The purpose of this experiment is to synthesize isopentyl acetate (3-methylbutyl acetate). |
|
Synthesis of Isopentyl Acetate
Synthesis of Isopentyl Acetate. 8. Objectives. To prepare isopentyl acetate from isopentyl alcohol and acetic acid by the Fischer esterification reaction. |
|
Lesson Plan: Synthesis of Isopentyl Acetate (Banana Oil)
In this lab you will perform a Fisher Esterification to synthesize isopentyl acetate from isopentyl alcohol and acetic acid. 2. Purpose. The purpose of this |
|
Chapter 5 Carboxylic Acids and Esters
Amyl acetate. Also known as banana oil and pear oil; the commercially available Reaction of a carboxylic acid with an alcohol to produce an ester and water. |
|
Organic Chemistry with a Biological Emphasis Volume II
Introduction: Why aren't identical twins identical? Just ask SAM. Section 1: Two mechanistic models for nucleophilic substitution. A: The SN2 mechanism. B: The |
|
Experiment 14A: Isopentyl Acetate
Oct 14 2020 In this experiment |
|
Bellevue College
In this experiment isopentyl acetate will be prepared from the reaction of acetic acid and isopentyl alcohol. Draw a structure for the ester that would be ... |
|
Synthesis of paracetamol by acetylation
Acetic acid. H+. H. + H+. +OH. O. O. R-OH. Scheme SM 3.1.5.1 – General acetylation reaction mechanism of an alcohol with acetic anhydride in acidic media. The |
|
Chapter 3 Alcohols Phenols
https://www.angelo.edu/faculty/kboudrea/index_2353/Chapter_03_2SPP.pdf |
|
EXPERIMENT 5 SYNTHESIS OF ESTERS USING ACETIC
(isoamyl alcohol isopentyl alcohol) |
|
Lesson Plan: Synthesis of Isopentyl Acetate (Banana Oil)
the reactants to the reaction mixture. The reaction mechanism involves initial ... isopentyl acetate from isopentyl alcohol and acetic acid. 2. Purpose. |
|
Synthesis of Isopentyl Acetate.pdf
To prepare isopentyl acetate from isopentyl alcohol and acetic acid by the Fischer esterification reaction. Introduction. Esters are derivatives of carboxylic |
|
A SN1 Reaction: Synthesis of tert-Butyl Chloride
Scheme SM 2.1.1.1 – Mechanism for the formation of tert-Butyl Chloride flavorings namely isoamyl acetate (bananas) |
|
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters and learn how to predict the products of ester synthesis and hydrolysis reactions. |
|
ISOAMYL ACETATE SYNTHESIS
This ester is formed from acetic acid and alcohol. The mechanism by which yeasts form esters is believed to be a reaction catalysed by an enzyme called alcohol |
|
Laboratory Manual
Experiment 3: Synthesis of a flavoring principle: Isopentyl Acetate . An acid-base extraction in which an organic acid or base is extracted from an ... |
|
12AL Experiment 8 (3 days): Synthesis of Isopentyl Acetate (aka
Today's synthesis a Fischer Esterification |
|
Ester Synthesis Lab (Student Handout)
reacting ethanol with acetic acid to give ethyl acetate is shown below. alcohol and carboxylic acid you will use in the synthesis reaction. |
|
Synthesis of paracetamol by acetylation
Acetic acid. H+. H. + H+. +OH. O. O. R-OH. Scheme SM 3.1.5.1 – General acetylation reaction mechanism of an alcohol with acetic anhydride in acidic media. |
|
Name Test 2 (4/10) CHEM 0203 (Organic) Spring 2019 1. (6 pts. ea
4. (14 pts.) Draw a mechanism for the acid catalyzed esterification of acetic acid and isoamyl alcohol to form isoamyl acetate. Remember to include electron |
|
Synthesis of Isopentyl Acetate (Banana Oil) - Thermo Fisher Scientific
The purpose of this experiment is to synthesize isopentyl acetate (3-methylbutyl acetate) via an esterification reaction between acetic acid and isopentyl alcohol |
|
Synthesis of Isopentyl Acetate - Diman Regional
Objectives To prepare isopentyl acetate from isopentyl alcohol and acetic acid by the Fischer The reaction mechanism is shown in Equations 2-6 Equation 2 |
|
Synthesis of Isopentyl Acetate - Ventura College Organic Chemistry
12AL Experiment 8 (3 days): Synthesis of Isopentyl Acetate (aka: Banana Oil) Wear gloves – concentrated acetic acid and sulfuric acid can cause burns – their Today's synthesis, a Fischer Esterification, reacts an alcohol with a carboxylic acid to important that you learn how to draw the mechanism for ANY Fischer |
|
10 Fisher Esterification: Preparation of Banana Oil - Web Pages
The overall mechanism for a general acid and alcohol is depicted in Figure 2 In your reaction, the synthesis of banana oil – isopentyl acetate, you will be using isopentyl alcohol (3-methyl-1-butanol) and acetic acid (ethanoic acid, see Figure |
|
Lab 1: Synthesis
Mechanism (In class) Esters are produced by the reaction between alcohols and carboxylic acids reacting ethanol with acetic acid to give ethyl acetate is shown below alcohol and carboxylic acid you will use in the synthesis reaction |
|
Synthesis of isopentyl acetate lab report conclusion - Squarespace
Synthesis of isopentyl acetate lab report conclusion and attribution Reaction to carboxylic acid and alcohol produces ester and water Draw the lower water layer Questions The specific weight of glacial acetic acid is 1 05 g/ml one of the reaction substances added to the reaction mixture 1 The reaction mechanism |
|
EXPERIMENT 5 SYNTHESIS OF ESTERS USING ACETIC
(isoamyl alcohol, isopentyl alcohol), 1-octanol (n-octyl alcohol), phenylmethanol ( benzyl alcohol) • 2-3 mL (2) acetic anhydride alcohol an acetate ester acetic acid side product HO C O CH3 overwhelm your olfactory sensing mechanism |
|
Lab 12: Synthesis of an Ester - California State University, Bakersfield
Run and interpret the 1H NMR of a reaction product Alcohol Carboxylic Acid Ester Scent Isopentyl Alcohol Acetic Acid Isopentyl Acetate Banana Ethanol |
|
Preparation of Banana Oil - Bellevue College
In this experiment isopentyl acetate will be prepared from the reaction of acetic acid and isopentyl alcohol The reaction does not yield significant amounts of the |
|
The Organic Chemistry II Laboratory Course Chem 302, Spring 2008
isopentyl acetate or isoamyl acetate, by heating isopentyl alcohol (3-methyl-1- butanol) with acetic and sulfuric acid: Because several of the components are |


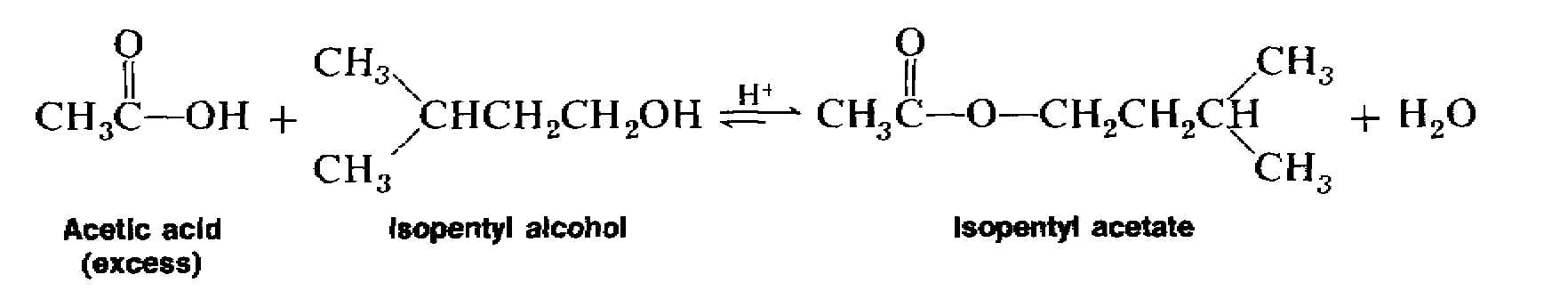







![Solved] A: Write the overall balanced equation and mechanism for Solved] A: Write the overall balanced equation and mechanism for](https://i.ytimg.com/vi/aKDPN8tTyOM/hqdefault.jpg)







![Solved] A: Write the overall balanced equation and mechanism for Solved] A: Write the overall balanced equation and mechanism for](https://i1.rgstatic.net/publication/225437592_Lipase-catalyzed_synthesis_of_isoamyl_butyrate_Optimization_by_response_surface_methodology/links/0912f508de91e81ffc000000/largepreview.png)