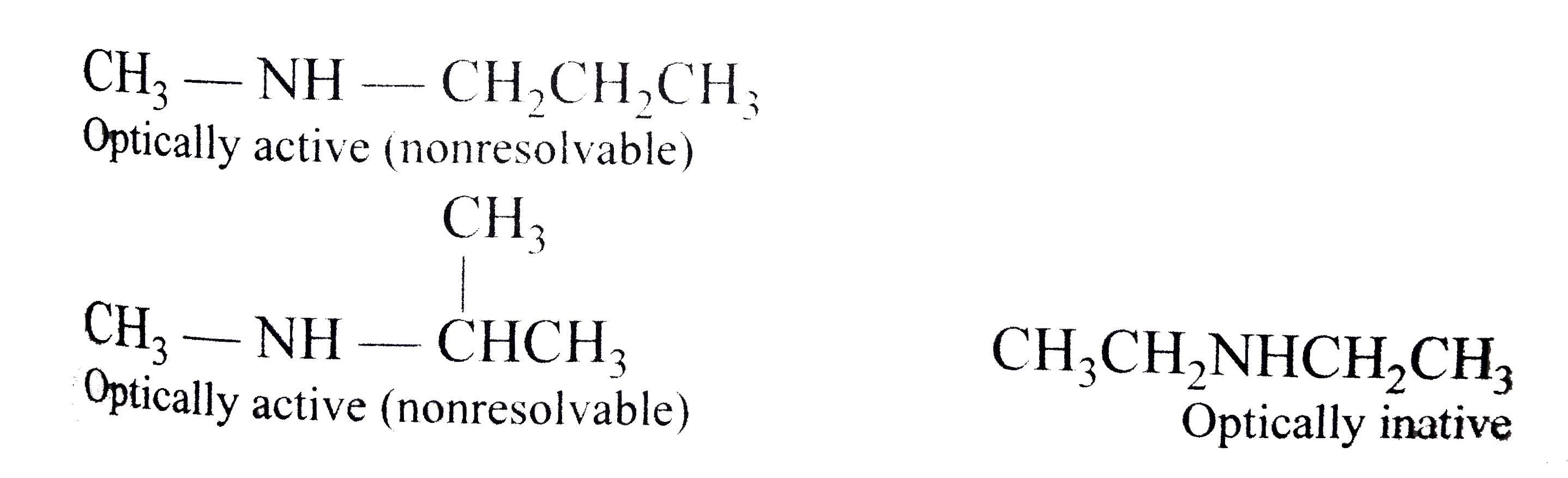c4h11n isomers
How many constitutional isomers are there with the molecular formula c4h11n?
There are eight constitutional isomers with the molecular formula C4H11N. Name and draw structural formulas for each. Classify each amine as primary, secondary, or tertiary. This problem has been solved! You'll get a detailed solution from a subject matter expert that helps you learn core concepts.
Why is C4H10 a branched molecule?
These isomers arise because of the possibility of branching in carbon chains. For example, there are two isomers of butane, C4H10 C 4 H 10. In one of them, the carbon atoms lie in a "straight chain" whereas in the other the chain is branched. Be careful not to draw "false" isomers which are just twisted versions of the original molecule.
What are the different types of optical isomers?
Note that we have to look beyond the first atom attached to the central carbon atom. The four circles indicate the four unique groups attached to the central carbon atom, which is chiral. Another type of optical isomer are diastereomers, which are non-mirror image optical isomers.
What are stereoisomers that are not geometric isomers?
Stereoisomers that are not geometric isomers are known as optical isomers. Optical isomers differ in the placement of substituted groups around one or more atoms of the molecule. They were given their name because of their interactions with plane-polarized light. Optical isomers are labeled enantiomers or diastereomers.
|
Tableau ”Amines” Cf page 89
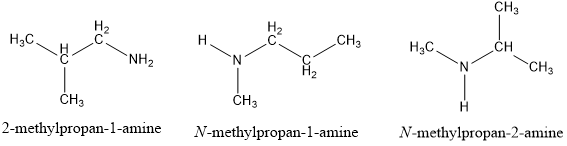
NN-dimethylmethanamine C4H11N butan-1-amine C4H11N butan-2-amine C4H11N 2-methylpropan-1-amine C4H11N 2-methylpropan-2-amine C4H11N |
|
Structural Isomers – Just how many structures can you make from a
Problem 4 – Draw all of the possible isomers of C4H11N Hint: There should be eight If you feel ambitious try and draw all of the possible isomers of |
|
Q1How many isomers have the molecular formula C5H12? A 2 B 3
Q3 How many secondary amines have the molecular formula C4H11N? A 2 B 3 C 4 |
|
AMINES
Molecular formula C3H9N represent three functional isomers Primary C4H11N CH3 – CH2 – CH2 – CH2 – NH2 [ Butan - 1- amine ( n butylamine)] |
|
Chapter 5: Stereoisomerism- three-dimensional arrangement
C4H11N 5 2 Introduction to Stereoisomerism Enantiomers: non-superimposable mirror image isomers Enantiomers are related to each other much like a right |
|
Stereochemistry three-dimensional arrangement of atoms (groups
Enantiomers: non-superimposable mirror image isomers Enantiomers are related to each other C4H11N different carbon skeleton different functional group |
|
Class XII Chapter 13 – Amines Chemistry Question 131
(i) Write structures of different isomeric amines corresponding to the molecular formula C4H11N (ii) Write IUPAC names of all the isomers (iii) What type of |
|
Aminespdf - cloudfrontnet
Primary amines secondary amines and tertiary amines are functional isomers Q 6 Write IUPAC names of all the isomers of C4H11N NCERT Chemistry (Vol 2) |
|
CHAPTER 21 ORGANIC CHEMISTRY
Isomers are distinctly different substances Resonance is the use of more than one Lewis structure to describe the bonding in a single compound |
|
Question#46700 – Chemistry - Assignment Expert
How many structural isomer of amine are possible with molecular formula C4H11N Answer: There are six isomers which have molecular formula C4H11N |
|
Sans titre
11 results · PDF Télécharger c4h11n isomers Gratuit PDF tert-Butylamine C4H11N CID 6385 - structure chemical names physical and chemical properties |
|
Butylamine C4H11N - PubChem
It is one of the four isomeric amines of butane It is known to have the fishy ammonia-like odor common to amines DrugBank |
|
Amines - NCERT
9 2 (i) Write structures of different isomeric amines corresponding to the molecular formula C4H11N (ii) Write IUPAC names of all the isomers |
|
Isomerism Unacademy
C4H11N ? No of 1° 2° and 3° amine Sol (1°amine) (2° amine) C—C—C—NH—C (3° amine) (10) C2FClBrI (No of structural isomerism) |
|
Each of the parts (a) to (e) below concerns a different pair of isomers
(c) Branched-chain structural isomers are possible for alkanes which have more than three How many secondary amines have the molecular formula C4H11N? |
|
Electron collisions with isomers of C4H8 and C4H10
The C4H8 isomers present a double bond between two carbons and the C4H10 isomers present only single bonds between the carbon atoms The structures of skew-1- |
|
C4H11N isomers - Molport
Molecule Directory By Molecular Formula / C4H11N Browse commercially available molecule directory by molecule formula Molecular formula compounds: |
|
Chapter 1 introduction to organic chemistry
The number of isomers you should be able to find is given in parentheses a) C3H9N (4) b)C4H11N(7) c) C3H5F(7;there are more) d) |
| Butane Isomers Structural Isomers - University of Wisconsin |
| Heptane All Isomers - Texas Commission on Environmental Quality |
| LEWIS FORMULAS STRUCTURAL ISOMERISM AND RESONANCE STRUCTURES |
| CRESOL (all isomers) and PHENOL 2546 - Centers for Disease |
| Decane All Isomers |
| Searches related to c4h11n isomers filetype:pdf |
|
PHYSIQUE-CHIMIE TS Chapitre 4 Tableau ”Amines” Cf page 89
N,N-dimethylmethanamine C4H11N butan-1-amine C4H11N butan-2-amine C4H11N 2-methylpropan-1-amine C4H11N 2-methylpropan-2-amine C4H11N |
|
Corrigé du Devoir Surveillé N°4 de Physique – Chimie - Eklablog
l'atome de carbone C est : 4 - l'atome d'oxygène O est : 2 - l'atome d'azote N est : 3 2) Les isomères de formule brute C4H11N sont : CH3 CH2 CH2 CH2 |
|
Three C4H10O isomers are shown below, along with their boiling
(b) Which has the weakest? (c) Explain briefly why n-butyl alcohol has a much higher bp than methyl propyl ether (d) Explain briefly the difference between the first |
|
Structural Isomers – Just how many structures can you make from a
As before, we will begin with a five carbon straight chain isomer There are 12 additional Problem 4 – Draw all of the possible isomers of C4H11N Hint: There |
|
Amines
(i) Write the structures of different isomeric amines corresponding to the molecular formula, C4H11N (ii) Write IUPAC names of all the isomers (iii) What type of |
|
Chemistry of Amines
compounds shown in the top row of the following diagram are all C4H11N isomers The first two are classified as 1º-amines, since only one alkyl group is bonded to the nitrogen; however, the alkyl group is primary in the first example and tertiary in the second |
|
AMINES
C4H11N CH3 – CH2 – CH2 – CH2 – NH2 [ Butan - 1- amine ( n butylamine)] 3 Position isomerism : This is due to difference in chain of amino group • C3H9N |
|
CHAPTER 21 ORGANIC CHEMISTRY
Methylcyclopropene is another cyclic isomer having the C4H8 formula For cis- trans isomerism (geometric isomerism), you must have at least two carbons with |
|
15 Isomers
molecules isomers or just different representations of the same molecule? ( Answer: These are b)C4H11N(7) c) C3H5F(7;there are more) d) C3H4Cl2(10) |
|
Relations disomérie entre les molécules - Serveur UNT-ORI
structures dans l'espace différentes (isomères de conformation cf chapitre 1, Des isomères de constitution ont même formule brute, mais des formules |