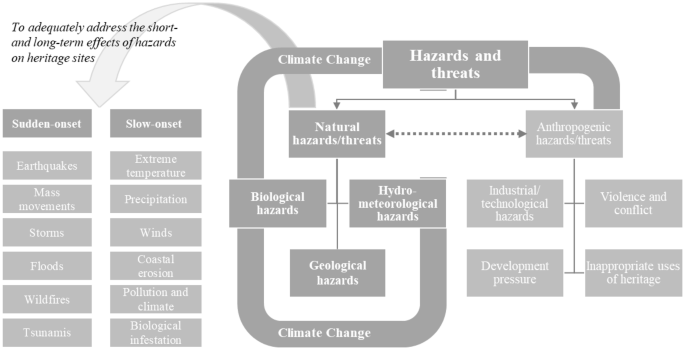
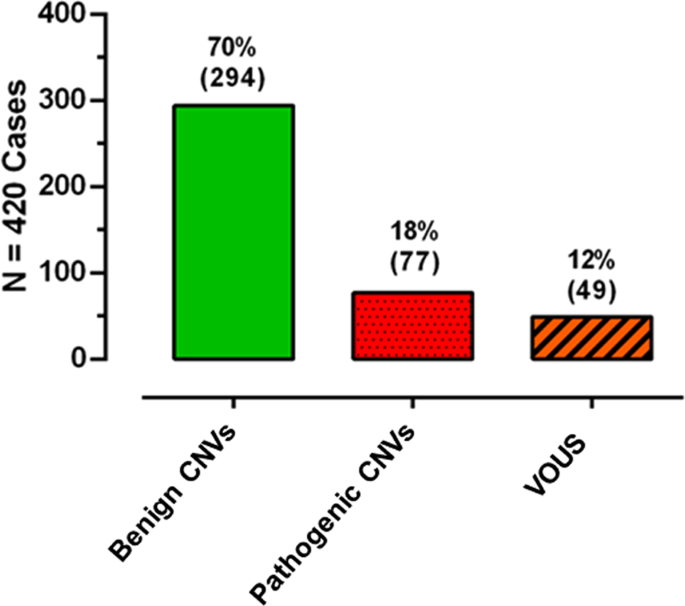
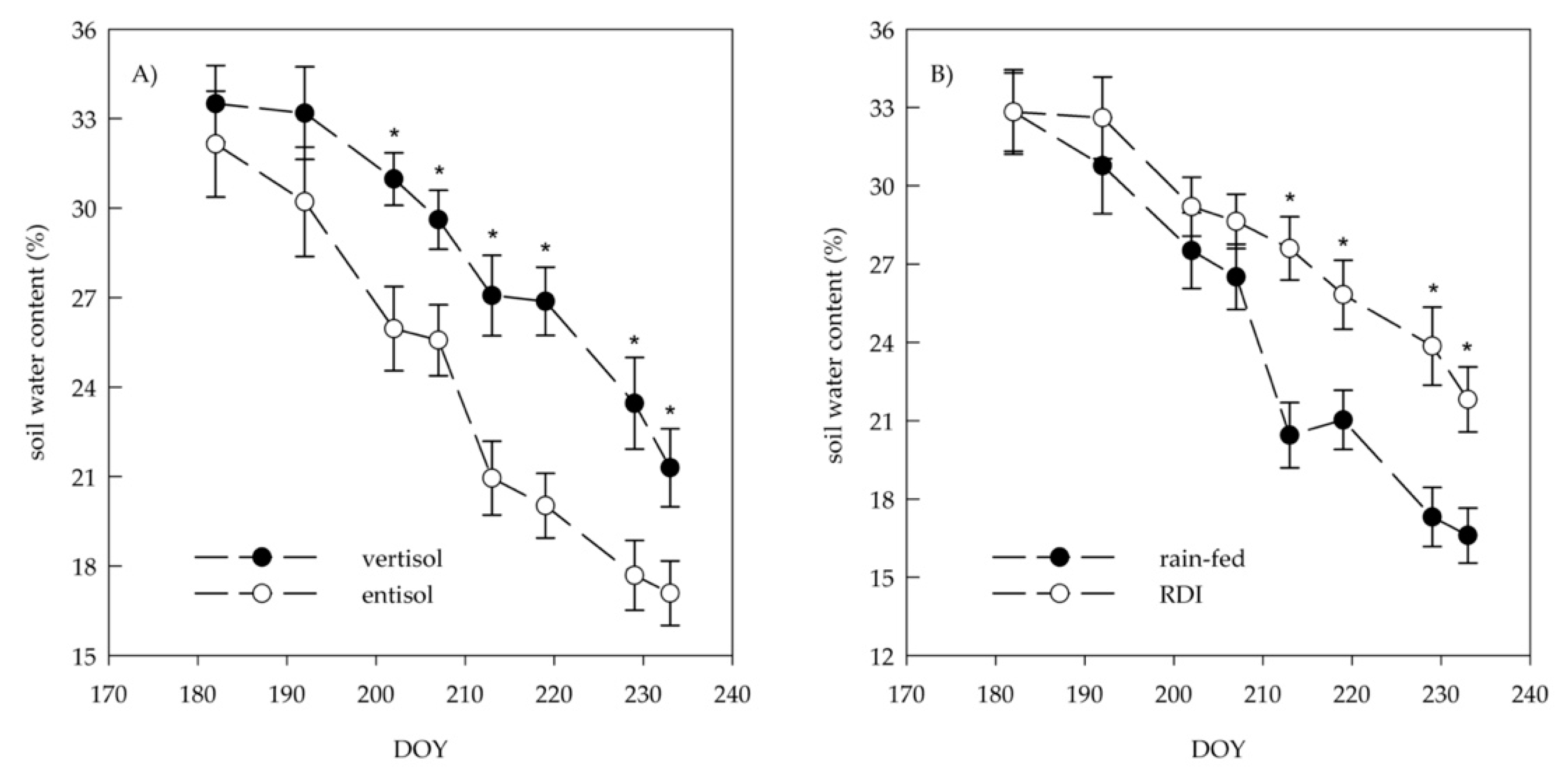
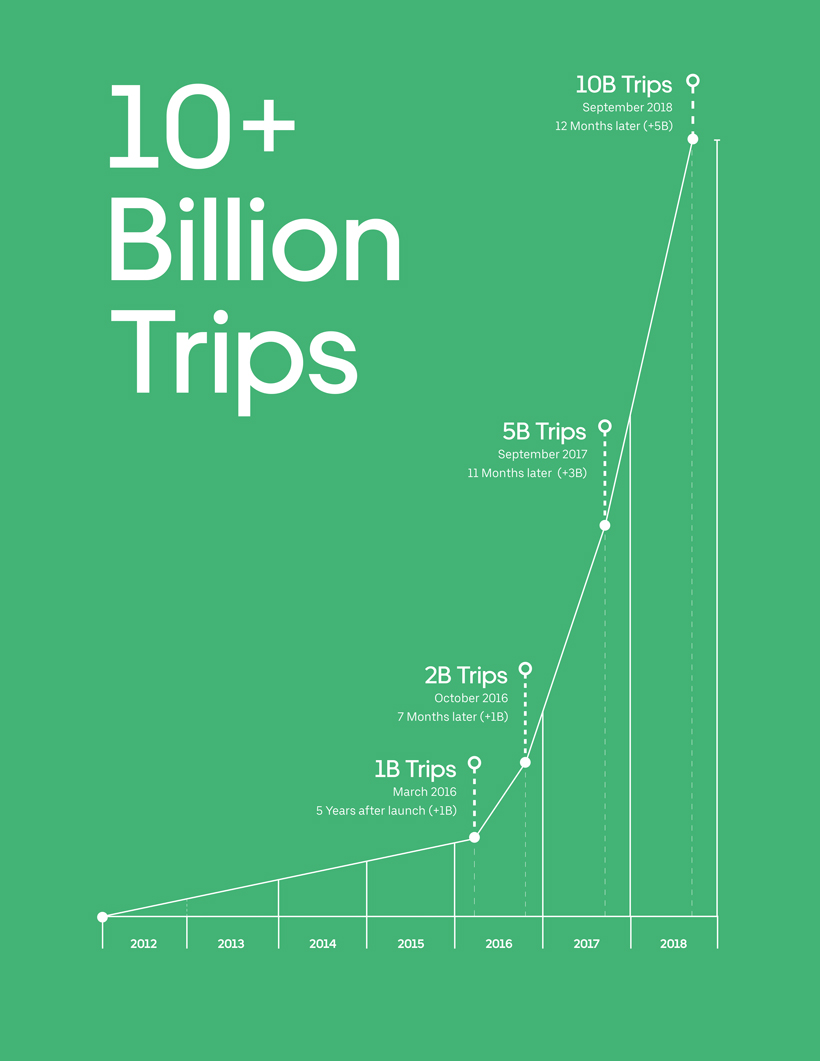
variation classification guideline 2016
|
The right time to modernise the eu variations
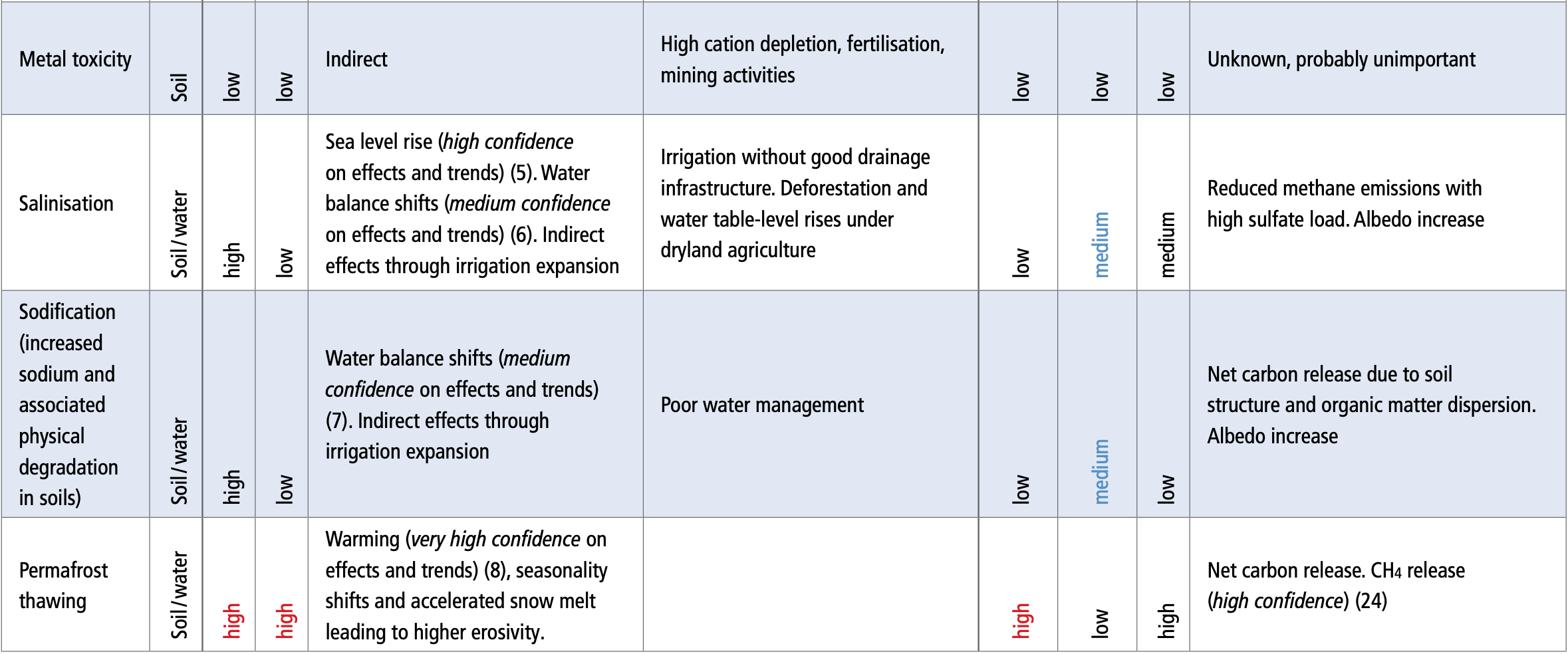
To consider the Variations Classification Guideline to be the EMA/HMA (CMDh) guideline instead of the EC guideline in view of more regular/frequent updates ( |
|
CMDv RfR Mar-Apr 14 EMA-CMDv-265840-2014
14 avr 2023 · The variations' guideline stipulates that the change in frequency and/or date of submission of the PSUR has been agreed by the competent |
|
EFPIA/EBE/Vaccines Europe Reflection Paper on a Revision of the
Variations Regulation) 2 Variation Classification Guideline (Guidelines 2013/C 223/01: Commission Guidelines on the details of the various categories of |
|
EU and US variation requirements
defines variation types a guideline lays out a harmonised list of anticipated variations with classification codes 1 A defined list of variations for European |
|
Variation regulation and classification guideline
1 jan 2019 · As of 1 February 2016 MA holders are no longer required to submit Type IA variations in relation to administrative changes to the QP |
|
ASEAN Variation Guideline for Pharmaceutical Products (R1)
EMA Classification Guidance On Minor Variations of Type IA Minor Variations of Type IB And Major Variations of Type II ii SUPAC-IR: Immediate-Release |
|
A GUIDELINE ON CHANGING THE CLASSIFICATION FOR THE
This guideline is for use by marketing authorisation holders applying to change the classification change requires the submission of a Type II variation |
What are the classification of variations in the EU?
A variation is a change in the dossier of an authorised product.
There are four different types of variations: Type IA, Type IB, Type II and Line extension.
The definitions of these variations are available in: The Regulations of the European Commission: Regulation (EC) 1234/2008 en Regulation (EU) 712/2012.What is an EMA Type 2 variation?
Commission Regulation (EC) No 1234/2008 (the Variations Regulation) defines a major variation of type II as a variation that is not an extension of the marketing authorisation (line extension) and that may have a significant impact on the quality, safety or efficacy of a medicinal product.
Article 5 of the Variation Regulation provides the basis for a marketing authorisation holder (MAH) to request EMA (in case of centralised marketing authorisations), the Reference Member State (RMS) (in case of Mutual Recognition or Decentralised marketing authorisations), or a national competent authority of a Member
What is the classification of variation?
Variations are classified either as continuous, or quantitative (smoothly grading between two extremes, with the majority of individuals at the centre, as height varies in human populations); or as discontinuous, or qualitative (composed of well-defined classes, as blood groups vary in humans).
|
Q&A on PSUSA Guidance Document for Assessors - update Oct 2017
31 oct. 2017 EMA/518909/2016 ... procedure in line with the Variations Classification guideline of Commission Regulation 1234/2008. |
|
A GUIDELINE ON CHANGING THE CLASSIFICATION FOR THE
This guideline is for use by marketing authorisation holders applying to change the classification for supply of a medicinal product. The guideline applies |
|
Guidelines on the application of the definition of default under Article
28/09/2016 where the default definition will change significantly the ... the basis for the assignment of exposures to the class of exposures in default ... |
|
European Medicines Agency post-authorisation procedural advice
20 juin 2022 Apr 2016 . ... NEW Dec 2016 . ... The Classification Guideline specifies which Type IA variations must be notified (submitted). |
|
Guideline on process validation for finished products - information
21 nov. 2016 EMA/CHMP/CVMP/QWP/749073/2016. Page 2/15. This guideline replaces the note for guidance on process validation (CPMP/QWP/848/96. |
|
C 223 Official Journal
2 août 2013 In addition the Annex to these guidelines provides details of the classification of variations into the following categories as. |
|
Prise en charge de lhypertension artérielle de ladulte
Haute Autorité de santé – 2016 Définition et classification de l'HTA . ... Clinical practice guidelines for the management of hypertension. |
|
Practical questions and answers to support the implementation of
01 June 2016 implementation of the variations guidelines1 in the ... New classification category C.1.11: introduction of or change(s) to |
|
Guideline on good pharmacovigilance practices (GVP) - Product- or
4 août 2016 Medicinal Products after a Change in the Manufacturing Process ... available product/class-specific guidance on immunogenicity evaluation ... |
|
ACGS Best Practice Guidelines for Variant Classification in Rare
4 févr. 2020 On 4th November 2016 a group of NHS clinical scientists and clinical geneticists met to discuss the implementation of the ACMG guidelines within ... |
What are the EU type of variations?
. There are four different types of variations: Type IA, Type IB, Type II and Line extension.
. The definitions of these variations are available in: The Regulations of the European Commission: Regulation (EC) 1234/2008 en Regulation (EU) 712/2012.
What is a Type 2 variation EU?
. Type II variations require a formal approval.
What are the types of variation in regulatory affairs?
. Type IA variations are minor changes with only minimal impact, or no impact at all, on the quality, safety and/or efficacy of a medicinal product.
What is Regulation EC No 1234 2008?
|
Practical questions and answers to support the implementation of
01 June 2016 EMA/427505/2013 Rev 132 implementation of the variations guidelines1 in the New classification category C 1 11: introduction of, or |
|
First webinar on regulatory and procedural aspects of type I variations
Q4 2016 Development of guidance on classification Q1 2017 • Analysis of VSI • Interaction •Variation Guidelines contains examples of changes which are |
|
Variation regulation and classification guideline
1 jan 2018 · As of 1 February 2016 MA holders are no longer required to submit Type IA variations in relation to administrative changes to the QP responsible |
|
WHY IS NOW THE RIGHT TIME TO MODERNISE THE EU
Variations Classification Guideline shall be considered under the mandate of the new 2007 2008 2009 2010 2011 2012 2013 2014 2015 2016 2017 2018 |
|
ASEAN Variation Guidelines For Pharmaceutical Products
ASEAN Variation Guideline for Pharmaceutical Products, 2012 – Final Adopted Document EMA Classification Guidance On Minor Variations of Type IA, Minor |
|
The new Classification Guideline of 2013 - DGRA
16 mai 2013 · In addition clear guidance is given in Annex II on variations that require a type II variation (such as a new indication or modification of existing |
|
Q&A - Pharmacovigilance Legislation - BfArM
(January 2016) variation guideline have been met • a type II A new RMP should be submitted as type II variation under classification category C I 11 b |
|
Guideline on the Regulation of Therapeutic Products in - Medsafe
N1 applications – new pack size/classification Guideline for the Regulation of Therapeutic Products in New Zealand Parts 1, 4, 5, 6, · and 10 1 3 Number Summary of Changes March 2016 1 0 Published version October 2019 1 1 |
|
Malaysian Variation Guideline for Biologics (MVGB) - NPRA
The Malaysian Variation Guidelines for Pharmaceutical Products (MVG) will still serve Note: Fees Implementation will commence 1st July 2016 in accordance to Justification for the classification of any manufacturing process and/or control |