ester react with sodium hydroxide
Does NaOH react with ester?
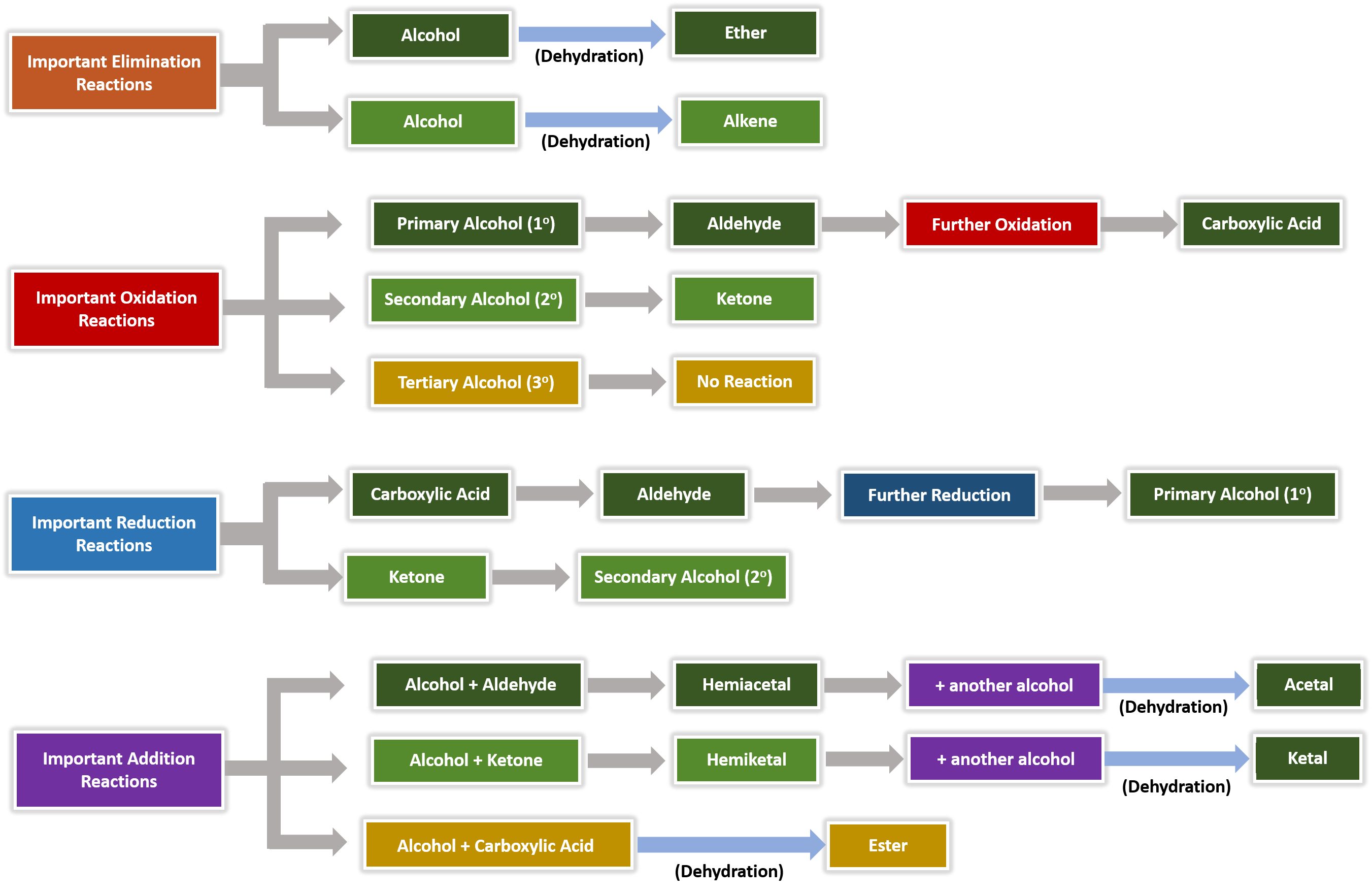
Ester Hydrolysis with NaOH or base catalysed ester hydrolysis is the reaction of an ester with water under a basic medium.
In it, an ester is heated under reflux with dilute NaOH to yield carboxylate salt and alcohol.
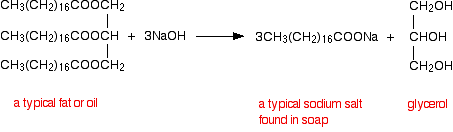
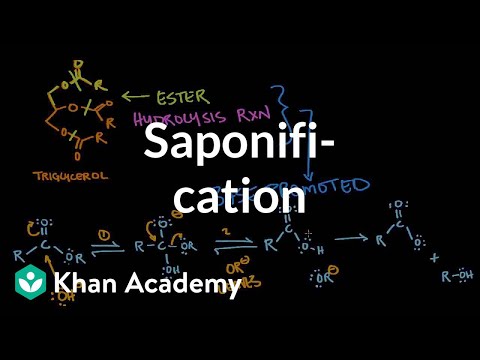
It is also known as saponification reaction, i.e. it is used to synthesise soap.
|
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters and learn how to sodium hydroxide (NaOH) and potassium hydroxide. |
|
Transesterification of Vegetable Oils with Ethanol and
Jun 5 2009 The ester preparation involved a two-step transesterification reaction |
|
SOME CONDENSATION REACTIONS INVOLVING THE
benzyl cyanide with methylene iodide in the presence of sodium hydroxide. The explanation of these peculiar reactions involving the elimination of ester |
|
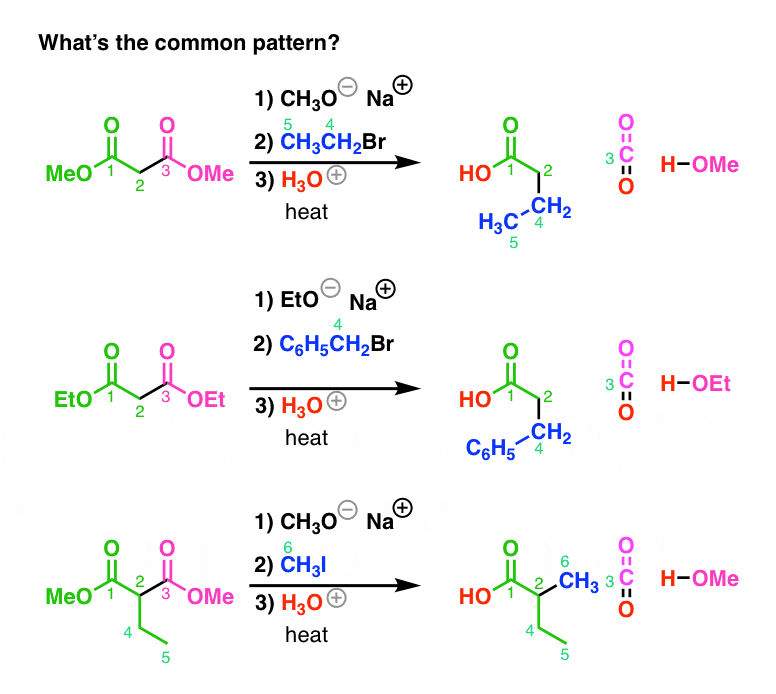
The Action of Sodium Methoxide on Methyl Esters1
of bromide ion to have been released. Reactions of Esters with Methanolic Sodium Hydroxide. —To a reaction flask containing a sealed ampoule of dry ester |
|
Effects of Sodium Hydroxide Pretreatment on Structural Components
The alkaline pretreatment reacts with the ester bonds linking the lignin to the hemicellulose in the LCC network. As these bonds are broken the LCC networking |
|
THE REACTION BETWEEN METALLIC SODIUM AND AROMATIC
Esters of aromatic acids react likewise |
|
THE ADDITION OF SODIUM MALONIC ESTER TO ALIPHATIC
in obtaining a complete reaction between sodium and the methyl ester. With cold aqueous sodium hydroxide it is possible to obtain one of the. |
|
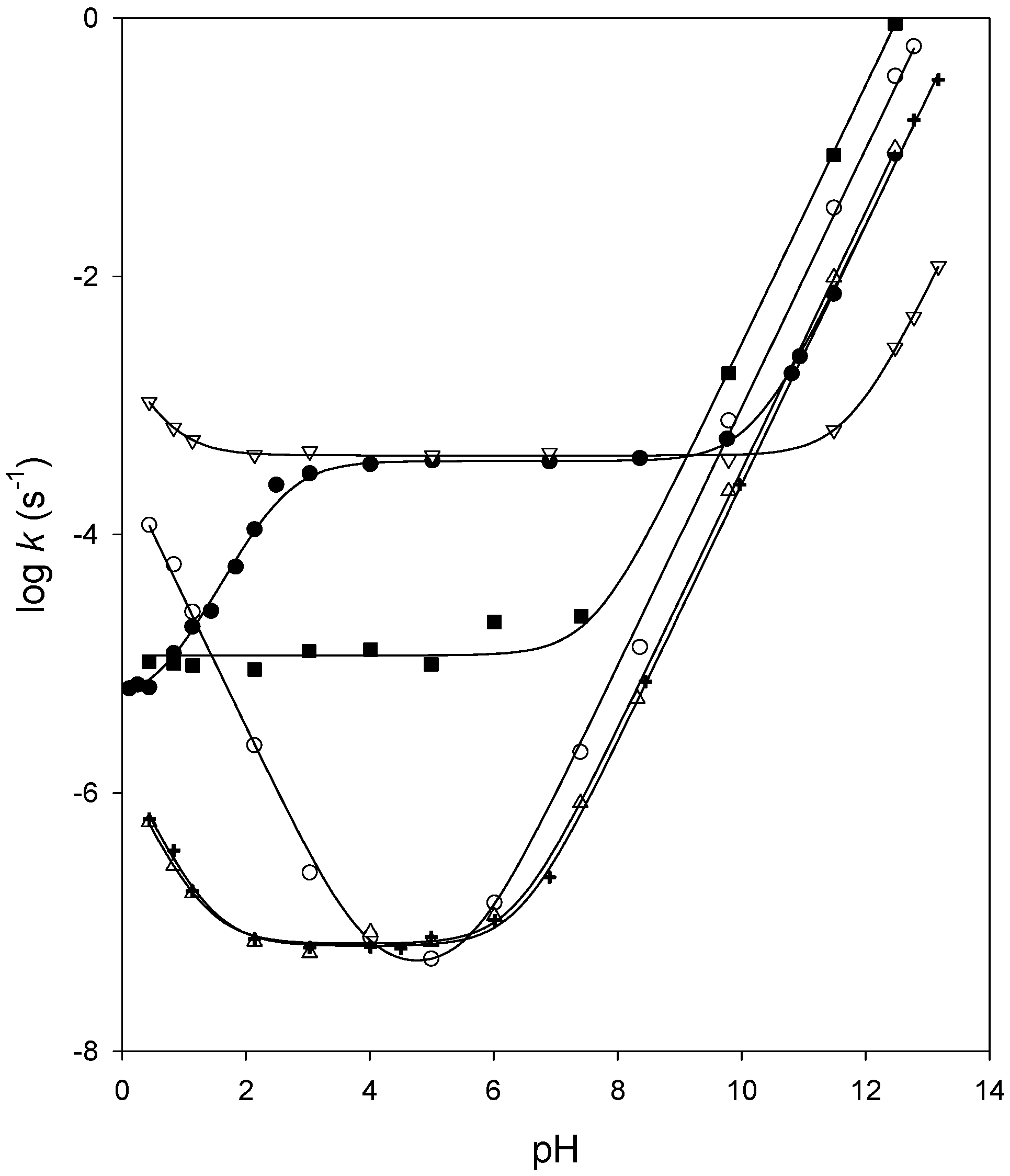
Reaction rate and rate constant of the hydrolysis of ethyl acetate with
In this hydrolysis of ester (ethyl acetate) with an alkali (sodium hydroxide) HCl was used as catalyst to accelerate it. 1ml and 2ml of ethyl acetate was |
|
Lab 14: Qualitative Organic Analysis
deprotonated by NaOH to form water-??soluble alkoxides. Only strong acids like carboxylic This reaction tests for the presence of amides and esters. |
|
Saponification of Ethyl Acetate
The rate constant k for chemical reactions is given by the. Arrhenius equation: fication of an ester by sodium hydroxide. |
|
Saponification of Ethyl Acetate
The rate constant k for chemical reactions is given by the Arrhenius equation: k = se-48a/RT fication of an ester by sodium hydroxide CH3COOC2H5 + Na+ + |
|
Benzoic Acid from Ethyl Benzoate by Base Hydrolysis
with aqueous sodium hydroxide, the reaction going to completion, to give sodium This process is called base hydrolysis (or saponification) of an ester and is |
|
39 Carboxylic acids and esters
+ NaOH → CH3CO2Na + H2O Sodium ethanoate 2CH3CO2H + Na2CO3 → 2CH3CO2Na + H2O + CO2 Sodium ethanoate Questions: 1) For the reactions |
|
Metal ion complex catalysis of amino acid ester hydrolysis - CORE
the reactions written for the base hydrolysis of esters is 2 one in which the 0 0067 M ester and 0 050 M KNOg with 0 206 M NaOH solution The pH of the |
|
Revisiting the mechanisms of low-temperature, base-catalysed ester
Abstract: Ester interchange reactions such as the interesterification of triglycerides and titative saponification reaction between the sodium hydroxide formed |
|
IV SEMMESTER
The hydrolysis of an ester occurs according to the equation (acetic acid) are titrated against a strong base (NaOH), strong acid reacts first followed by a |
|
Chemical Properties and Derivatives of Glycerol - Science The
or sodium dichromate and chromic acid oxidize glycerol the presence of potassium or sodium hydroxide(l4), Esters can be prepared by reacting glycerine |


















![Chapter 5 Carboxylic Acids and Esters - [PDF Document] Chapter 5 Carboxylic Acids and Esters - [PDF Document]](https://d10lpgp6xz60nq.cloudfront.net/physics_images/GRB_ORG_CHM_P2_C12_E01_083_S02.png)

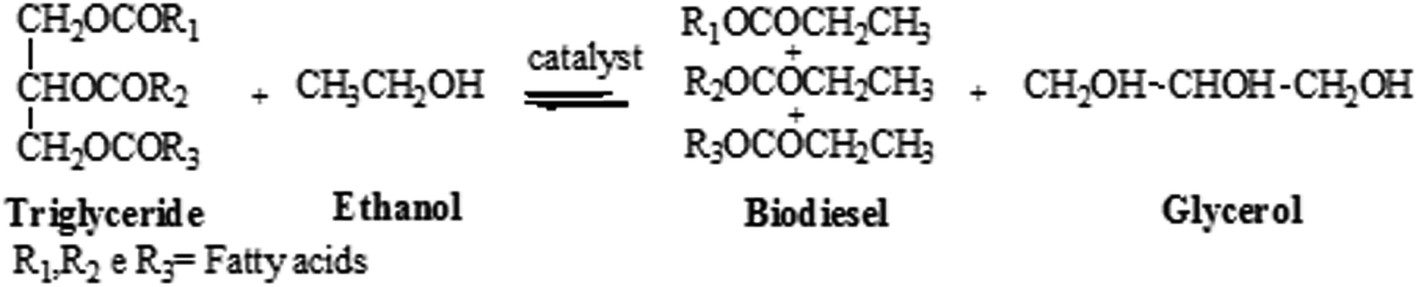






![Esterification Manufacture of Malonic Ester - [PDF Document] Esterification Manufacture of Malonic Ester - [PDF Document]](https://cdn.masterorganicchemistry.com/wp-content/uploads/2019/12/4-mechanism-for-reduction-of-aldehydes-with-sodium-borohydride.gif)

:max_bytes(150000):strip_icc()/a-bar-of-white-soap-with-soap-suds-on-it-77937370-5915b4ed3df78c7a8c566360.jpg)