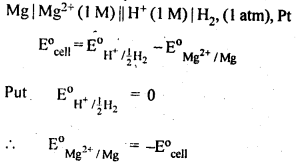electrochemistry solutions class 12
What is NCERT solutions for Class 12 chemistry Chapter 3?
NCERT Solutions for Class 12 Chemistry Chapter 3 Electrochemistry play a pivotal role in the CBSE Class 12 Chemistry board examination. NCERT Solutions for Class 12 Chemistry are comprehensive materials that have answers to the exercise present in the NCERT Textbook.
Are Vedantu solutions useful for Class 12 chemistry students?
With Vedantu's Electrochemistry Class 12 NCERT solutions, students can enhance their understanding of electrochemistry and improve their performance in examinations. Overall, Vedantu's solutions are a valuable tool for Class 12 Chemistry students. Is this page helpful? 1. What is Electrochemistry all About?
What is Ch 3 chemistry class 12?
Ch 3 Chemistry Class 12 is quite a tough chapter so students can make use of Electrochemistry Class 12 solutions. After studying the chapters students can begin their preparation on past year papers and sample papers. These will help students to gain confidence and experience in solving these types of problems.

Electrochemistry Class 12 Chemistry Chapter 3 NCERT Solutions Questions 3.1 to 3.9

Electrochemistry Class 12 One Shot Class 12 Board Exam Preparation Arvind Sir

Electrochemistry Exercise Solutions Class 12 Board Exam
|
TOPIC 6: ELECTROCHEMISTRY
Grade 12 Chemistry • Topic 6: Electrochemistry. SUGGESTIONS FOR INSTRUCTION. Entry-Level Knowledge. In Topic 1: Reactions in Aqueous Solutions |
|
CBSE NCERT Solutions for Class 12 Chemistry Chapter 3
[1 mark]. Solution: Page 2. Class- XII-CBSE-Chemistry. Electrochemistry. Practice more on Electrochemistry. Page - 2 www.embibe.com. Substances that are |
|
NCERT Solutions for Class 12 Chemistry Chapter 3 - Electrochemistry
NCERT Solutions for Class 12 Chemistry Chapter 3. Electrochemistry. Q 3.1: Arrange the following metals in the order in which they displace each other from |
|
Test4 ch19 Electrochemistry Practice-answers-Marked
Spontaneous Voltaic Electrochemical Cells p4 Nonstandard Concentrations and Cell Potential p11. Cell Potentials p5 Electrolysis p12. |
|
NCERT Exemplar Solution of Class 12 Chemistry Chapter 3
(iv) It provides a surface for a redox reaction. Solution: Option (iv) is the answer. 6. An electrochemical cell can behave like an electrolytic cell when |
|
Grade 12 Chemistry: A Foundation for Implementation
21 janv. 2011 Contents • Grade 12 Chemistry. Grade 12 Chemistry. Topic 1: Reactions in Aqueous Solutions 1. Topic 2: Atomic Structure 1. |
|
Electrochemistry Electrochemistry
In dilute solutions it is equal to concentration. You will study more about it in higher classes. 3.1 Electrochemical. Electrochemical. Cells. |
|
Chemistry Class 12 Chapter 3 NCERT Solution-electrochemistry
Class XII. Chapter 3 – Electrochemistry. Chemistry. Page 1 of 25. Chemistry Class 12 Chapter 3 NCERT Solution www.mywayteaching.com. |
|
CBSE Test Paper-01 Class 12 Chemistry (Electrochemistry) 1. Given
Predict the products of electrolysis in the following: A solution of H2SO4 with platinum electrode. 14. Zinc rod is dipped in 0.1 M solution of ZnSO4. The salt |
|
Chemistry Notes for class 12 Chapter 3 Electrochemistry .pdf
The solution remains neutral on both sides. 3. The reaction and now of electrons stops after sometime. Daniell Cell. An electrochemical cell of zinc and copper |
|
Chemistry Class 12 Chapter 3 NCERT Solution - Mywayteaching
(i) Zn electrode (anode) is negatively charged Class XII Chapter 3 – Electrochemistry Chemistry Page 1 of 25 Chemistry Class 12 Chapter 3 NCERT Solution |
|
Chemistry Notes for class 12 Chapter 3 Electrochemistry - Ncert Help
The solution remains neutral on both sides 3 The reaction and now of electrons stops after sometime Daniell Cell An electrochemical cell of zinc and copper |
|
Electrochemistry - NCERT
solutions, it is equal to concentration You will study more about it in higher classes 3 1 Electrochemical Electrochemical Cells Fig 3 1: Daniell cell having |
|
Electrochemistry - Mahesh Tutorials Science
12 SPONTANEITY OF A REACTION ∆G = – nFE CELL For a spontaneous cell reaction ΔG should For example, H2 gas is used with a dilute solution of HCl |
|
CHEMISTRY CLASS-XII - edudel
10 Unit II Solutions 10 Unit III Electrochemistry 12 23 Unit IV Chemical Kinetics 10 Unit V Surface Chemistry 08 Unit VI General Principles and Processes |
|
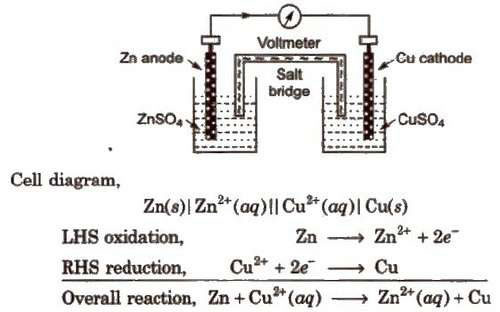
Class-XII Chapter 3: Electrochemistry Electrochemistry is that branch
Salt-bridge generally contains solution of strong electrolyte such as KNO3, KCl etc When an electrode is in contact with the solution of its ions in a half-cell, it has |
|
NCERT Exemplar Solution of Class 12 Chemistry Chapter 3 - Byjus
NCERT Exemplar Solution of Class 12 Chemistry Chapter 3 Electrochemistry I Multiple Choice Questions (Type-I) 1 Which cell will measure standard |
|
NCERT SOLUTIONS CLASS-XII CHEMISTRY CHAPTER-3
In the order of their reactivity, i e how they displace each other from their salt solutions, allign the metals in decreasing order Cu, Fe, Al, Zn and Mg Answer: |