ketoconazole hra notice-eu - Chirurgie
|
Ketoconazole HRA INN-ketoconazole
Ketoconazole HRA was designated as an orphan medicinal product EU/3/12/965 on 23 April 2012 Ketoconazole HRA was designated as an orphan medicinal product in the following indication: treatment of Cushing syndrome |
|
Ketoconazole HRA INN-ketoconazole
Ketoconazole HRA in the approved indication remains favourable and therefore recommended the renewal of the marketing authorisation with unlimited validity PSUSA/10316 /201811 Periodic Safety Update EU Single assessment - ketoconazole (centrally authorised product only) 14/06/2019 n/a PRAC Recommendation - maintenance |
|
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
Ketoconazole HRA should be stopped immediately and should not be reintroduced due to the risk of serious hepatic toxicity Ketoconazole HRA should be discontinued without any delay if clinical symptoms of hepatitis develop In case of long term treatment (more than 6 months): |
Does ketoconazole cause hepatoxicity?
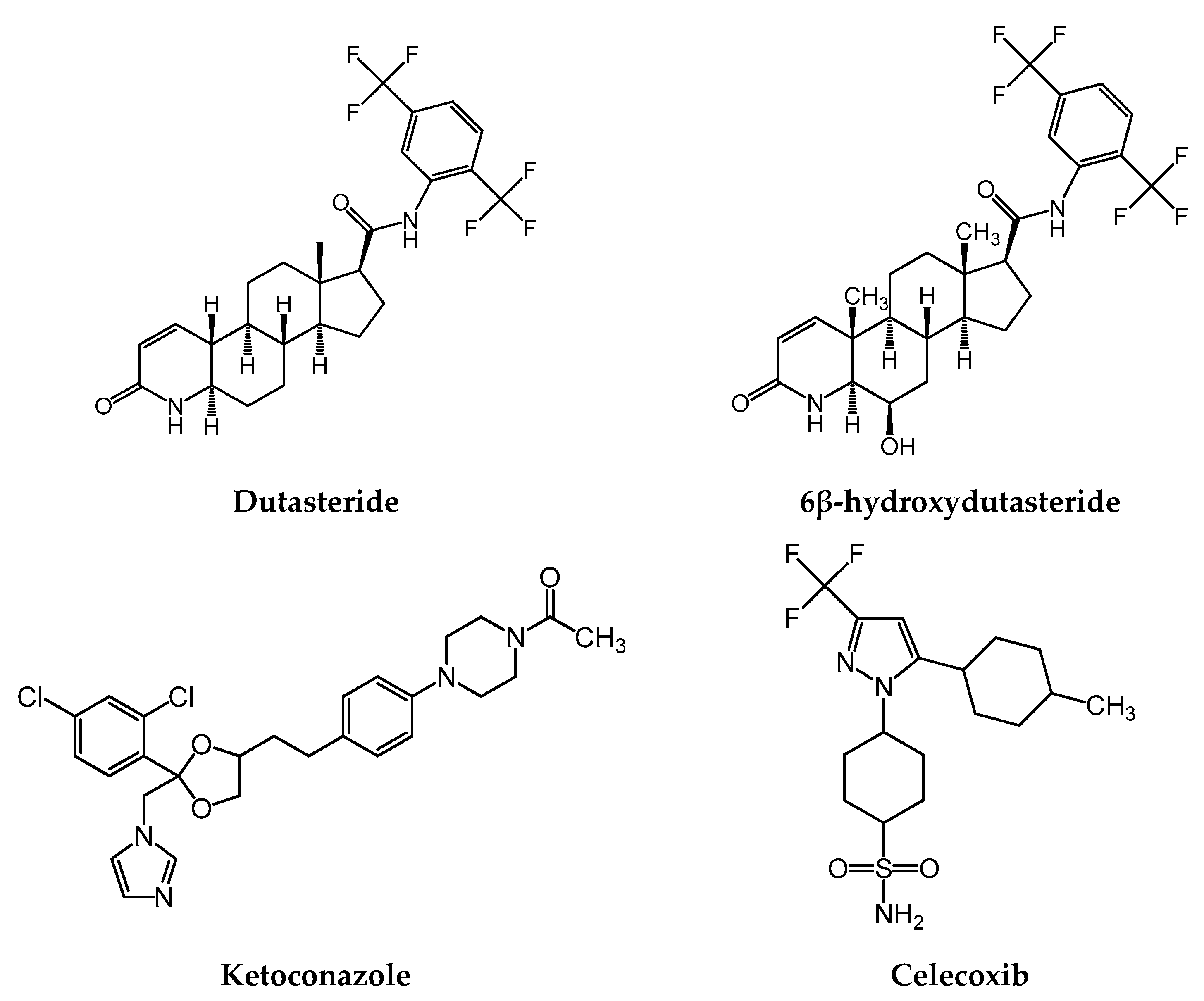
In total two cases out of 24 developed severe hepatoxicity including one case of fatal liver failure due to ketoconazole was reported in a 14 year-old girl while treated for Cushing’s disease with ketoconazole 200 mg twice daily (Zollner, 2001) and this is included in section 4.8 of the SmPC.
Does ketoconazole inhibit hERG?
Moreover, one report has shown that ketoconazole directly inhibits the hERG current and reduces the surface membrane expression of hERG channels by disrupting channel protein trafficking (Takemasa, 2008). Please refer to module 2.4 and module 2.6.2 for further preclinical and clinical data presentation and discussion.
Is ketoconazole HRA an orphan medicinal product?
Ketoconazole HRA was designated as an orphan medicinal product EU/3/12/965 on 23 April 2012. Ketoconazole HRA was designated as an orphan medicinal product in the following indication: treatment of Cushing syndrome. The applicant applied for the following indication: Treatment of Cushing’s syndrome.
What is the European public assessment report (epar) for ketoconazole HRA?
This is a summary of the European public assessment report (EPAR) for Ketoconazole HRA. It explains how the Agency assessed the medicine to recommend its authorisation in the EU and its conditions of use. It is not intended to provide practical advice on how to use Ketoconazole HRA.
|
Ketoconazole HRA INN-ketoconazole
Ketoconazole HRA est indiqué dans le traitement du syndrome de Cushing demande accrue en glucocorticoïdes (par ex. en cas de stress chirurgie ou ... |
|
Ketoconazole-hra-epar-product-information_fr.pdf
Ketoconazole HRA est indiqué dans le traitement du syndrome de Cushing demande accrue en glucocorticoïdes (par ex. en cas de stress chirurgie ou ... |
|
Ketoconazole HRA INN-ketoconazole
une chirurgie). Contrôle de la fonction hépatique. Avant de commencer le traitement il est obligatoire : - de doser les enzymes hépatiques (ASAT |
|
Braftovi INN-encorafenib
Feb 11 2019 des modèles pour le kétoconazole suggèrent une augmentation multipliée par environ ... Une interruption ou une modification de la dose a eu. |
|
Abraxane INN-paclitaxel
chirurgie potentiellement curative et/ou une radiothérapie. des médicaments inhibiteurs du CYP2C8 ou du CYP3A4 (kétoconazole et autres antifongiques. |
|
COMMISSION DE LA TRANSPARENCE
Jul 20 2016 KETOCONAZOLE HRA |
|
RELEVE DE DECISIONS
Nov 12 2014 juin 2014 et visant à créer deux actes de chirurgie par coelioscopie ... sociale de la spécialité KETOCONAZOLE HRA 200 mg (kétoconazole). |
|
Services de santé non assurés : Liste des médicaments - septembre
Sep 20 2020 1. le formulaire de Santé Canada: Notification concernant un effet indésirable ... le patient a eu une récidive clinique et/ou il y a eu une. |
|
FORMULAIRE THERAPEUTIQUE TUNISIEN
les patients ayant subi une angioplastie ou une chirurgie de revascularisation. cisapride par certains médicaments (e.a. kétoconazole itraconazole |
|
Journal N°102 Janvier-Mars 2015 _V4_
Kétoconazole HRA® : Risque d'hépatotoxicité . non concluante puisqu'il n'y a pas eu de ... l'atenolol : dissection aortique (2 vs 0) |




![Synthesis of Ketoconazole Derivatives - [PDF Document] Synthesis of Ketoconazole Derivatives - [PDF Document]](https://www.yumpu.com/fr/image/facebook/16731618.jpg)