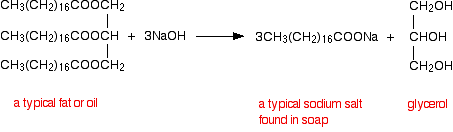
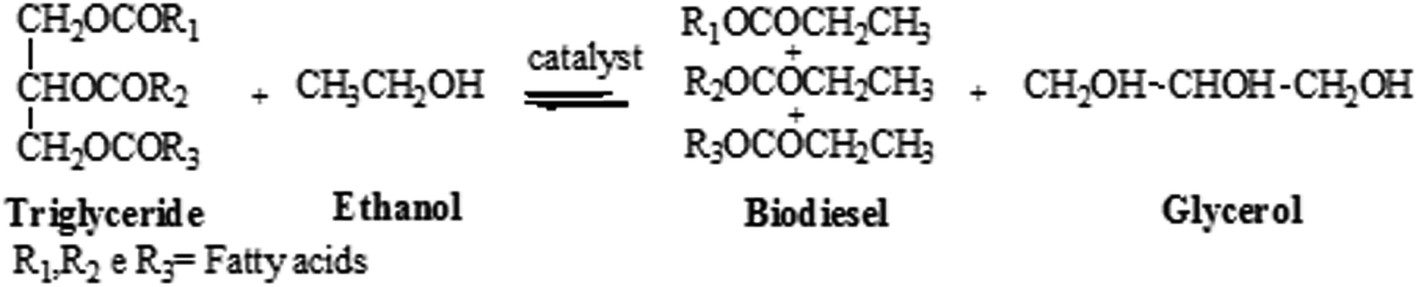esters reaction with sodium hydroxide
What will be the order of reaction if ester is hydrolysed by using NaOH?
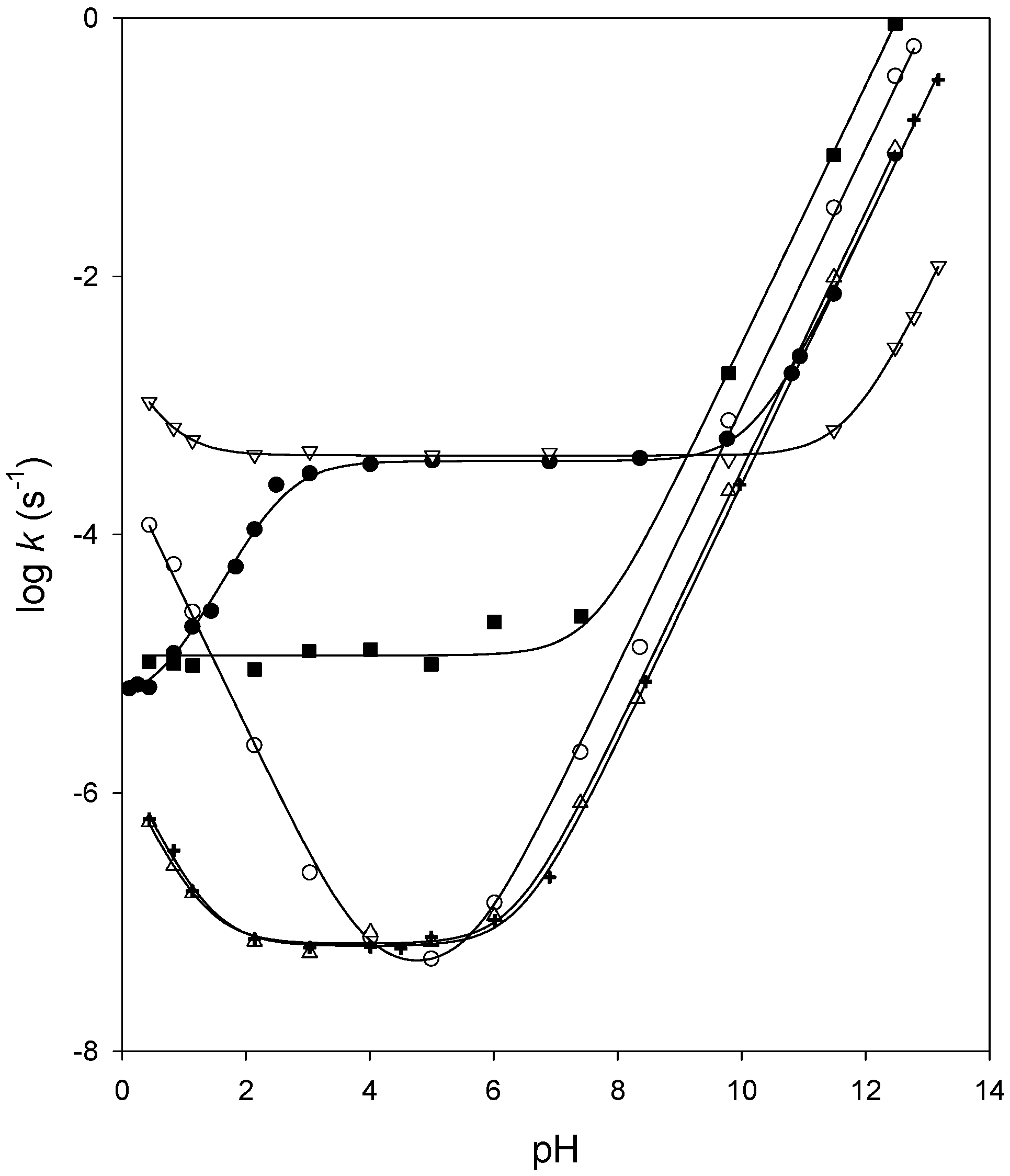
Hydrolysis of an ester in dilute acid follow 1st order, while in the presence of dilute alkali, it follows 2nd order.
|
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters and learn how to sodium hydroxide (NaOH) and potassium hydroxide. |
|
Transesterification of Vegetable Oils with Ethanol and
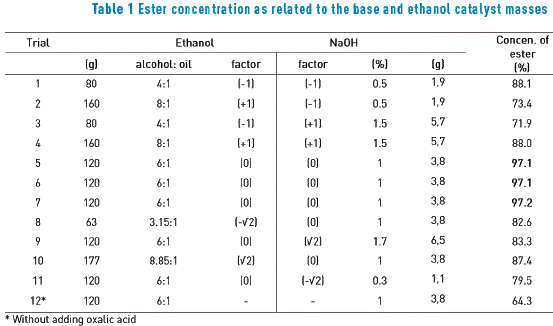
5 juin 2009 Effect of the mass ratio of NaOH to oil on ethyl ester yield. Ethanol/oil molar ratio 12:1; reaction temperature |
|
THE HYDROLYSIS OF PHOSPHATE DIESTERS WITH BARIUM
In addition to hydrolysis of the phosphate ester bonds of DNA hot effect of varying the concentratioil of sodium hydroxide in the reaction mixture. |
|
THE PRODUCTION OF METHYL ESTERS FROM VEGETABLE
presence of sodium or potassium hydroxide as catalyst (0.1-0.5 wt. % based on the weight of oil). The methyl esters produced from this reaction were fiuther |
|
The Reaction of Various Alkyl Benzoates with Sodium Alkoxides
from the reaction of ethyl benzoate with sodium With this amount of ester in the reaction a con- ... by washing with 20% sodium hydroxide solution. The. |
|
Selective reduction of esters with sodium trimethoxyborohydride
The reaction mixture was The rate of reduction of representative methyl esters with sodium ... one mole-equivalent of sodium hydroxide in. |
|
HYDROLYSIS OF METHYL SALICYLATE
This ester will be treated with aqueous base. The hydrolysis reaction that occurs will form methanol water |
|
Revisiting the mechanisms of low-temperature base-catalysed ester
Abstract: Ester interchange reactions such as the interesterification of titative saponification reaction between the sodium hydroxide formed. |
|
The Study of Chemiluminescence of Acridinium Ester in Presence of
the Chemi Luminescence (CL) reaction is acridone and that the blue light produced with out the hydrogen peroxide acridinium ester and sodium hydroxide. |
|
39 Carboxylic acids and esters
+ NaOH → CH3CO2Na + H2O Sodium ethanoate 2CH3CO2H + Na2CO3 → 2CH3CO2Na + H2O + CO2 Sodium ethanoate Questions: 1) For the reactions |
|
Saponification of Ethyl Acetate
The rate constant k for chemical reactions is given by the Arrhenius equation: k = se-48a/RT fication of an ester by sodium hydroxide CH3COOC2H5 + Na+ + |
|
613 revision guide carboxylic acids and esters - chemrevise
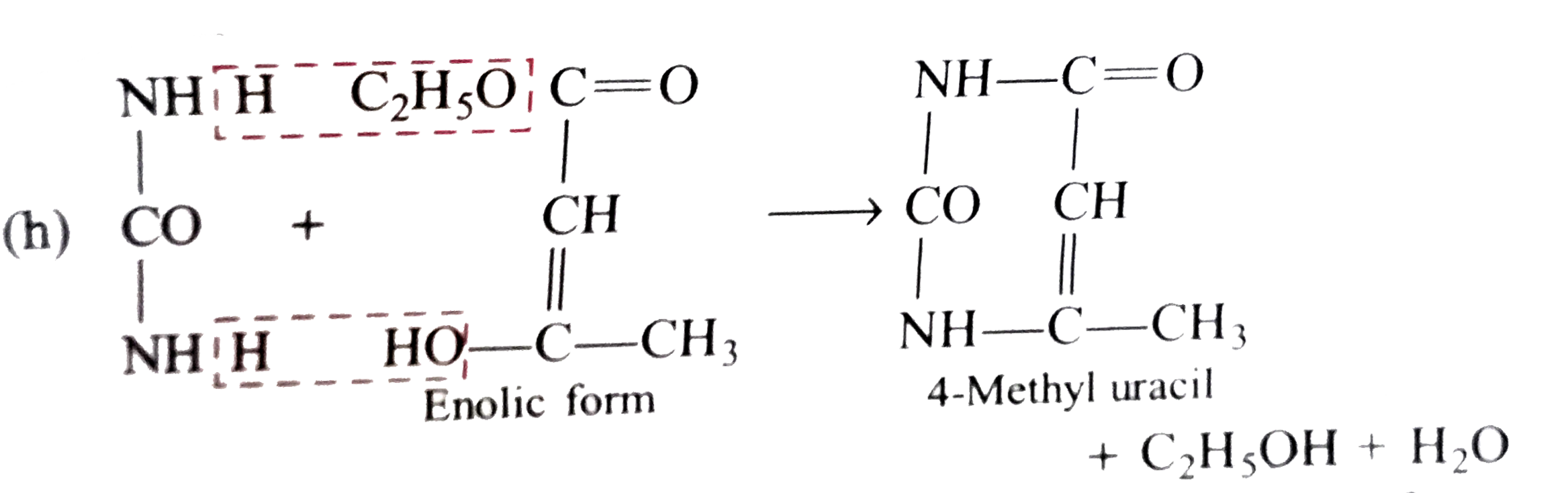
Esters can be hydrolysed and split up by either heating with acid or with sodium hydroxide The carboxylic acid salt product is the anion of the carboxylic acid The anion is resistant to attack by weak nucleophiles such as alcohols, so the reaction is not reversible |
|
Benzoic Acid from Ethyl Benzoate by Base Hydrolysis
esters It is a sweet smelling, colourless, liquid used in perfumery under the name with aqueous sodium hydroxide, the reaction going to completion, to give |
|
Metal ion complex catalysis of amino acid ester hydrolysis - CORE
the reactions written for the base hydrolysis of esters is 2 one in which the 0 0067 M ester and 0 050 M KNOg with 0 206 M NaOH solution The pH of the |
|
Synthesis of Carboxylic Acids
Since the reaction with NaOH is always downhill, all of these reactions work Conversion of the acid chloride to the ester is a “downhill” reaction energetically |