0.45 sodium chloride (1/2 normal saline)
|
045% Sodium Chloride Injection USP
It is a parenteral solution containing sodium chloride in water for injection intended for intravenous administration Each 100 mL of 0 45 Sodium Chloride |
|
045% Sodium Chloride Injection USP
3 déc 2018 · 0 45 Sodium Chloride Injection USP is a sterile nonpyrogenic hypotonic solution composed of sodium chloride (NaCl) and water The |
Tell your doctor right away if you have any serious side effects, including: swelling hands/ankles/feet, muscle cramps, unusual weakness, headache, nausea, extreme drowsiness, mental/mood changes (such as confusion), seizures.
What is d5 0.45 NaCl used for?
Sodium Chloride 0.45 % w/v and Glucose 5.0 % w/v solution is indicated for: - Treatment of dehydration or hypovolaemia in cases where supply of water, sodium chloride and carbohydrates is required due to restriction of the intake of fluids and electrolytes by normal routes.
What would happen if you injected a patient with 0.45% saline solution?
The intravenous administration of 0.45% Sodium chloride injection can cause fluid and/or solute overloading resulting in dilution of serum electrolyte concentrations, overhydration, congested states or pulmonary edema.
Is .45 half normal saline?
The 0.9% solution may simply be called "normal saline", abbreviated NS.
The 0.45% solution may be referred to as "half normal saline" (since 0.45% is half of 0.9%) and abbreviated 1/2 NS.
|
Making up intravenous fluids
0.45% Sodium Chloride 10% glucose 500ml bag. Method 1: If 0.45% Sodium Chloride with 5% dextrose is available a) Remove and discard 56 ml from a 500ml bag of |
|
5% Dextrose and 0.45% Sodium Chloride Injection USP
DESCRIPTION. 5% Dextrose and 0.45% Sodium Chloride Injection USP solution is sterile and nonpyrogenic. It is a large volume parenteral solution. |
|
0.45% Sodium Chloride Injection USP - VisIV
When administered intravenously these solutions provide a source of water and electrolytes. Solutions which provide combinations of hypotonic or isotonic |
|
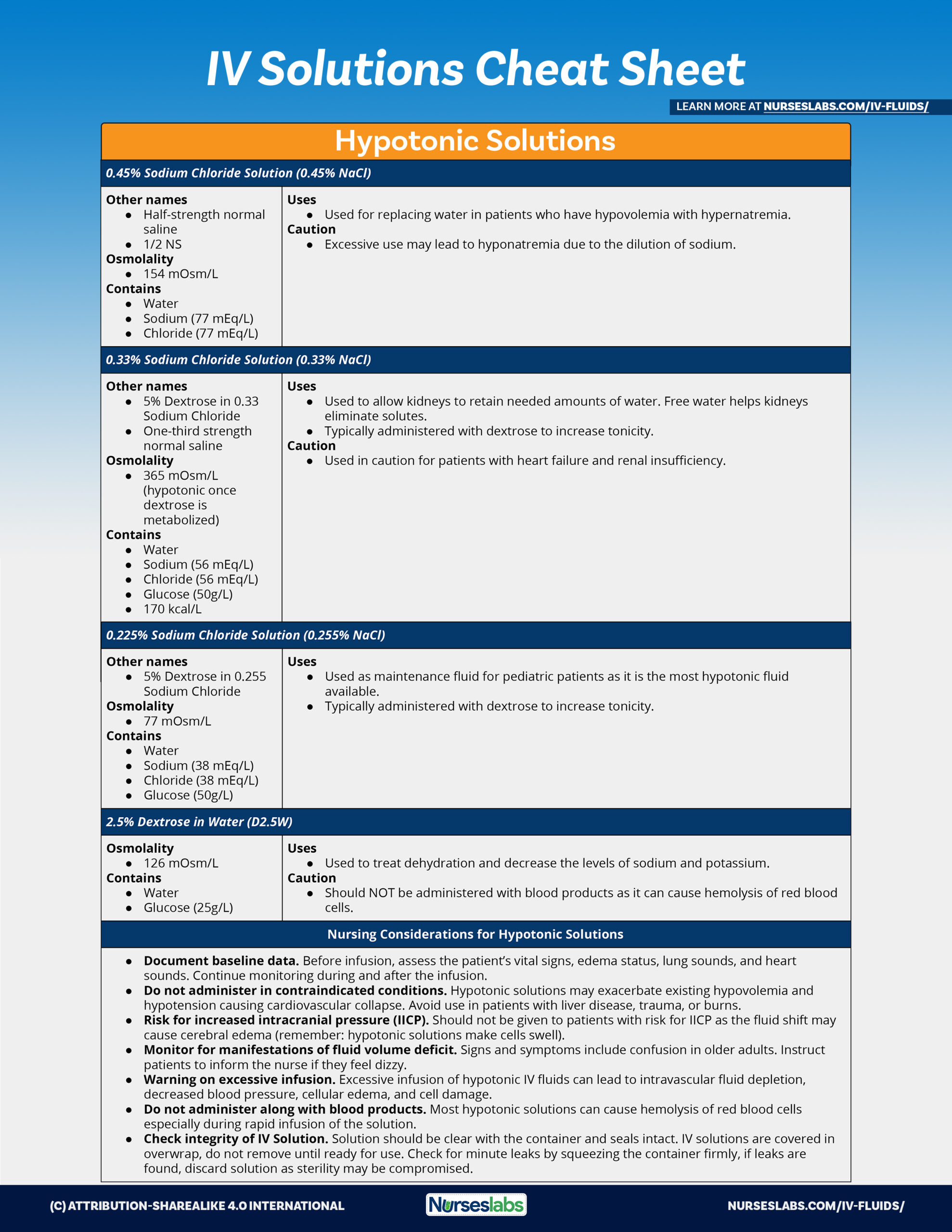
Potassium Chloride in 0.45% Sodium Chloride Injection USP in
10 Dec 2018 Potassium Chloride in 0.45% Sodium Chloride Injection USP is a sterile |
|
0.45% Sodium Chloride Injection USP
3 Dec 2018 0.45% Sodium Chloride Injection USP is a sterile |
|
Intravenous-fluid-therapy.pdf
10 Feb 2020 Normal saline (0.9% sodium chloride) which has been a life-saving ... 0.45% sodium chloride [D5. ½ NS] |
|
Intravenous (IV) Fluids
2 Dec 2022 Avoid in patients with liver disease trauma or burns. I.V. Fluid. Osmolarity. Composition. Uses/Clinical Considerations. 0.45% NaCl. (1/2 NS). |
|
ZITHROMAX®
1/2 Normal Saline (0.45% sodium chloride). 5% Dextrose in Water. Lactated Ringer's Solution. 5% Dextrose in 1/2 Normal Saline (0.45% sodium chloride) with 20 |
|
Pediatric Sodium Disorders
20 May 2016 ... 0.45% sodium chloride. (D5 1/2 NS).5. Hypertonic intravenous solution – an intravenous solution whose osmotically effective solutes result in ... |
|
NaCl 0.45% (% NS)
A. The LPN performing IV therapy must have successfully completed education and training provided by an IV therapy integated accredited nursing program or |
|
5% Dextrose and 0.45% Sodium Chloride Injection USP
DESCRIPTION. 5% Dextrose and 0.45% Sodium Chloride Injection USP solution is sterile and nonpyrogenic. It is a large volume parenteral solution. |
|
0.45% Sodium Chloride Injection USP - VisIV
When administered intravenously these solutions provide a source of water and electrolytes. Solutions which provide combinations of hypotonic or isotonic |
|
Making up intravenous fluids
0.45% Sodium Chloride 10% glucose 500ml bag. Method 1: If 0.45% Sodium Chloride with 5% dextrose is available a) Remove and discard 56 ml from a 500ml bag |
|
IV Fluids
02-Sept-2021 remaining saline and electrolytes act isotonic expanding the ... 0.45% NaCl. (1/2 NS) ... Dextrose 5% in 0.45%. NaCl. (D5 ½ NS). 406 mOsm/L. |
|
J-Code to National Drug Code (NDC) Billing Cross-Reference Guide
Ampicillin sodium per 1.5 gm. 1.5 GM. HCPC. CODE. NDC UNIT. QUALIFIER. NDC. STRENGTH HEPARIN SODPORK IN 0.45% NACL ... Normal saline solution infus. |
|
0.45% Sodium Chloride Injection USP
03-Dec-2018 0.45% Sodium Chloride Injection USP is a sterile |
|
Calculation Refresher Ratio and Proportion Method To solve for x
100 ml bag of normal saline (the “piggyback”). – Normal saline is 0.9% sodium chloride in water. Word Problem Conversion So is 0.5 ½ |
|
Intravenous-fluid-therapy.pdf
10-Feb-2020 *D5 1/2 NS + 20 KCl in children less than 1 year ... Normal saline (0.9% sodium chloride) which has been a ... 0.45% NaCl/Dextrose 5%. |
|
Potassium Chloride in 0.45% Sodium Chloride Injection USP in
10-Dec-2018 Potassium Chloride in 0.45% Sodium Chloride Injection USP is a sterile |
|
ACS/ASE Medical Student Core Curriculum Fluids and Electrolytes
Basic: Serum Sodium Potassium |
|
Chlorure de sodium injectable à 0,45 %, USP - Baxter
3 déc 2018 · chlorure de sodium (NaCl) et d'eau Le tableau 1 montre la Cl- Chlorure de sodium injectable à 0,45 , USP 1 000 00060186 4,5 77 77 154 voie intraveineuse à l'aide d'un équipement stérile Il est recommandé de |
|
Solution injectable de dextrose et de chlorure de sodium - Baxter
14 déc 2018 · Solution injectable de dextrose à 3,3 et de chlorure de sodium à 0,3 de chlorure de sodium, USP, est stérile et apyrogène et ne contient aucun agent 2 ,0 g 321 3,5 à 6,5 34 34 170 Dextrose à 5 et NaCl à 0,45 |
|
5% Dextrose and 045% Sodium Chloride Injection, USP - FDA
It is a large volume parenteral solution containing dextrose and sodium chloride in water for injection intended for intravenous administration Each 100 mL of 5 |
|
045% Sodium Chloride Injection, USP - FDA
A hypertonic concentration of sodium chloride may be used to repair severe salt depletion syndrome Sodium chloride in water dissociates to provide sodium (Na |
|
Intravenous fluids
when the serum sodium is low 150 150 50 0 45 NaCl with 5 dextrose and KCl 20 mmol/l (1/2 Normal saline with glucose and potassium) Use for mildly to |
|
Solutés de perfusion
Solutés: – salés isotoniques: chlorure de sodium 0,9 ou pH normal car compensation par Cristalloïdes: NaCl® isotonique ou hypertonique, 1-2 mL/ min |
|
IV Fluids - NursingCentercom
2 jan 2019 · Avoid in patients with liver disease, trauma or burns I V Fluid Osmolarity Composition Uses/Clinical Considerations 0 45 NaCl (1/2 NS) |
|
Making up intravenous fluids - BIMDG
9 sept 2016 · 0 45 Sodium Chloride 10 glucose 500ml bag Method 1: If 0 45 Sodium Chloride with 5 dextrose is available a) Remove and discard 56 |
|
NaCl 045% (% NS) - Mississippi Board of Nursing
MEDTCATTON(S) RECOMMENDED DOSAGE LIMITATIONS Sodium Chloride Solutions NaCl 0 225 (1/4 NS) 20 ml to 150 ml per hour NaCl 0 45 ( NS) |
|
Intravenous Fluid Therapy - Childrens Hospital Colorado
10 fév 2020 · of 120 mL/hr *D5 1/2 NS + 20 KCl in children less than 1 year (0 9 sodium chloride16) ½ NS (0 45 sodium chloride) Plasma- Lyte18 |


















![PDF] The effect of intravenous lactated Ringer's solution versus PDF] The effect of intravenous lactated Ringer's solution versus](https://els-jbs-prod-cdn.jbs.elsevierhealth.com/cms/attachment/3e8e7dad-7c04-4626-af76-1ff28ec217a4/gr1_lrg.jpg)











![PDF] (Ab)normal saline and physiological Hartmann's solution: a PDF] (Ab)normal saline and physiological Hartmann's solution: a](https://img.17qq.com/images/biihifgpofz.jpeg)





