fischer esterification method
What is the Fischer esterification mechanism?
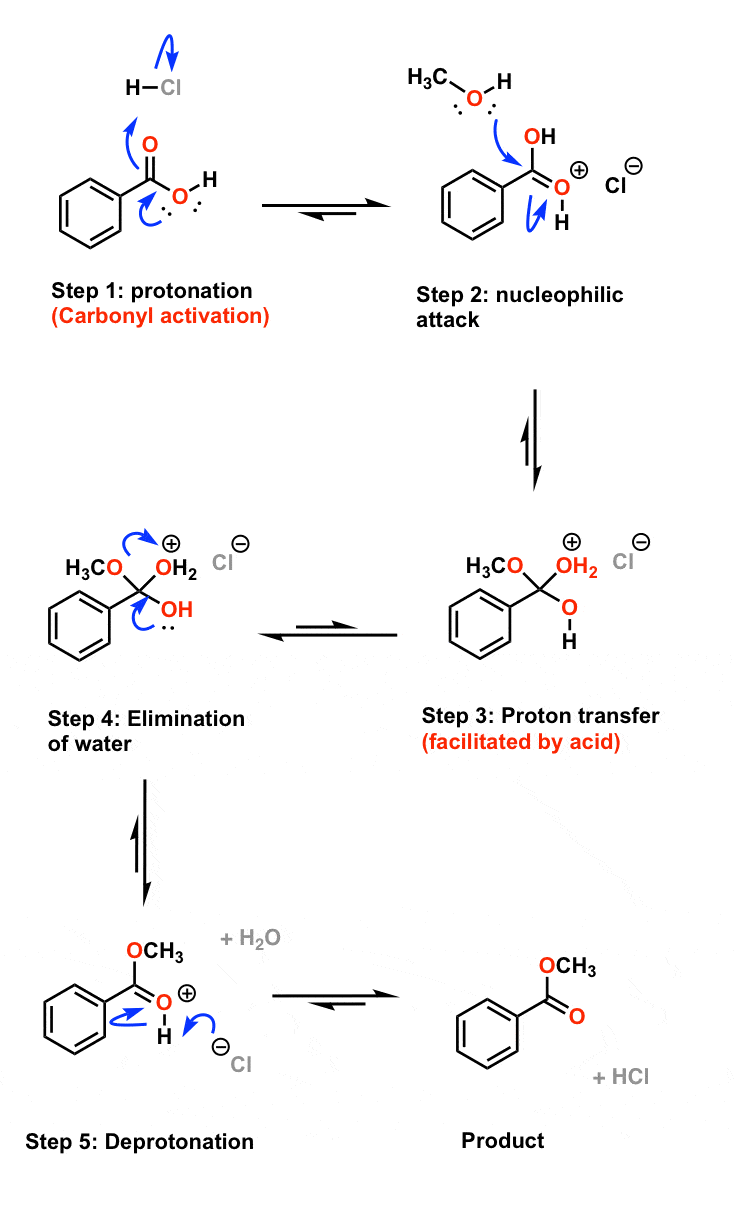
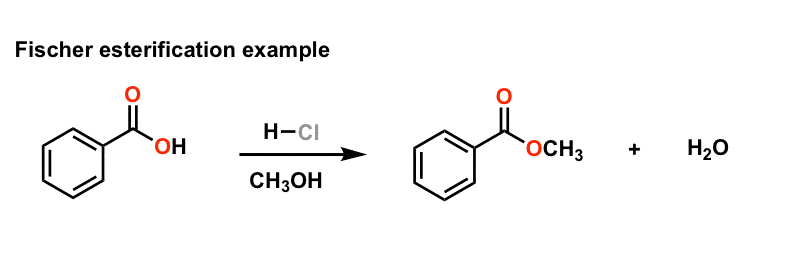
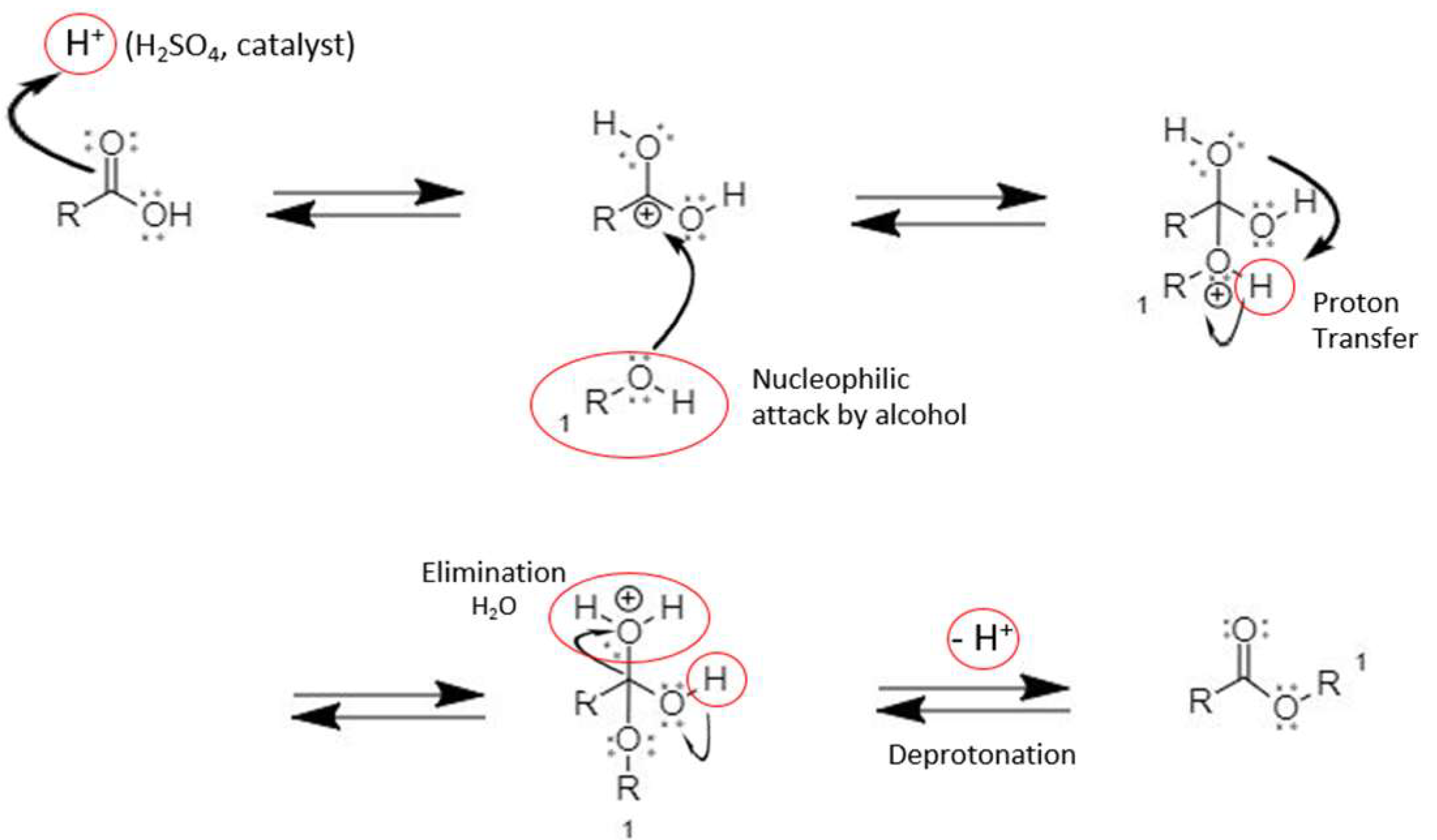
Thus, the required ester is produced. The Fischer esterification Mechanism can simply be described as – Protonation of the carbonyl, followed by the nucleophilic attack on the carbonyl, the proton transfer to the OH group, the removal of water and finally the deprotonation step.
Are Fischer esterification routes mutually exclusive with acetic anhydride routes?
However, in many carefully designed syntheses, reagents can be designed such that acid anhydrides are generated in situ and carboxylic acid byproducts are reactivated, and Fischer esterification routes are not necessarily mutually exclusive with acetic anhydride routes.
What is esterification in chemistry?
Esterification is the reaction of an acid (acid carboxyl group condensation) in the presence of a catalyst with alcohol (alcohol hydroxyl group). Put your understanding of this concept to test by answering a few MCQs. Click ‘Start Quiz’ to begin!

Preparation of esters via Fischer esterification Organic chemistry Khan Academy

Fischer Esterification Reaction Mechanism

Fischer esterification Carboxylic acids and derivatives Organic chemistry Khan Academy
|
Fischer Esterification
Introduction: Esters are one of the most common derivatives of carboxylic acids and are widely distributed in both nature and industry. A typical procedure |
|
5.310 (F19) Fischer Esterification Lab Manual
For the ester above the mechanism involves a second H atom transfer. Fischer Esterification-15. Page 16. EXPERIMENT #5 Fischer Esterification. D. NUCLEAR |
|
22. The Fischer Esterification
Fischer Esterification to Prepare Isopentyl Acetate. Figure 4 shows the mechanism for the acid catalyzed Fischer esterification. First the carbonyl oxygen |
|
Green and Efficient Esterification Method Using Dried Dowex H+/NaI
May 22 2019 is the Fischer esterification |
|
29. The Fischer Esterification
Fischer Esterification to Prepare Isopentyl Acetate. Figure 4 shows the mechanism for the acid catalyzed Fischer esterification. First the carbonyl oxygen |
|
PicoSpin™ 45: The Fisher Esterification Reaction Synthesis of
The simplest approach is. Fisher esterification. By this method esters are produced by refluxing a carboxylic acid and an alcohol in the presence of a |
|
Comparing esterification methods: preparation of aspirin and oil of
The yields for the two experiments show that esterification using an activated carboxylic acid derivative is more efficient than Fischer esterification as the |
|
Simulations and Economic Analyses of Ethyl Acetate Productions by
May 30 2014 ... method have revealed that |
|
A solvent-reagent selection guide for Steglich-type esterification of
Jul 29 2021 7. Methodology often employed in the synthesis of esters includes the traditional acid-catalysed Fischer–Speier method. (catalytic HCl |
|
Electrolytic oxidative coupling of alcohols with aldehydes to form
Traditionally the Fischer esterification method has been employed in the synthesis of esters starting from carboxylic acids and alcohols under acid |
|
Fischer Esterification
A typical procedure to synthesize esters is the Fischer esterification wherein a carboxylic acid is treated with an alcohol in the presence of a mineral |
|
Green and Efficient Esterification Method Using Dried Dowex H+/NaI
May 22 2019 method is regioselective |
|
Exp 22 Fischer Esterification_2017
the pure organic ester which will be a liquid. Figure 3. Fischer Esterification to Prepare Isopentyl Acetate. Figure 4 shows the mechanism for the acid |
|
Synthesis and Investigation of Thermal Properties of Highly Pure
Jun 30 2018 Synthesis Mechanism and Instrumentation. The esters were synthesized based on Fischer esterification process with H2SO4 as a catalyst. |
|
Understanding and Control of Dimethyl Sulfate in a Manufacturing
Dec 23 2011 Process: Kinetic Modeling of a Fischer Esterification Catalyzed ... impurities with a validated analytical method |
|
Exp 29 Fischer Esterification_2016 final
Figure 4 shows the mechanism for the acid catalyzed Fischer esterification. First the carbonyl oxygen of acetic acid is protonated by the acid catalyst. |
|
PicoSpin™ 45: The Fisher Esterification Reaction Synthesis of
By this method esters are produced by refluxing a carboxylic acid and via an esterification reaction between acetic acid and isopentyl alcohol ... |
|
10. Fisher Esterification: Preparation of Banana Oil
Fischer Esterification Figures 19.32 and 19.33 |
|
A Convenient Synthesis of Amino Acid Methyl Esters
May 8 2008 This method is not only compatible with natural amino ... Keywords: Amino acid methyl ester hydrochlorides; amino acids; ... |
|
5.310 (F19) Fischer Esterification Lab Manual
Fischer Esterification: The Organic Synthesis Isolation |
|
Fischer Esterification - MIT OpenCourseWare
Fischer Esterification: The Organic Synthesis, Isolation, Purification, and Fundamental Chemical Engineering Separation Process principles on refluxing |
|
Synthesis of an Ester: Fischer Esterification The ester is synthesised
Weigh compounds directly into the reaction flask Be careful with butyric acid – rancid odour Don't forget to add the sulfuric acid Page 2 Reaction Mechanism |
|
Silica Sulphuric Acid Catalyzed Fischer Esterification - Zenodo
In conclusion the present method is a very efficient and selective protocol for esterification reactions of carboxylic acids with alcohols and a high yield synthesis of |
|
Improved Fischer Esterification of Substituted Benzoic Acid - USM
Abstract: Ethyl-4-fluoro-3-nitro benzoate was synthesised in good yield under closed vessel conditions using a single-mode microwave This simple method |
|
Esters An Introduction
Mixing an alcohol with a carboxylic acid will produce no ester A strong-acid catalyst This is the method used in your esterification reaction Think of this as |
|
Esterification - Beyond Benign
Reference: Fischer Esterification, Organic Chemistry Laboratory Manual, A B Padfas, Greener method introduces the concepts of biobased resources and |
|
Lab 12: Synthesis of an Ester - California State University, Bakersfield
You will be assigned one of two possible esters Esters can be prepared by this method in the presence of an acid catalyst To force the reaction equilibrium to |