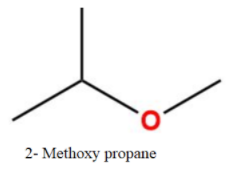
c4h10o isomers
How many isomers are possible for C4H10?
What are the seven isomers of C4H10O? C4H10O => CnH (2n+1)O With the above-mentioned general formula two types of organic compounds are possible. One is alcohol and other is ether. Alcohols: 1) CH3—CH2—CH2—CH2—OH Butanol / Butyl alcohol 2) CH3—CH2—CH (OH)--CH3 Butan-2-ol 3) CH3—CH (CH3)—CH2—OH 2-methylpropanol
How much isomers does C5H10 have?
There are ten structural isomers possible for molecular formula C5H10,Since degree of unsaturation is 2,it indicates that whether it is an alkene or cycloalkane.The following structural isomers exists:
Is C4H10O ionic or covalent?
Coming to our question which bond is stronger – covalent or ionic? 15th February 2021 In Uncategorised. Answer = C4H10 ( BUTANE ) is Polar What is polar and non-polar? Ionic and covalent bonds are the two extremes of bonding. Chem., 1997, 36 (14), pp 3022–3030) So while nothing is completely covalent or ionic, BeF2 is extremely toward ionic.

Constitutional isomers of C4H10O Alcohol & Ether

Lecture Problem: Draw all the constitutional isomers of C4H10O

How to Draw Structural Isomers containing Oxygen (C4H10O)
|
¨Ubung 1 Es gibt 4 isomere Butanole mit der Summenformel
Es gibt 4 isomere Butanole mit der Summenformel C4H10O. Die 1H- und 13C-Spektren dieser Verbindungen sind abgebildet. Welche Spek-. |
|
1. (15 points) Three C4H10O isomers are shown below along with
(15 points) Three C4H10O isomers are shown below along with their boiling points. (CH3)3C–OH. CH3–CH2–CH2–CH2–OH. CH3–CH2–CH2–O–CH3 tert-butyl alcohol. |
|
Homework 3 1. The following proton spectra were obtained for
of C4H10O. (Hint: There are just 7 structural isomers possible for this molecular formula. - the (CH3)2CHOCH3 isomer's NMR is NOT shown.). |
|
Grundkurs Alkohole 1975/V 3. a) Butanol-(1) bzw. Butanol-(2
2.1 Zeichnen Sie die Strukturformel des in Frage kommenden isomeren Alkohols Vom Butanol (Summenformel: C4H10O) sind fünf Isomere bekannt. Sie werden. |
|
Structural Isomers – Just how many structures can you make from a
structural or constitutional isomers for our saturated formula of C5H12. Problem 3 – Draw all of the possible isomers of C4H10O. |
|
Untitled
10.06.2016 Compounds A B |
|
Q. No. 2 Total number of isomeric alcohols with formula C4H10O
Total number of isomeric alcohols with formula C4H10O. Option 1. 2. Option 2. 1. Option 3. 3. Option 4. 4. Correct Answer. 4. Explanation. Q. No. 3. |
|
2.10 Alcohols.rtf
There are four structurally isomeric alcohols of molecular formula C4H10O. Graphical formulae of these isomers labelled A |
|
Lesson Plan: Regioisomers of Butanol pS45
Ethyl methyl ether is a constitutional isomer while the propanol isomers are positional isomers. Let us now consider the isomers of C4H10O. There are eight |
|
Lesson Plan: Regioisomers of Butanol pS80
Ethyl methyl ether is a constitutional isomer while the propanol isomers are positional isomers. Let us now consider the isomers of C4H10O. There are eight |
|
1 (15 points) Three C4H10O isomers are shown below along with
(15 points) Three C4H10O isomers are shown below along with their boiling points (CH3)3C–OH CH3–CH2–CH2–CH2–OH CH3–CH2–CH2–O–CH3 tert-butyl alcohol |
|
STEPS FOR DRAWING ISOMERS
The following five steps will help you to draw the isomers once you are given a molecular formula I am using the formula C6H14 as an example 1 Draw the main |
|
Electron collisions with isomers of C4H8 and C4H10
The C4H8 isomers present a double bond between two carbons and the C4H10 isomers present only single bonds between the carbon atoms The structures of skew-1- |
|
Geometric Isomerismpdf
The individual isomers are known as Metamers Page 15 Methyl propyl ether and diethyl ether both have the molecular formula C4H10O The number |
|
3/08 1 Unit 4 Chemistry of Carbon Organic Chemistry studies the
Some isomers have the same molecular formula but different functional group C4H8O2 Carboxylic acid Ester C4H6 Alkene Diene C4H10O Alcohol |
|
Isomerism Questions - No Brain Too Small
An alcohol A with the molecular formula C4H10O can exist as enantiomers (optical isomers) (a) (i) State the structural requirement for a molecule to be |
|
AP® CHEMISTRY 2010 SCORING GUIDELINES (Form B)
The compound butane C4H10 occurs in two isomeric forms n-butane and (a) Draw the structural formula of each of the isomers (include all atoms) |
|
2-Butanol C4H10O - PubChem
2-Butanol C4H10O CID 6568 - structure chemical names physical and chemical (R)-isomer; 2-butanol lithium salt (S)-isomer; sec-butyl alcohol |
|
Chapter 24 Organic Chemistry
Which one of these hydrocarbons does not have isomers? A C7H16 B C6H14 C C5H10 D C4H8 E C3H8 7 How many structural isomers are there of C4H10? |
How many isomers are there in C4H10O?
There are seven isomers in C4H10O. Out of these seven isomers, four are alcohol and three are ether.What are the 7 isomers of C4H10O?
C4H10O
Butanols. n-Butanol. sec-Butanol. tert-Butanol. Isobutanol.Ethoxyethane (Diethyl ether)1-Methoxypropane (methyl propyl ether)2-Methoxypropane (isopropyl methyl ether)What are the 4 alcohol isomers of C4H10O?
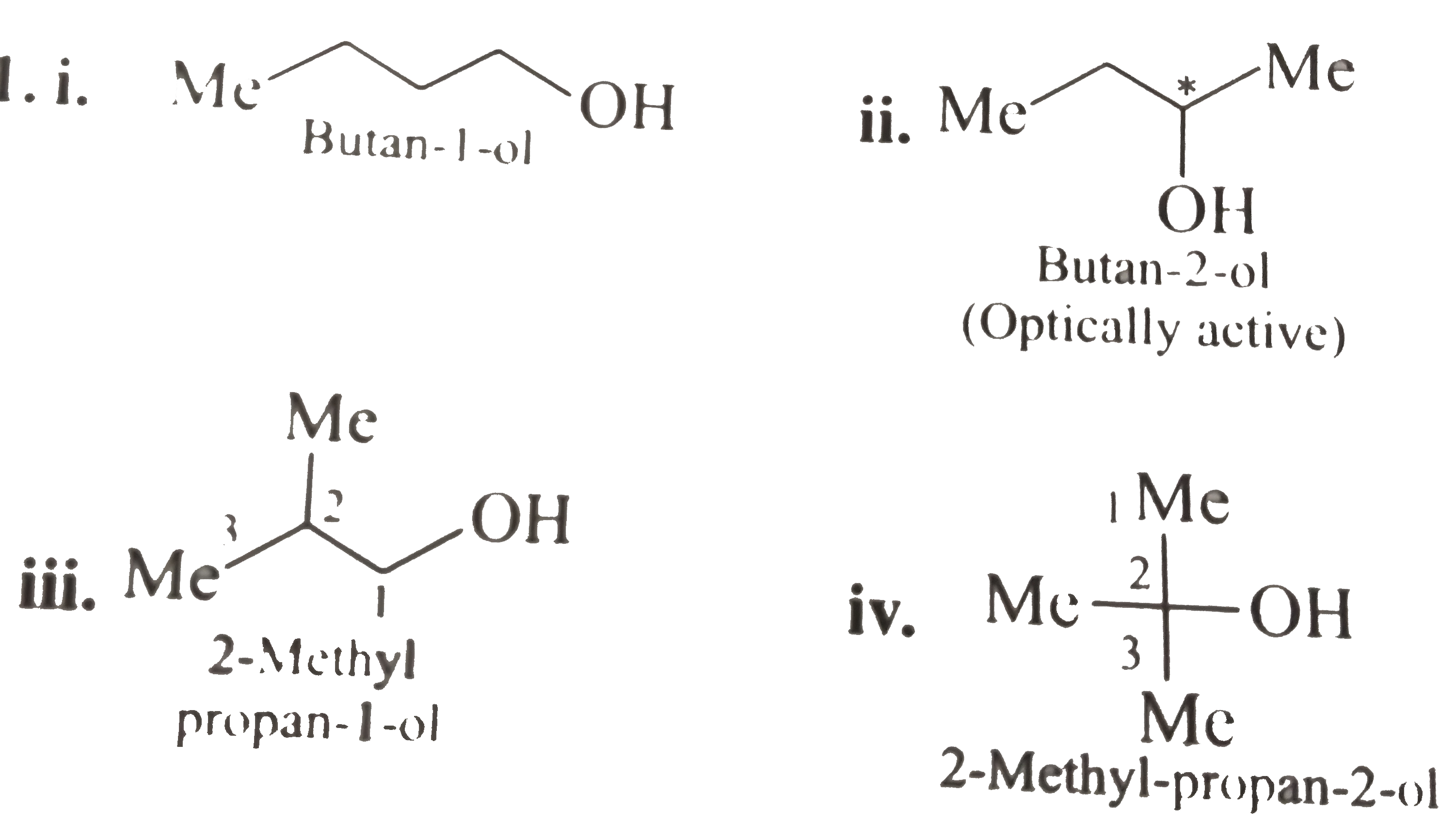
The four isomers of alcohol C4H10O are butan-1-ol, butan-2-ol, 2-methylpropan-1-ol and 2-methylpropan-2-ol. Isomers are compounds that contain the same number of atoms but have a different spatial arrangement.- There are three possible ethers.
| Functional Group Isomers CH OH Isomers that contain different |
| Chemistry 130 Experiment 7: Isomerism |
| What Are the Structural Isomers of C4H10O? - Referencecom |
| C4h10o ether isomers |
| Structural isomers of c4h10o |
| Searches related to c4h10o isomers filetype:pdf |
What are the names of the seven isomers of C4H10O?
- Butanols. n-Butanol. sec-Butanol. tert-Butanol.
. Isobutanol. - Ethoxyethane (Diethyl ether)
- 1-Methoxypropane (methyl propyl ether)
- 2-Methoxypropane (isopropyl methyl ether)
How many isomers are possible for C4H10?
- What are the seven isomers of C4H10O? C4H10O => CnH (2n+1)O With the above-mentioned general formula two types of organic compounds are possible.
. One is alcohol and other is ether.
. Alcohols: 1) CH3—CH2—CH2—CH2—OH Butanol / Butyl alcohol 2) CH3—CH2—CH (OH)--CH3 Butan-2-ol 3) CH3—CH (CH3)—CH2—OH 2-methylpropanol
How much isomers does C5H10 have?
- There are ten structural isomers possible for molecular formula C5H10,Since degree of unsaturation is 2,it indicates that whether it is an alkene or cycloalkane
.The following structural isomers exists:
Is C4H10O ionic or covalent?
- Coming to our question which bond is stronger – covalent or ionic? 15th February 2021 In Uncategorised.
. Answer = C4H10 ( BUTANE ) is Polar What is polar and non-polar? Ionic and covalent bonds are the two extremes of bonding.
. Chem., 1997, 36 (14), pp 3022–3030) So while nothing is completely covalent or ionic, BeF2 is extremely toward ionic.
|
Three C4H10O isomers are shown below, along with their boiling
(b) Which has the weakest? (c) Explain briefly why n-butyl alcohol has a much higher bp than methyl propyl ether (d) Explain briefly the difference between the first |
|
Q No 2 Total number of isomeric alcohols with formula C4H10O
Total number of isomeric alcohols with formula C4H10O Option 1 2 Option 2 1 Option 3 3 Option 4 4 Correct Answer 4 Explanation Q No 3 Which of the |
|
NMR Key 3
of C4H10O (Hint: There are just 7 structural isomers possible for this molecular formula - the (CH3)2CHOCH3 isomer's NMR is NOT shown ) We have not yet |
|
Formules semi-développées des alcools de formule brute C4H10O
Exercice : Identification d'isomères (22 points) 1) (4 pts) Formules semi- développées des alcools de formule brute C4H10O : CH2 CH2 CH2 OH CH3 |
|
Lesson Plan: Regioisomers of Butanol, pS45 - Thermo Fisher
An easy example to consider is isomers of C3H8O This molecular formula has three possible stable isomers, two alcohols and an ether Placing an alcohol group on the terminal, or 1, position of propane (C3H8) yields the primary alcohol 1-propanol (propan-1-ol) |
|
Structural Isomers – Just how many structures can you make from a
As before, we will begin with a five carbon straight chain isomer There are 12 additional Problem 3 – Draw all of the possible isomers of C4H10O Hint: There |
|
ISOMERS 2019:1 - No Brain Too Small
(b) Draw four structural (constitutional) isomers of C4H10O that are alcohols (ii ) Explain why compound A exists as geometric (cis-trans) isomers while |
|
MOSTLY ALCOHOLS 2019:1 (also included in Collated Isomers
(iii) One of the other alcohol isomers of C4H10O can also be oxidised to form a carboxylic acid • Identify this isomer by name or structural formula • Explain your |
|
Oct 2017 - Chemistry
21 oct 2017 · 21 There are four isomeric alcohols with the molecular formula C4H10O (a) Two The other two isomers are alcohol X and alcohol Y (i) Draw |
|
210 Alcoholsrtf - Physics & Maths Tutor
There are four structurally isomeric alcohols of molecular formula C4H10O Graphical formulae of these isomers, labelled A, B, C and D, are shown below H H H |





![615 isomerism - chemrevise - [PDF Document] 615 isomerism - chemrevise - [PDF Document]](https://demo.vdocuments.mx/img/378x509/reader019/reader/2020031415/5857bcac1a28ab6e328cc48f/r-1.jpg)