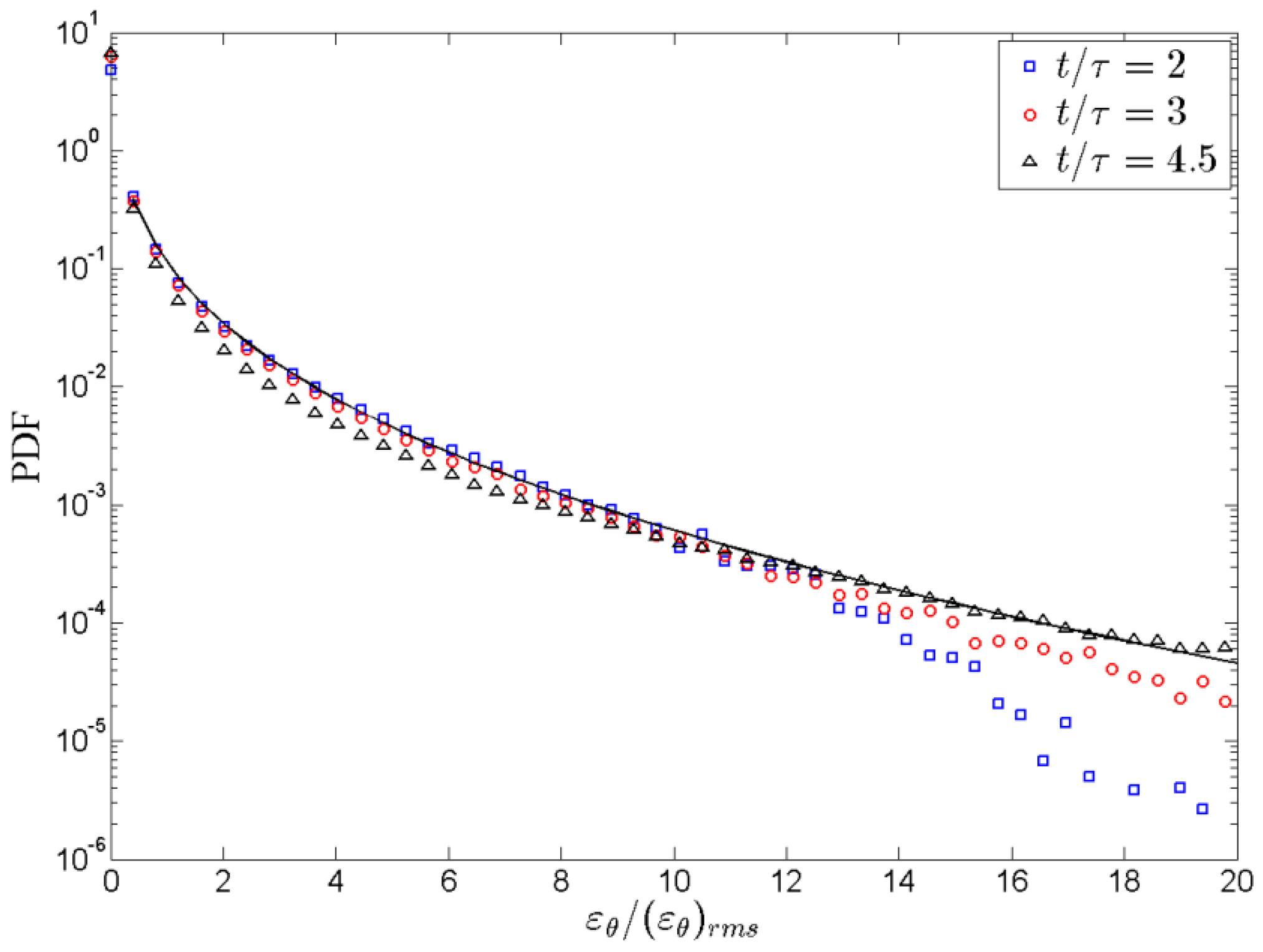
entropy of mixing
|
Entropy of mixing gas
An adiabatic free expansion of an ideal gas i e where a greater volume suddenly becomes available to the gas is an irreversible process which proceeds |
Does entropy increase with mixing?
When two pure substances mix under normal conditions there is usually an increase in the entropy of the system.
This is qualitatively easily visualised in terms of the increased disorder brought about by mixing.What is the formula for the entropy of mixing?
If you had 1 mole each of two ideal gases at the same temperature and pressure, the entropy of mixing the two gases would be: Δ m i x S = − 2 R ( 1 2 ln 1 2 ) = R ln
The enthalpy of mixing (or heat of mixing or excess enthalpy) is the enthalpy liberated or absorbed from a substance upon mixing.
When a substance or compound is combined with any other substance or compound theenthalpy of mixing is the consequence of the new interactions between the two substances or compounds.
|
Entropy and Mixing for Amenable Group Actions
Entropy and mixing for amenable group actions. By DANIEL J. RUDOLPH and BENJAMIN WEISS*. Abstract. For r a countable amenable group consider those actions |
|
Excess Entropy of Mixing of Liquids
where X is the mole fraction. (IV) The mixture of rigid spheres is compressed to the volume of the solution vt. The entropy |
|
Gibbs paradox of entropy of mixing: experimental facts its rejection
Gibbs paradox statement of entropy of mixing has been regarded as the theoretical foundation of statistical mechanics quantum theory and biophysics. |
|
Entropy of mixing gas Masatsugu Sei Suzuki Department of Physics
Entropy change of mixing gas. ((Kubo Thermodynamics)). Two kinds of ideal gases at equal pressure and temperature |
|
Entropy of mixing exists only for classical and quantum-like theories
In this paper we consider thermodynamical entropy of mixing in a specific series of theories of GPTs called the regular polygon theories |
|
Combinatorial Entropy of Mixing for Molecules Differing in Size and
In reducing experimental vapor-liquid equilibrium data it is common to calculate the Gibbs energy of mixing in excess of that for a solution whose entropy |
|
Entropy of Mixing: Gibbs Paradox
Entropy of Mixing: Gibbs Paradox. Programme: B. Sc. Physics. Semester: VI. Dr. Ajai Kumar Gupta. Professor. Department of Physics. Mahatma Gandhi Central. |
|
IV. Polymer Solutions and Blends
To characterize enthalpy of mixture the interaction parameter ? is introduced: The contribution of a polymer to entropy of mixing is ~1/N ... |
|
V. Ihnatovych
Where pi – partial pressure of i-th gas in a mixture. 3. Derivation and the preliminary analysis of formulas for entropy of mixing of ideal gases. At mixing of |
|
Entropy of mixing revisited [tln50]
Entropy of mixing revisited [tln50]. Consider two dilute gases in a rigid and insu- lating box separated by a mobile conducting. |
|
Entropy of mixing - Pradeep Research Group
by diffusion), always results in increase in entropy Mixing is spontaneous This is in general true of any material, liquid or solid Spontaneous processes → net increase in entropy → increase in randomness of distribution (mixing) |
|
Gibbs paradox of entropy of mixing - Molecular Diversity
Gibbs paradox statement of entropy of mixing has been regarded as the theoretical foundation of statistical mechanics, quantum theory and biophysics A large |
|
(Lec 3 Solution Models)
Molar Gibbs free energy of mixing ∆HM Molar enthalpy of mixing ∆SM Molar entropy of mixing ∆eG Excess Gibbs free energy per mole of solution ∆eH |