acids and bases practice problems with answers
|
Practice Problems on Acid-‐Base Chemistry -‐ Answers
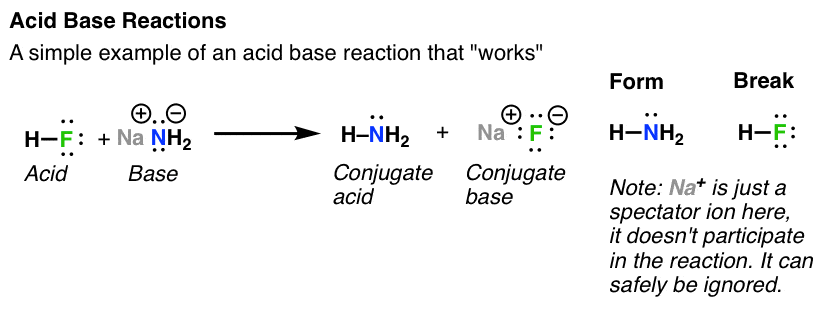
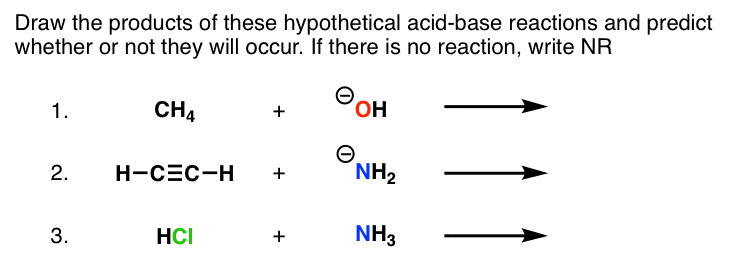
Practice Problems on Acid-‐Base Chemistry -‐ Answers 1 For each of the following acid base reactions place one of the arrows inside |
|
Acid Base Practice Test
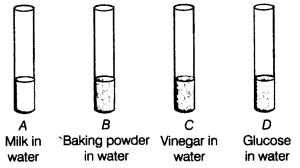
Acid Base Practice Test ____ 1 Acids taste a sweet c bitter b sour d M KOH solution? a 3 c 9 b 5 d 11 ____ 19 What is the molarity of an HCl |
|
Acids and Bases Extra Practice Problems
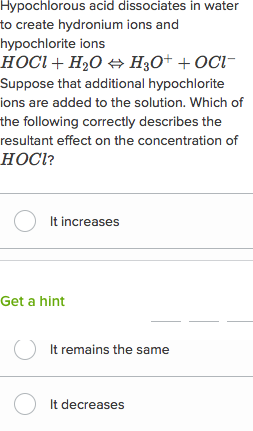
Extra Practice Problems Using Dissociation Constants What is the difference Solution The strong base will have the lowest [Hj; strong base weak base |
|
Acids and Bases
Practice Test: Acids and Bases Answer using a #2 pencil Version A Bubble the correct answer on the scantron Page 2 14 When you test a substance with a pH |
|
Acids and Bases
Chemistry Acids and Bases Practice Problems ANSWERS 1a NaOH → Na1+ + OH1 pH = 13 (base) 1b pH = 13 2a pH = - log [H1+] 5 = - log [H1+] (on your |
|
ACIDS PRACTICE pKa and pKb SMOsNotes
1 Methanoic acid (HCOOH) is a weak acid with pKa=3 75 (a) Calculate the pH of a 0 100 mol dm-3 solution of methanoic acid [3] Ka |
|
Solution Equilibrium (Acids Bases Buffers and Hydrolysis
Page 1 Page 2 Page 3 Page 4 Page 5 Page 6 Page 7 Page 8 Page 9 Page 10 Page 11 Page 12 Page 13 Page 14 Page 15 |
|
Organic chemistry i practice problems for bronsted-lowry acid-base
PRACTICE PROBLEMS FOR BRONSTED-LOWRY ACID-BASE CHEMISTRY 1 For each of the ANSWERS 1 HF CH3CH2OH H3O H2O CH3CH3 CH3CN HC CH H2 CH3OH2 RNH3 F |
|
UNIT 3: ACIDS AND BASES
Four possible answers are given for the following questions Each question has only ONE correct answer Choose the correct answer and mark the applicable |
What are the questions on the acid test?
Definition.
The Supreme Court has now confirmed that to determine whether a person is deprived of their liberty, there are 2 key questions to ask, described as the 'acid test': Is the person free to leave.
Is the person subject to continuous supervision and control.What is acid question?
An acid is a substance that donates protons or accepts electrons.
It dissociates in an aqueous solution to give H+ ions.
A base is a substance that abstracts protons or donates electrons.What is the strongest acid question?
The strongest of them all
That title falls to fluoroantimonic acid – a superacid mixture of antimony pentafluoride and hydrofluoric acid.K+ is the conjugate acid of the strong base KOH.
|
Jasperse Acid-Base Chemistry. Extra Practice Problems
The base ionization constant Kb describes which of the following reactions for a weak base B |
|
Practice Problems Section 11.1 – Acids and Bases Goal
A buffer solution maintains pH by neutralizing small amounts of an acid or base. Most buffer solutions consist of nearly equal concentrations of a weak acid and |
|
Chemistry additional sample examination questions
Support your answer with a labelled diagram. Mapping grid: Content. Syllabus outcomes. Bands. Mod 7 Reactions of Organic Acids and Bases. CH12–4 CH12–7 |
|
Acid-Base Chemistry. Extra Practice Problems
ANSWERS. Acid-Base Chemistry. Extra Practice Problems. 1. A. 2. B. 3. A. 4. D. 5. B. 6. B. 7. A. 8. C. 9. B. 10. B. 11. B. 12. C. 13. A. 14. E. 15. C. 16. D. 17 |
|
Weak Acids and Bases Practice -- Chemistry 121A Hanson OK
A 0.10 M solution of an acid HA is found to be 3.5% ionized. What is the Ka of Google weak acid practice or weak base problems or such. You will find plenty! |
|
Organic Chemistry Jasperse Acid-Base Practice Problems
Acid-Base Practice Problems. A. Identify each chemical as either an “acid” or a “base” in the following reactions and identify “conjugate” relationships |
|
1. For each of the species below identify the most acidic proton and
PRACTICE PROBLEMS FOR BRONSTED-LOWRY ACID-BASE CHEMISTRY. 1. For each of the ANSWERS. 1. HF. CH3CH2OH. H3O. H2O. CH3CH3. CH3CN. HC CH. H2. CH3OH2. RNH3. F. |
|
Stanley Acids and Bases Study Guide
Examples are given with solutions/answers some explanations are provided This may assist especially when answering multiple choice questions. Base 1. Acid 1. |
|
Topic 4 – Acid-Base Neutralization Reactions
Practice Examples a. What volume of 2.0 M HNO3 is needed to neutralize 40. ml of 5.0 M NaOH? b. In a titration 27.4 mL of 0.0154 |
|
Ch_15 Acid Base Eq - Buffer Practice Problems - key.pdf
Questions 5-8 refer to these answers. (a) a solution A certain buffer solution contains equal concentration of the weak acid HX and it conjugate base ion. |
|
Test2 ch17a Acid-Base Practice Problems
p4 Answers p12. Ka: Sense + Calculations. Using Ka or pKa to Calculate. [H+] and/or pH; using pH to calculate Ka or pKa p5. Conceptual Questions. Acids |
|
Organic Chemistry Jasperse Acid-Base Practice Problems
Acid-Base Practice Problems. A. Identify each chemical as either an “acid” or a “base” in the following reactions and identify “conjugate” relationships. |
|
Arterial Blood Gas Case Questions and Answers
26 Base Excess 1. On his chemistry panel |
|
Chapter 11 - Acids and Bases - Practice Problems - Section 11.1
by one H. Conjugate acid-base pairs: H2SO4/HSO4 andH3O+ /H?O. El. Practice Problems. |
|
Chapter 11 – Acids and Bases – Practice Problems Section 11.1
When H2O acts as a base it gains one H+ |
|
Acid Base Practice Test - Houston ISD
neither acids salts |
|
Weak Acids and Bases Practice -- Chemistry 121A Hanson OK
A 0.10 M solution of a weak base B is found to have pH 9.20. What is the Kb of this base? Want more? Google weak acid practice or weak base problems or such. |
|
Access Free Chapter 20 Acids Bases Practice Problems Answers
2 days ago Acid-Base Equilibrium Practice Problems. Here are two hints about these problems in general: When it gives you a concentra- tion of acid in the ... |
|
Practice Set Answer Keys Organic Chemistry I Table of Contents
Acid-Base Practice Problems. A. Identify each chemical as either an “acid” or a “base” in the following reactions and identify “conjugate” relationships. |
|
Sample Exercise 16.1 Identifying Conjugate Acids and Bases
conjugate base of the acid H2O. Practice Exercise. Solution Note: In this problem and all that follow we assume |
|
Ch 17a Acid-Base Practice Problems
se Chemistry Extra Practice Problems between pH and pOH p4 Answers p12 In the Brønsted–Lowry definition of acids and bases, an acid ______ a is a proton donor |
|
Acid-Base Practice Problems
Chem341PDF |
|
Chapter 11 – Acids and Bases – Practice Problems Section
O acts as a base, it gains one H+, which forms its conjugate acid, H3O+ Practice Problems 8 |
|
Practice Problems on Acid-‐Base Chemistry -‐ Answers 1 For
e Problems on Acid-‐Base Chemistry -‐ Answers 1 For each of the following acid base reactions |
|
Solutions to Review Problems for Acid/Base Chemistry - CSUN
mL of 6 0 M HCl are added to one sample, and 5 0 mL of 6 0 M NaOH are added to the other, what is |
|
Acid Base Practice Test - Houston ISD
se Practice Test ____ 1 Acids taste Which of the following chemical reactions represents an acid-base reaction? a HBr + KOH → KBr + Base Practice Test Answer Section |
|
Chem 1B Practice problems for Exam 2 Once again, this is not a
s acid, Bronsted-Lowry acid d) Lewis base, Bronsted-Lowry base Answer d 2 Identify from the |
|
Sample Exercise 161 Identifying Conjugate Acids and Bases
he reaction that occurs, and identify the conjugate acid–base pairs Note: In this problem and all that follow, we assume, unless stated otherwise, Answers: (a) 3 42, (b) [H+] = 5 3 ×10–9 M, so pH = 8 28 |