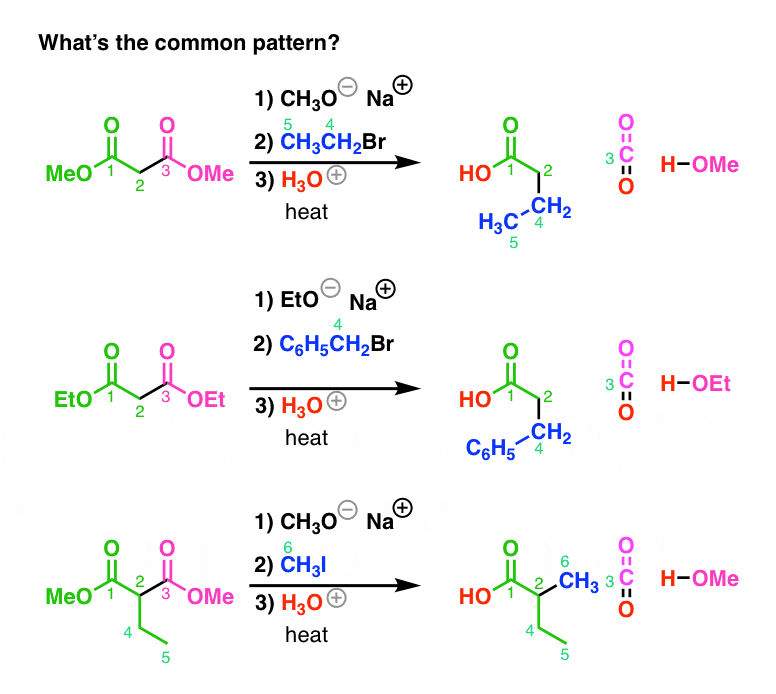
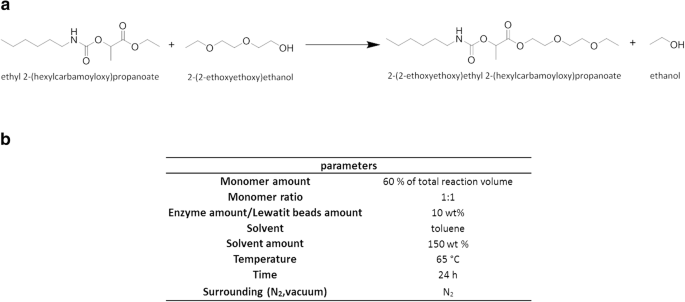
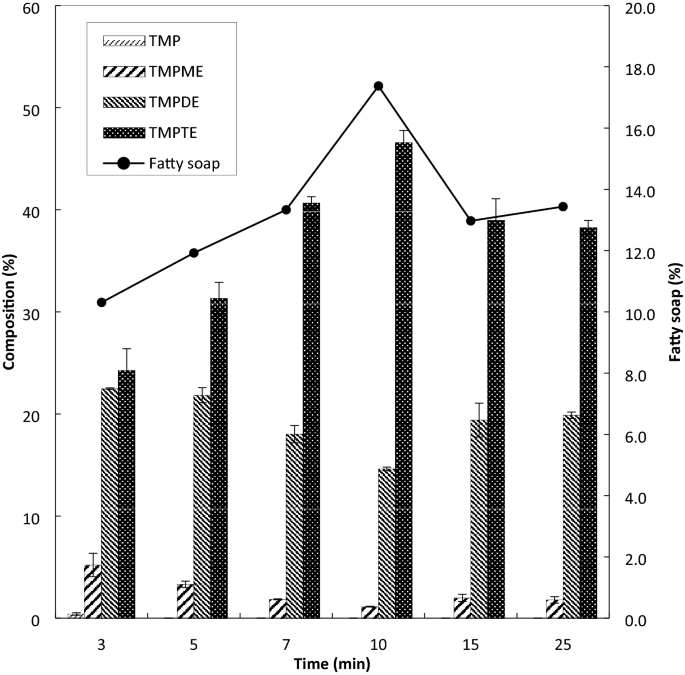
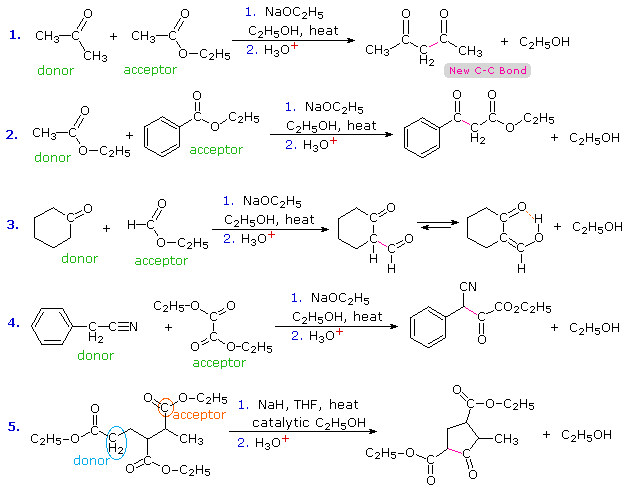
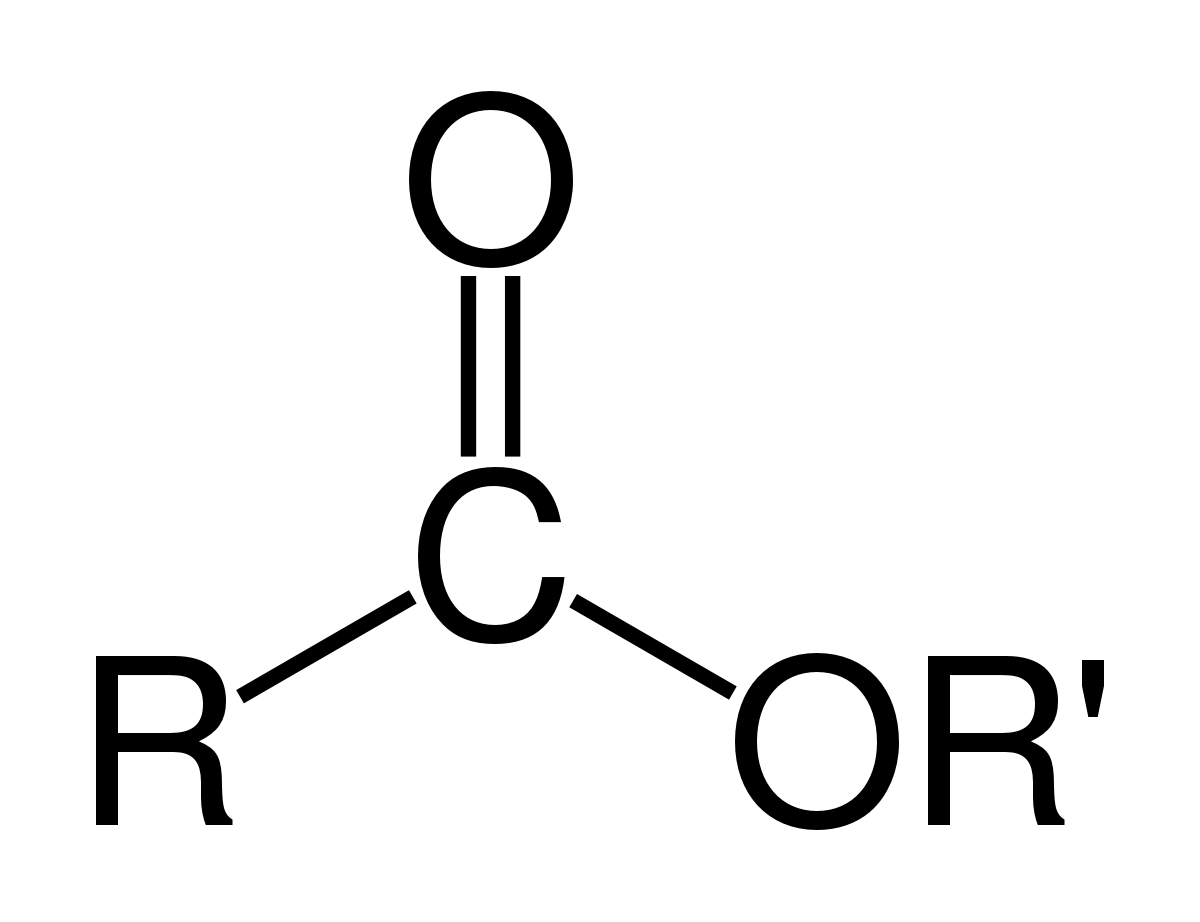
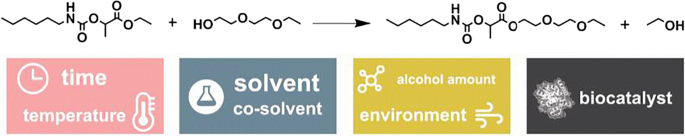
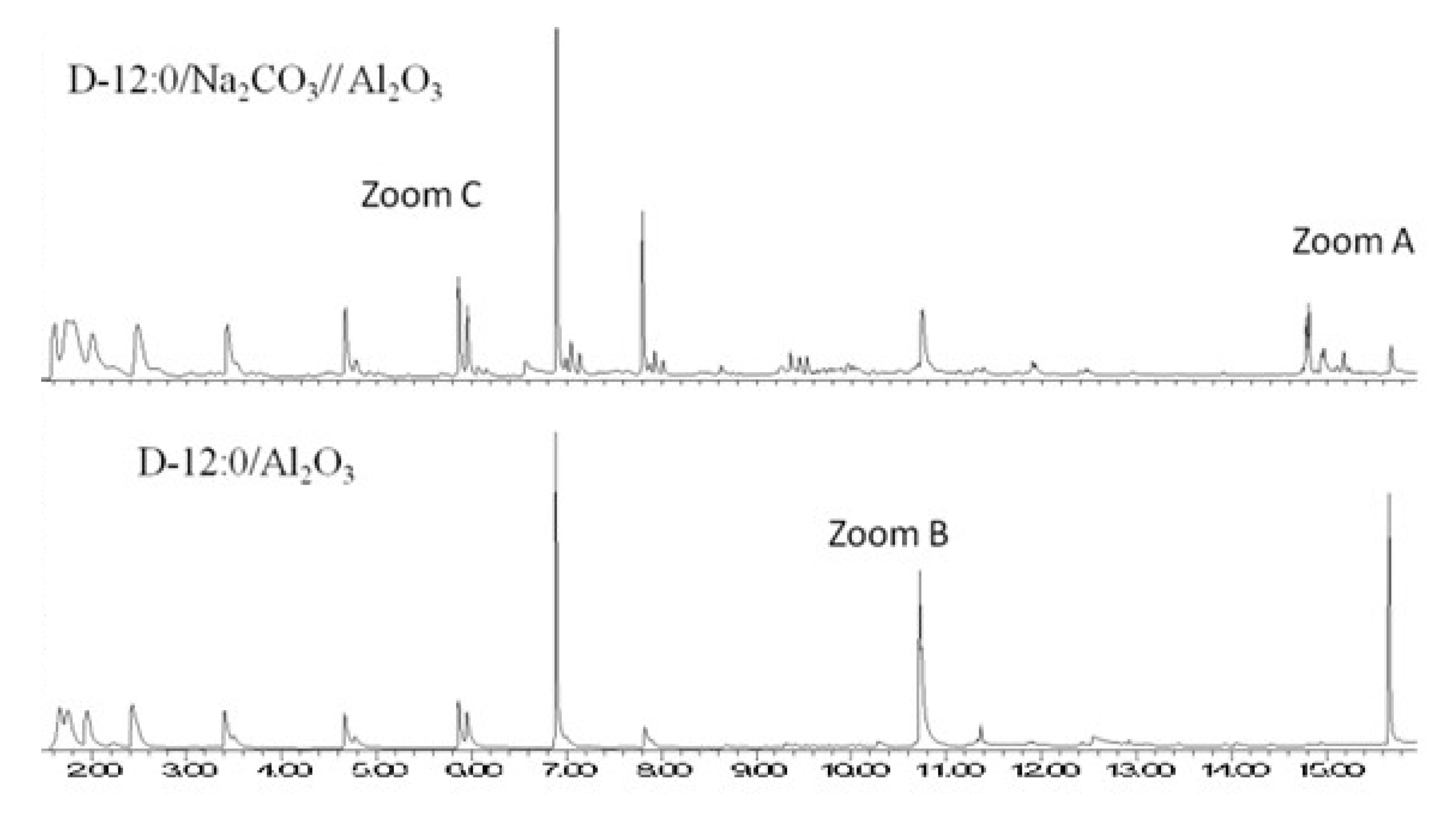
esters react with sodium carbonate
Sodium carbonate is referred to as washing soda.
It is basic in nature and reacts with acids.
What happens when sodium reacts with esters?
When esters are treated with sodium hydroxide, they are converted into carboxylate salts, which upon neutralization yield carboxylic acids.
This process is called basic hydrolysis of esters, or saponification.
The carboxylic acid is then deprotonated to give a carboxylate.
|
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters and learn how to The sodium salt of the amino acid glutamate;. |
|
ESTERS
ESTERS. An Introduction to Organic Chemistry Reactions sodium carbonate Na2CO3 10% solution magnesium sulfate (anhydrous) |
|
THE ACETOACETIC ESTER CONDENSATION. II. THE REACTION
esters react with sodium to give compounds other than the acetoacetic sodium carbonate. The carbonate extract was then acidified with sulfuric acid and. |
|
N Goalby chemrevise.org 1 Carboxylic Acids
enough an acid to react with sodium carbonate. In phenol the lone pair of carboxylic acids esters |
|
750 VOl. 53 THE ACETOACETIC ESTER CONDENSATION. 11
esters react with sodium to give compounds other than the acetoacetic sodium carbonate. The carbonate extract was then acidified with sulfuric acid and. |
|
THE CLEAVAGE OF DISUBSTITUTED MALONIC ESTERS BY
ing acetic esters and ethyl carbonate by means of alcoholic sodium ethoxide is pointed out. The diphenylmalonic ester reaction yielded principally di-. |
|
Base-induced Reactions of Certain Benzyl Esters
With the ester IX the reaction with sodium benzyl oxide is stoichiometric and yields dibenzylacetonitrile and sodium benzyl carbonate in high. |
|
Testsforfunctionalgroups - inorganiccompounds
Both the above reactions are used as tests for unsaturation. solution containing equal volume of 1% sodium carbonate solution. ... C. Ester test. |
|
The Acid-Catalyzed Alcoholysis of ?-Naphthyl Esters
samples of esters were allowed to react with solutions of hydrogen chloride in the exchange alcohol and the ß- naphthol liberated was. |
|
NZQA - NCEA Level 3 Chemistry (91391) 2018
Identifies alkene / C=C in products of elimination reaction. • Links presence of carboxylic acid group to its reaction with sodium carbonate and the structural |
|
39 Carboxylic acids and esters
They react as acids to form salts called carboxylates Ethanoic acid + sodium carbonate → e chemical equation showing the esterification reaction: Uses of |
|
I9-dI - PNAS
(enzyme mimic/biocatalyst/directscreening/immunoassay/ester hydrolysis) DAN S TAWFIK*t of reaction, substrate, or catalyst (enzymes, catalytic antibod- ies, chemical ester) and then hydrolyzed (0 1 M sodium carbonate, 3 hr) to give the |
|
Dialkyl Carbonate-Induced Transesterification for Analysis of Fatty
of sodium methoxide and the methyl esters are formed ifieation reactions for preparation of fatty acid esters for methanol with two parts of dimethyl carbonate |
|
Synthesis, Isolation, and Purification of an Ester
A solution of saturated sodium carbonate is added to a separatory funnel along with the contents of the reaction flask These contents include any unreacted acetic |
|
Lab 14: Qualitative Organic Analysis - California State University
This reaction tests for the presence of amides and esters Interpretation: Benedict's reagent contains copper(II) sulfate, sodium citrate, and sodium carbonate |
|
Reaction of Pyrroles with Ethyl 2-Nitroso- and 2-Azo - CORE
2,5-diphenyl-pyrrole The reactions of the nitroso and azo esters with pyrroles are react in organic solvents with sodium carbonate in suspension to produce |