variation classification guideline word
|
Guidelines-for-Variation-of-an-immunological-veterinary-productpdf
This Guideline is intended to provide guidance to applicants on the conditions to be fulfilled and the type of documentation to be submitted before a Variation |
|
Annex 3
The variation guidelines have been completely updated and expanded bringing them into line with the principles of the new generic quality guidelines WHO |
|
The new Classification Guideline of 2013
16 mai 2013 · aender-aenderungPar29- pdf __blob=publicationFile&v=3 (12 02 2014 variation is missing in the updated Classification Guideline The |
|
Variation regulation and classification guideline
1 jan 2019 · Classification Guideline (key document) – In other words an eCTD is the submission of (mostly) PDF leaf documents stored in the eCTD |
|
C 223 Official Journal
2 sie 2013 Guidelines on the details of the various categories of variations on the operation of the procedures laid down in Chapters II |
|
European Medicines Agency post-authorisation procedural advice
20 cze 2022 The Classification Guideline specifies which Type IA variations must be ... and EN tracked Word together with the variation application. |
|
EMA-CMDv VRA guidance
17 cze 2022 Guidance on the details of the classification of variations ... may decide to use other timetable than those detailed in this guideline. |
|
Guidance on the details of the classification of variations requiring
9 wrz 2021 Classification of additional new variations not already listed . ... decide to use other timetable than those detailed in this guideline. |
|
Guidance for the applicants for the preparation of the precise scope
30 cze 2017 EMA/220707/2017. Page 3/50. ANNEX. EXAMPLES OF 'PRECISE SCOPE' SECTION WORDINGS FOR EACH. CATEGORY OF THE VARIATION CLASSIFICATION GUIDELINE ... |
|
Overview of comments received on Guideline on stability testing for
19 sie 2014 example comparative 3 months/1 batch of accelerated and long-term data ... The risk-based approach of the Variations change classification ... |
|
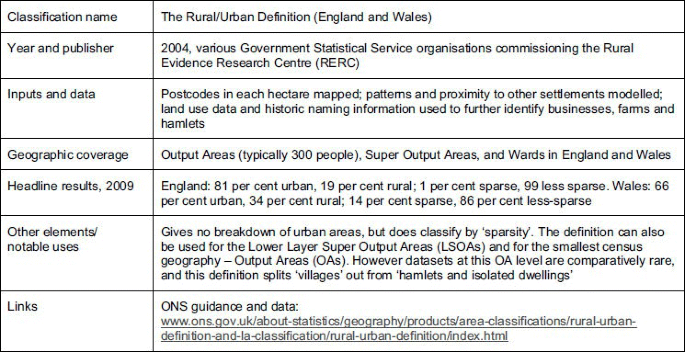
A GUIDELINE ON CHANGING THE CLASSIFICATION FOR THE
Medicinal products not subject to a medical prescription should be approved primarily for short term treatment e.g. when the possibility of ”masking” could |
|
Final feedback from European Medicine Agency (EMA) to the EU
8 wrz 2021 Regulatory procedures to register replacements in excipients: From the regulatory point of view the EC variation classification guideline ... |
|
Procedural guidance for variation for variant update to coronavirus
8 cze 2022 This guideline lays down the procedural aspects related to the submission of a variation to change the composition of a marketing authorisation ... |
|
Variation regulation and classification guideline
1 sty 2018 Type I and type II variations submitted: ... Classification Guideline (key document) ... In other words an eCTD is the submission of. |
|
Classification guideline
It should be noted that the general documentation accompanying every application for variations to the term of a marketing authorisation is laid down on Annex IV |
|
04 - Vol 2B Variation AF - Revision 2017-05-19 TC - European
Number and title of variation, as per the classification guideline or highlight the changed words presented in the table above or provide as a separate Annex |
|
Post-authorisation - European Medical Writers Association
6 déc 2018 · The Classification Guideline specifies which Type IA variations must be notified (Annex IIIB) as a full set of annexes per EU language (word |
|
Variation regulation and classification guideline
1 jan 2018 · Classification Guideline (key document) Classification Guideline – Structure specifications In other words, an eCTD is the submission of |
|
Q&A - List for the submission of variations according - Acta Sanitaria
Which documents have to be submitted for a variation Type IA, IB or II or a grouped application requirement 1 of category A8 of the classification guideline? identified, preferably using track-changes function in an editable ( word) format |
|
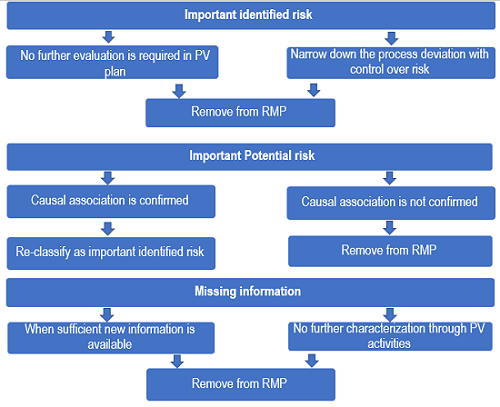
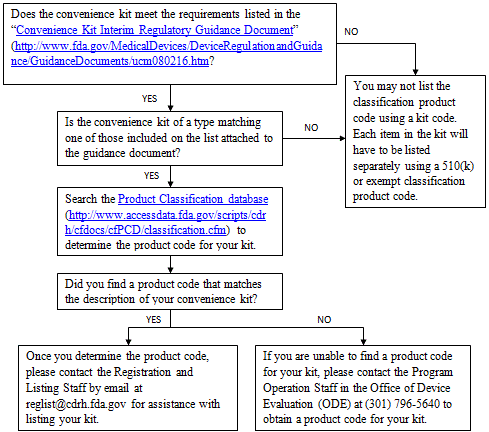
EU and US variation requirements
Alongside the European legislation that defines variation types, a guideline lays out a harmonised list of anticipated variations with classification codes 1 |




![Examples of Groupings for Variations - [PDF Document] Examples of Groupings for Variations - [PDF Document]](https://www.ncbi.nlm.nih.gov/core/assets/clinvar/images/revstat_fig_1.png)


![Examples of Groupings for Variations - [PDF Document] Examples of Groupings for Variations - [PDF Document]](https://www.ncbi.nlm.nih.gov/core/assets/clinvar/images/revstat_fig_2.png)















































![The Emotion Wheel: What It Is and How to Use It [+PDF] The Emotion Wheel: What It Is and How to Use It [+PDF]](https://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41598-019-38487-w/MediaObjects/41598_2019_38487_Fig3_HTML.png)