variation classification guideline
|
CMDv RfR Mar-Apr 14 EMA-CMDv-265840-2014
14 avr 2023 · Variation classification C II 8 in classification guideline C (2013) 2804 dd 16/05/2013 The CMDv discussed the use of variation classification |
|
Classification guideline
Consequently this guideline provides details of the classification of variations into the following categories as defined in Article 2 of the variations |
|
Annex 3
The variation guidelines have been completely updated and expanded bringing them into line with the principles of the new generic quality guidelines WHO |
|
The new Classification Guideline of 2013
16 mai 2013 · variation An appropriate variation IA is lacking in the Classification Guideline For adaption to QRD-Template and fulfilment of sanctions |
|
EU and US variation requirements
Alongside the European legislation that defines variation types a guideline lays out a harmonised list of anticipated variations with classification codes 1 A |
|
ASEAN Variation Guideline for Pharmaceutical Product Revision 2
EMA Classification Guidance On Minor Variations of Type IA Minor Variations of Type IB And Major Variations of Type II ii SUPAC-IR: Immediate-Release |
|
Variation regulation and classification guideline
1 jan 2019 · Because of the Type IB default guideline needs to cover all types of changes including admin quality clinical all types of changes including |
|
European variations for medicinal products for human use
Details regarding the classification of variations into the various categories can be located in the “Commission Guideline on the details of the various |
|
Guidance-details-classification-variations-requiring-assessment
guideline Grouped variations requiring assessment will be processed according to the longest timetable applicable to any of the included variations For |
|
ASEAN Variation Guideline for Pharmaceutical Product Revision 2
This ASEAN Variation Guideline concerns the variation applications submitted by the EMA Classification Guidance On Minor Variations of Type IA ... |
|
C 223 Official Journal
2 Aug 2013 Commission with the task of drawing up guidelines on the ... the classification of variations into the following categories as. |
|
Classification_guideline_adopted.pdf
CLASSIFICATION GUIDANCE ON MINOR VARIATIONS OF TYPE IA 3 of the variations regulation and that the Annex to this Guideline does not. |
|
Malaysian Variation Guideline for Biologics (MVGB)
The Malaysian Variation Guidelines for Pharmaceutical Products (MVG) will still Justification for the classification of any manufacturing process and/or ... |
|
EMA-CMDv VRA guidance
17 Jun 2022 Guidance on the details of the classification of variations ... may decide to use other timetable than those detailed in this guideline. |
|
Guidance for the applicants for the preparation of the precise scope
30 Jun 2017 variations in addition to the EMA/CMDh explanatory notes on variation application ... CATEGORY OF THE VARIATION CLASSIFICATION GUIDELINE ... |
|
Guidelines on the details of the various categories of variations on
2 Aug 2013 the classification of variations into the following categories as defined in Article 2 of the Variations Regulation: minor vari. |
|
The Variations Regulation (EC) No 1234/2008: The legal framework
14 Jun 2011 Now: - The “Commission guideline on the details of the various categories of variations” ('the Classification Guideline') provides details ... |
|
Procedural guidance for variation for variant update to coronavirus
8 Jun 2022 variation covid-19 vaccines |
|
Guideline on stability testing for applications for variations to a
21 Mar 2014 A Type IB variation is the default category under the Commission Regulation (EC) No 1234/2008. The associated classification guideline ... |
|
Classification guideline
The variations regulation lays down general rules on the types and classification of variations in Articles 2 and 3 and in Annex II In addition, Article 4(1)(a) charges |
|
Procedural guideline - European Commission
categories of variations” (hereinafter referred to as 'the Commission Classification Guideline') set-out a list of changes to be considered as minor variations of |
|
EMA practical guidance on type IA and IB variation application form
9 nov 2018 · application form for centralised type IA and IB variations for veterinary medicinal products This document is Classification guideline extract |
|
Variation regulation and classification guideline
1 jan 2018 · Classification Guideline (key document) the MS requests the submission of a type IB variation in case of submission after 12 months due to |
|
WHY IS NOW THE RIGHT TIME TO MODERNISE THE EU
To consider the Variations Classification Guideline to be the EMA/HMA (CMDh) guideline, instead of the EC guideline in view of more regular/frequent updates |
|
Pre-notification check for type IA/IAIN Variations - BfArM
Type IB variation notifications by Marketing Authorisation Holders (MAHs) Guidance for Copy of the relevant page(s) from the Classification Guideline is/ are |
|
Q&A - List for the submission of variations according - Acta Sanitaria
The implementation of product information updates after a PSUR-WS can be submitted as a type IB variation under category C I 3 z of the classification guideline |
|
EU and US variation requirements
Alongside the European legislation that defines variation types, a guideline lays out a harmonised list of anticipated variations with classification codes 1 |
|
Best practice of worldwide product variations regarding - DGRA
Guidance on the classification of variations is given in the respective guideline by the Food and Drug Administration (FDA) [9] Major changes or “Prior Approval |












![PDF] ACGS Best Practice Guidelines for Variant Classification 2019 PDF] ACGS Best Practice Guidelines for Variant Classification 2019](https://i1.rgstatic.net/publication/273148351_Standards_and_Guidelines_for_the_Interpretation_of_Sequence_Variants_A_Joint_Consensus_Recommendation_of_the_American_College_of_Medical_Genetics_and_Genomics_and_the_Association_for_Molecular_Patholo/links/5554e2e608ae980ca60ad446/largepreview.png)



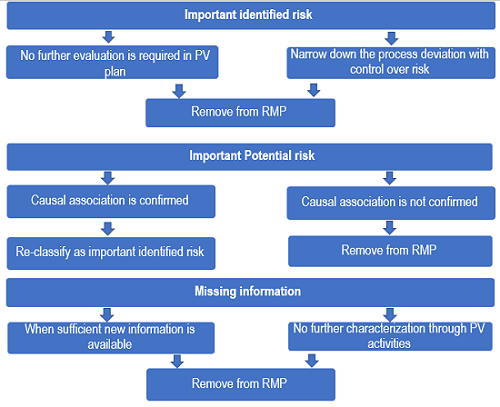


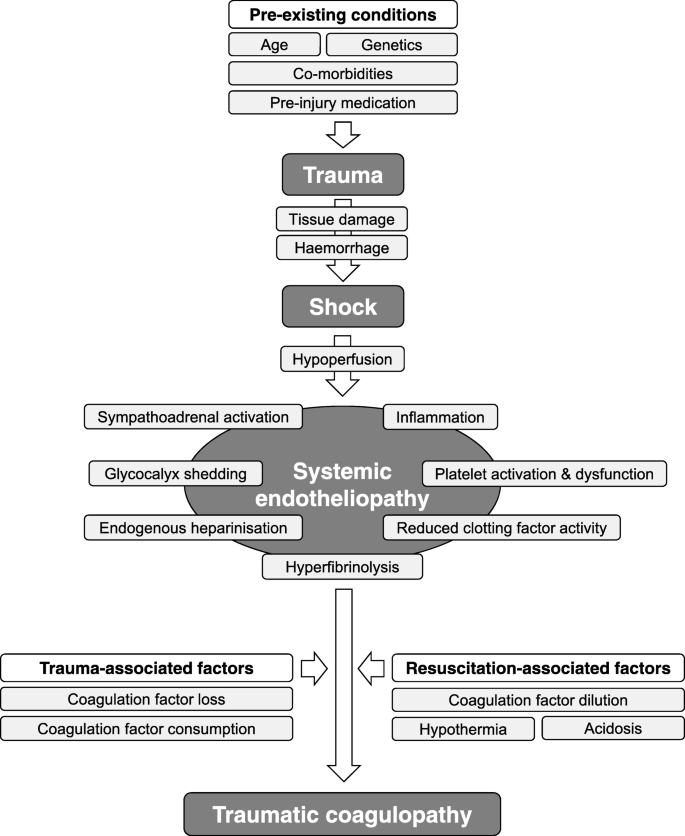

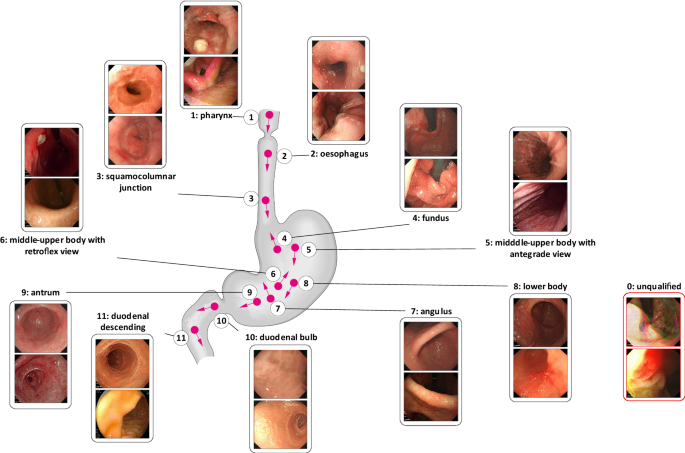


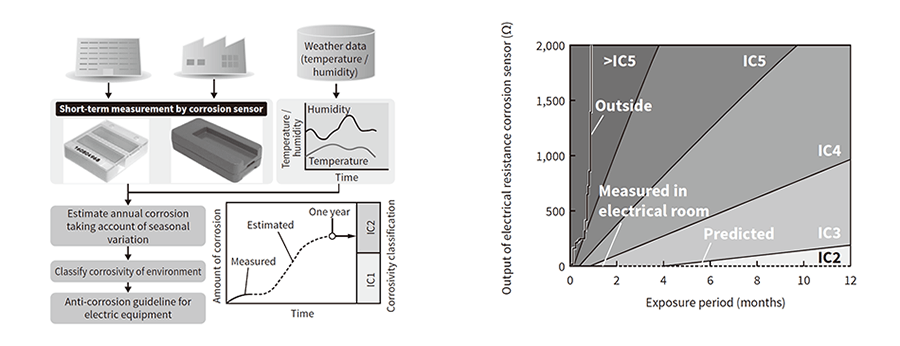

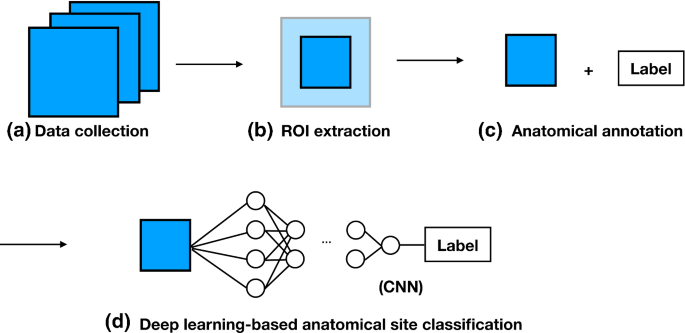







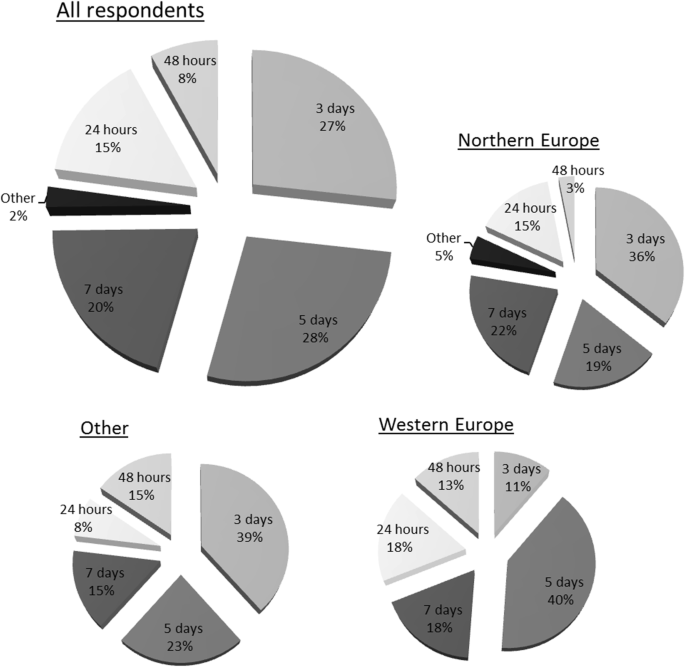


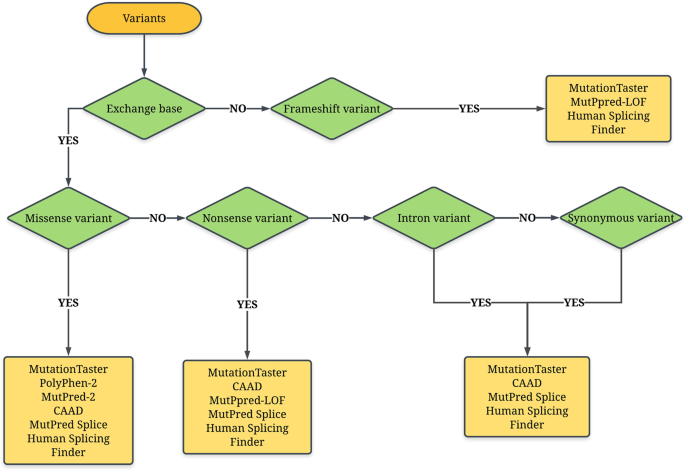

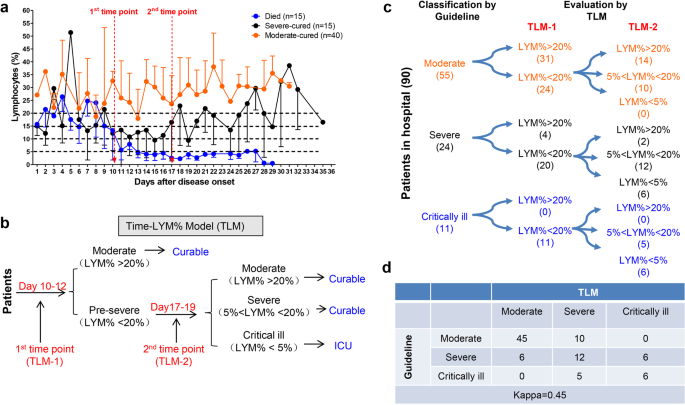










![PDF] ACGS Best Practice Guidelines for Variant Classification 2019 PDF] ACGS Best Practice Guidelines for Variant Classification 2019](https://vertassets.blob.core.windows.net/image/dd477265/dd477265-8b54-43b8-acdb-7c19ab9bb689/eu_rmp_fig1.png)