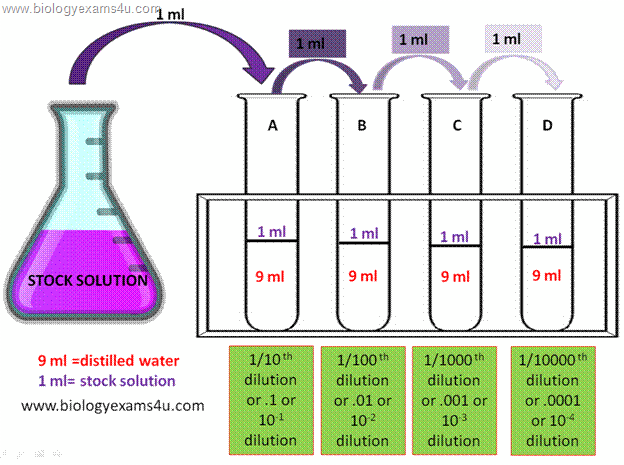
1/5 dilution factor
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
1 + 4 = 5 = dilution factor) Example 1: To dilute a streptavidin solution 1:300 Mix 1 unit volume of streptavidin solution with 299 volumes of solvent medium |
How do you calculate dilution factor?
The formula for dilution factor (or DF for short) is as follows: DF = (final volume of cells + stain)/(initial volume of cells).
For example, If you mix your sample 1:1 with AO/PI, you'll need to add 20 uL AO/PI to 20 uL cells, for a total of 40 uL.
So, DF = ( 40 uL)/(20uL cells) = 2.What does a 1 5 dilution mean?
For example, in a solution with a 1:5 dilution ratio, entails combining 1 unit volume of solute (the material to be diluted) with 5 unit volumes of the solvent to give 6 total units of total volume.
format, 1:4 means you have 1 part of sample for 4 parts of water, or 5 parts of water + sample in total.
To get from 1 part to 5 parts you need to multiply by 5.
|
PREPARING SOLUTIONS AND MAKING DILUTIONS
1:5 dilution (verbalize as "1 to 5" dilution) entails combining 1 unit volume of diluent ( factor). Example 1: To dilute a streptavidin solution 1:300. |
|
DILUTION CHART.pdf
MIXTURE. Dilution Chart. 1:1. 1:5 1:10 1:20 1:25 1:50 1:100 1:150 1:200 1:250 1:500 1:1000. 500ml 200ml 100ml 50ml. |
|
Dilutions Help
“The dilution factor is 5". “It was a 5 fold dilution”. “It was diluted 1/5". These all mean the same thing that there is 1 volume part of sample and 4 |
|
Dr. Gs DILUTION PRIMER
this is a 1 to 5 dilution or 1:5 dilution. This process can be used to determine how to perform any dilution. All you need to know is what the dilution factor |
|
Laboratory Math II: Solutions and Dilutions
The dilution factor is the factor by which you will dilute your sample at each step. Mathematically it is equal to the sum of the volume of your stock solution |
|
Dilutions Occasionally a solution is too concentrated to be used as it
dilution factor and so on until the final concentration is known. Example: A 5M solution of HCl is diluted 1/5. The resulting solution is diluted 1/10. |
|
1. dilutions and concentrations. liquid and solid samples
Concentration Factor. 1.6. We have prepared the followings dilutions from different sample: 1:10 1:5 |
|
IDEXX VetTest Chemistry Analyzer Dilution Protocol: Urine Plasma
Dilution Factor: The total number of parts in the diluted sample; the. VetTest analyzer automatically multiplies the result by this number to correct. |
|
2 Amount and concentration: making and diluting solutions
In all cases you are diluting by the same factor. 1:5 dilution = 1 unit volume of diluent + 4 unit volumes of solvent;. ? dilution factor = 5. |
|
Comparison of four matrixes for diluting insulin in routine clinical
13 avr. 2020 We calculated that using the original diluent (valid- ity: 21 days 10 mL; dilution factor: 1:5; 40 ?L) to dilute high-insulin serum could result ... |
|
Dilutions Occasionally a solution is too concentrated to be used as it
dilution factor, and so on until the final concentration is known Example: A 5M solution of HCl is diluted 1/5 The resulting solution is diluted 1/10 Determine |
|
Dilution Factor:
Dilution Factor: A water sample was tested (with dithizone in methylene chloride to make a color solution) for lead content, but was diluted prior to obtaining the |
|
261_Labs_Making Solutions and Dilutions - Penguin Prof Pages
dilution factor is the total number of unit volumes in which your material will be For example, a 1:5 dilution (verbalize as "1 to 5" dilution) entails combining 1 |
|
Dr Gs DILUTION PRIMER
this is a 1 to 5 dilution or 1:5 dilution This process can be used to determine how to perform any dilution All you need to know is what the dilution factor is, and |
|
Laboratory Math II: Solutions and Dilutions - NIH Office of Intramural
The dilution factor is the factor by which you will dilute your sample at each step Mathematically it is equal to the sum of the volume of your stock solution you are |
|
How to Make Simple Solutions and Dilutions
l Simplc Dilution (Dilution Factor Nlethod based on ratios) A sinple diluted) + { unit volumes ofthe solvent medium (hence, + 4= 5 = dilution factor) The |
|
DILUTION CHART NEW - Caprichem
DILUTION CHART 1:1 1:5 1:10 1:20 1:25 1:50 1:100 1:150 1:200 1:250 1:500 1: 1000 CHEMICAL 500ml 200ml 100ml 50ml 40ml 20ml 10ml 7ml 5ml 4ml 2ml |