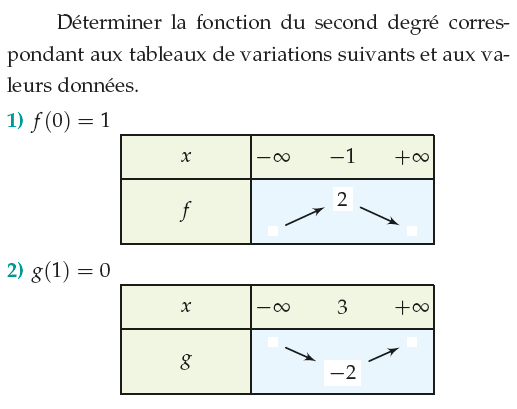
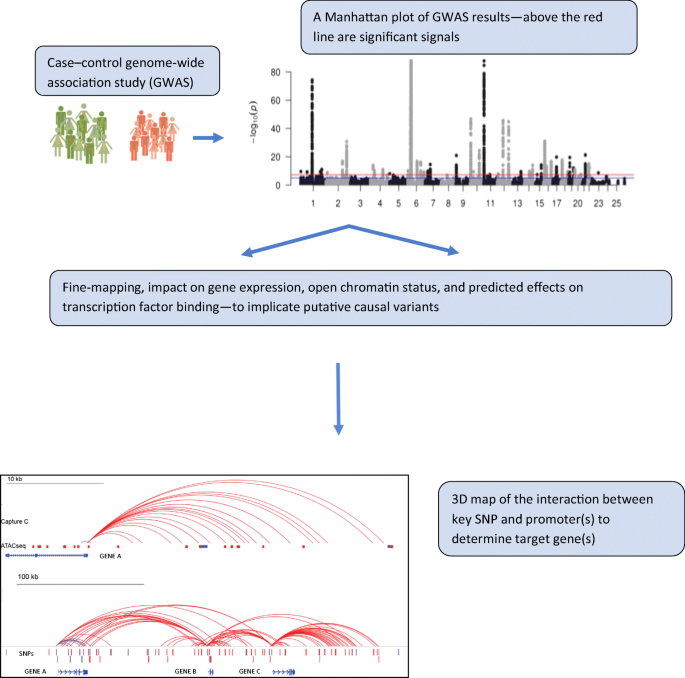
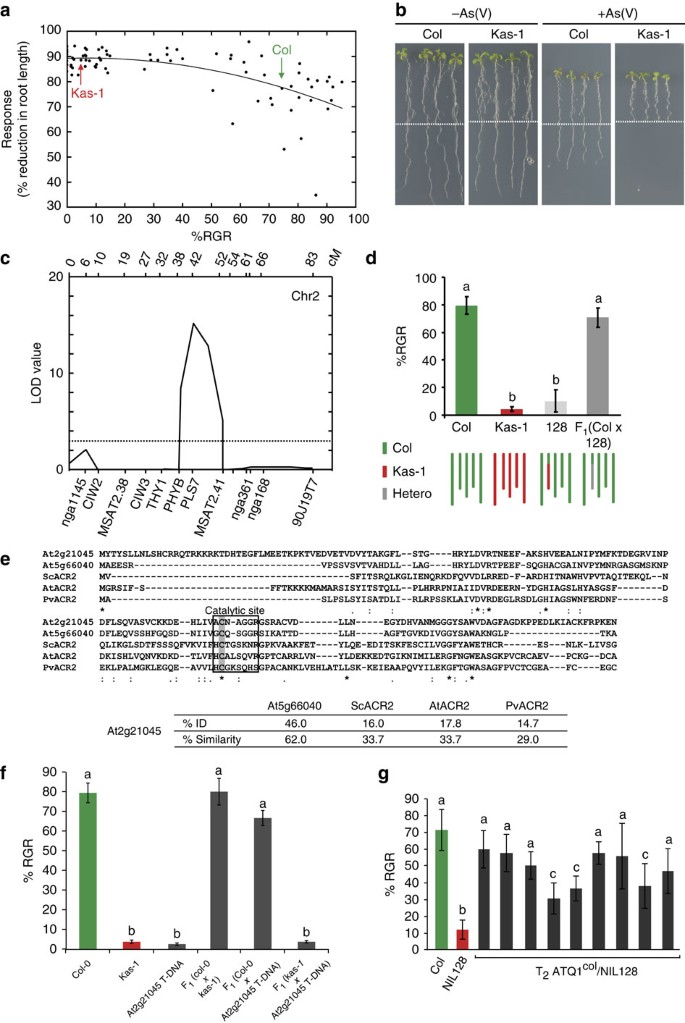
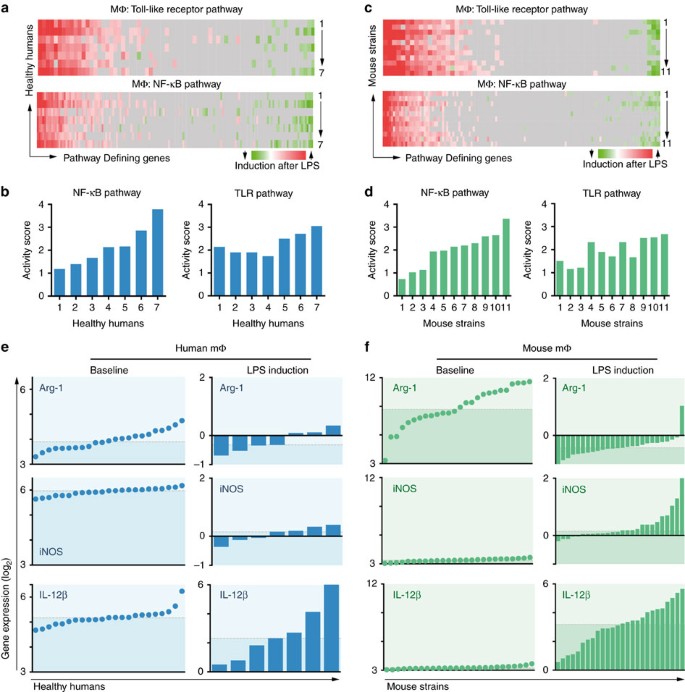
type 1a variation
|
Classification guideline
The variations regulation lays down general rules on the types and classification of variations in Articles 2 and 3 and in Annex II In addition Article 4(1)(a |
|
Marketing Authorisation Variations
The following changes concerning own label suppliers/distributors will be handled under Type 1A change code A5: • A change to the name and/or address of an |
|
Chapter 3
This Best Practice Guide has been produced by the CMDh in order to facilitate the processing of Type IA minor variations in the MR procedure |
|
European variations for medicinal products for human use
Type IA variations are minor variations which have minimal impact or no impact at all on the quality safety or efficacy of the medicinal product The |
|
Pre-notification check for type IA/IAIN Variations 1
This pre-notification checklist is aimed at facilitating submission of complete and correct Type IA and Type IAIN variation notifications by Marketing |
|
BEST PRACTICE GUIDE For Type IA Variations CMDv/BPG/004
11 avr 2014 · These are identified as IAIN variations in the above guideline 1 3 It is possible for a MAH to include a Type IA variation in the submission of |
|
Guideline on dossier requirements for Type IA and IB notifications
7 juil 2006 · A consequential variation to a Type IA notification can only be another Type IA notification while a consequential variation to a Type IB |
|
Lifecycle management: - EU and US variation requirements
Variations that can be submitted as Type IA must be implemented and then the required submission made within one year of the implementation date. For changes. |
|
European variations for medicinal products for human use
Type IB variations are minor variations which are not a Type IA variation nor a Type. II variation nor an extension. Approval from the competent authority is |
|
Pre-notification check for type IA/IAIN Variations 1
The BfArM strongly recommends that this checklist is used in advance of submission of any Type IA or Type IAIN variation; you should be able to answer to |
|
Pre-notification check for type IA-IAIN variations
30-Jun-2017 This pre-notification checklist is aimed at facilitating submission of complete and correct Type IA and. Type IAIN variation notifications ... |
|
Pre-notification check for type IA/IAIN variations: Ensuring the quality
12-May-2021 Ensuring the quality of veterinary type IA/IAIN variation submissions. Background. Type IA and IAIN (type IA immediate notification) ... |
|
Practical guidance on the application form for centralised type IA
25-Feb-2021 application form for centralised type IA and IB variations. This document is intended as guidance to facilitate the completion of the latest ... |
|
EMA practical guidance on type IA and IB variation application form
09-Nov-2018 application form for centralised type IA and IB variations for veterinary medicinal products. This document is intended as guidance to ... |
|
Guideline on variations to marketing authorisations for medicinal
However certain type IA variations must be notified to the Danish Medicines Agency immediately after implementation due to administrative circumstances. Type |
|
Variations in eCTD format Q&A document
05-Jan-2015 or extension type. For example a group consisting of a Type IA and a Type IB variation should use the submission type value “var-type1b”; ... |
|
Guidance for the applicants for the preparation of the precise scope
30-Jun-2017 form when submitting Type I and Type II variation applications ... of Competent Authority (CMDh Art. 5 Recommendation) (IA IB or II. |
| Searches related to type 1a variation |
Overview
Variations are either: 1. an administrative change such as a change of company name and/or address 2. a change to the characteristics of a product that can affect its quality, such as a change to its composition 3. a change to the safety, efficacy or pharmacovigilance of the product Changes are classed as major (type II) or minor. Minor changes are...
Marketing Authorisation Types
From 1 January 2021, the following Market Authorisation types will be possible in the United Kingdom (UK): (Guidance relating to application for a licence to market a medicine in the UK) PL – authorised for use in United Kingdom 1. as a purely national UK wide MA 2. as part of an MR/DC procedure involving Northern Ireland as CMS, but a UK wide MA P...
Major Variations
These changes are more complex and may have a significant impact on the quality, safety and/or efficacy of the product. MHRA needs to approve major variations before they are made. One example of a major change is adding a new therapeutic indication to a product or updating the current indication (the medical needs the product is used for). See the...
Common Invalidation Errors and Pre-Submission Checklist
Following analysis of submissions MHRA has developed a pre-submission checklist of submission errors. Applicants are strongly advised to use the pre-submission checklist below which will reduce the likelihood of the submitted application being invalidated or rejected. See our pre-submission checklist(PDF, 26.9KB, 2 pages The marketing authorisation...
Extensions
Although extensions are still considered a type of variation, their impact on a product is so significant that you will need to follow the application process to apply for a new MA. The form for this process has a section on extensions. Extensions can be: 1. changes to the active substance(s), including the salt/ester, isomer or biological active s...
Revised Labels, Leaflets and/or Packaging
If your variation (eg change of manufacturer) affects details of the labels, leaflets and or packaging (also known as livery) of the product, you will need to include the updated versions with your submission to be assessed. However, you don’t need to submit a variation if there hasn’t been change to the product and you only want to make style chan...
Submit grouped Changes
You can apply multiple changes to a single product using the ‘grouping’ format, as long as the changes are directly related. For example, you can make a change to the manufacturing site of the finished product, change in batch size and the manufacturing process to 1 product. The type of procedure (type IA, IB, II) depends on the extent of the chang...
Variations to Multiple Products
In some circumstances, you can use the worksharing format if you’re submitting the same type IB or type II change or group of changes to multiple MAs. As far as any product which is authorised in the UK is concerned only products which are authorised in Northern Ireland either centrally, purely nationally in Northern Ireland only, or as part of an ...
Composite Coordination Collection
If you want to apply for changes to one or more product licences (licence for each product under a marketing authorisation) that would affect the product information you can submit a composite coordination collection (CCC). Product information includes the summary of product characteristics, leaflets and labels. With a CCC you can: 1. submit only o...
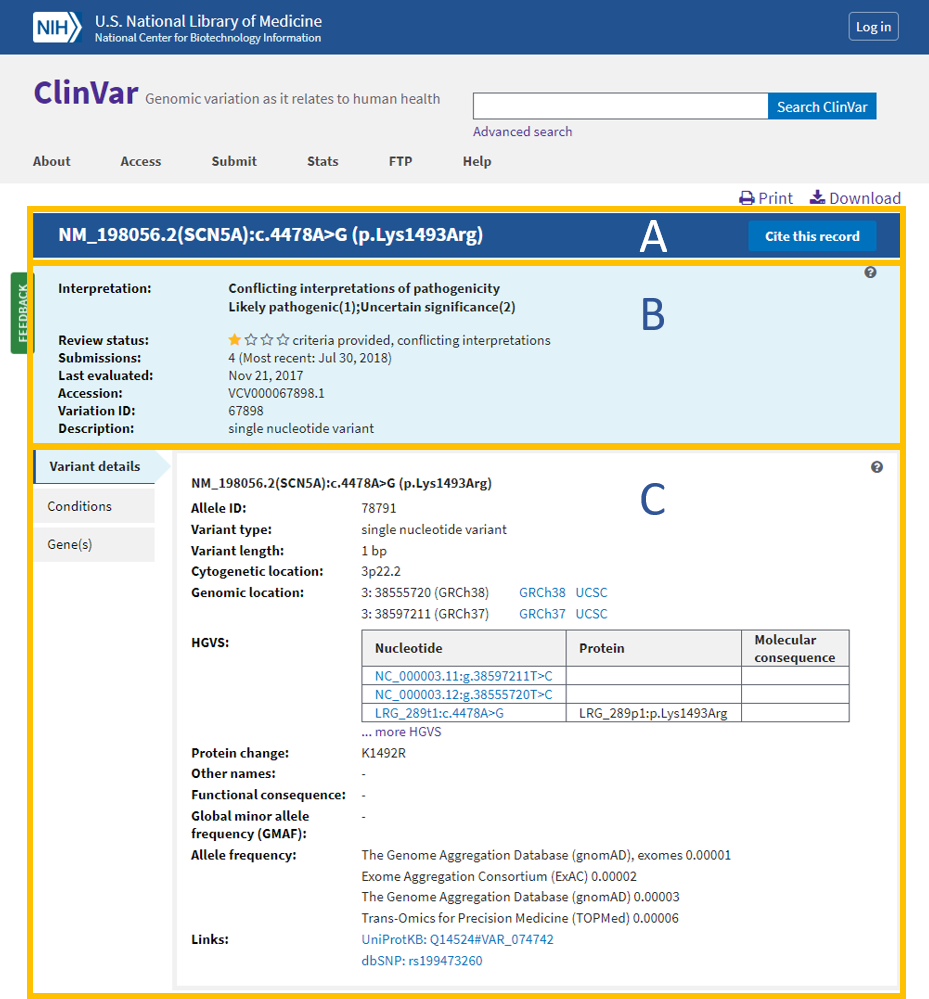
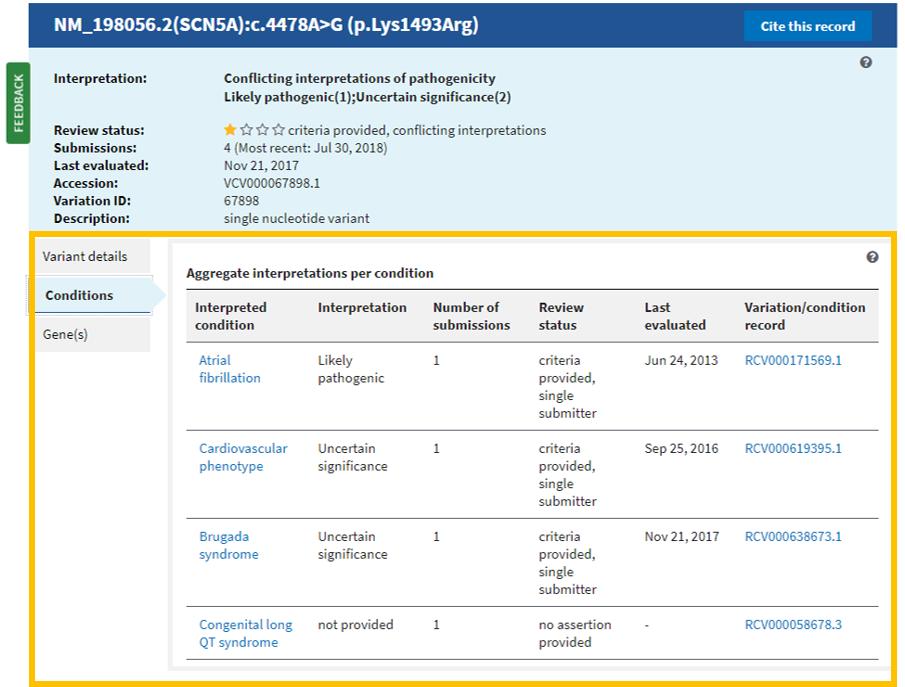
What is a Type Ia variation?
- Type IA variation A minor change to a marketing authorisation that has a minimal or no impact on the quality, safety or efficacy of the medicine and does not require prior approval before implementation by the marketing authorisation holder. For more information, see type IA variations: questions and answers.
What are the subcategories of Type-Ia variations?
- Such minor variations are classified in two subcategories, which impact on their submission: Type-IA variations requiring immediate notification ('IA IN') Type-IA variations not requiring immediate notification ('IA')
What is possible grouping of Type-Ia and IA in changes only?
- Possible grouping of type-IA and -IA IN changes only Several type IA or IA IN affecting one medicinal product: This means, for instance, that a type-IA variation, which is normally not subject to immediate notification, can be included in the submission of a type-IA INvariation;
How long does it take to implement a Type-Ia variation?
- Type-IA and -IA IN variations do not require prior approval before implementation ('do-and-tell' procedure), i.e. they can be implemented and notified to the Agency either immediately for type-IA variations requiring immediate notification ('IA IN ') or within 12 months for type-IA variations not requiring immediate notification ('IA').
|
EMA practical guidance on type IA and IB variation application form
9 nov 2018 · This document is intended as guidance to facilitate the completion of the application form for type IA and IB variations to be submitted in the |
|
Guidance for the applicants for the preparation of the precise scope
30 jui 2017 · form when submitting Type I and Type II variation applications For default Type IB variations i e 'z'- category (except Type IB variations |
|
Classification des différents types de variations - FAVA-Multi
22 jui 2018 · ▫ Type de variation : Non-sens, frameshift, splice, délétion d'exon(s) Faux sens |
|
Guidance pour les variations de type II impliquant une - AFMPS
9 jan 2012 · demandes de variation de type II impliquant une révision des sections RCP 4 6 et 5 3 ces derniers mois, une guidance plus détaillée est |
|
EU and US variation requirements
Variations that can be submitted as Type IA must be implemented and then the required submission made within one year of the implementation date For changes |
|
Variations et formules
Plus formellement, l'équation Type I est appelée variation directe et celle du Type II est une variation partielle Dans la prochaine section, tu examineras les |
|
Pre-notification check for type IA/IAIN Variations - BfArM
This pre-notification checklist is aimed at facilitating submission of complete and correct Type IA and Type IAIN variation notifications by Marketing Authorisation |
|
ZL300_00_001e_WL Guidance document Variations - Swissmedic
1 mar 2021 · These are minor variations which are neither a minor variation of type IA/IAIN, nor a major variation of type II nor a marketing extension These are |