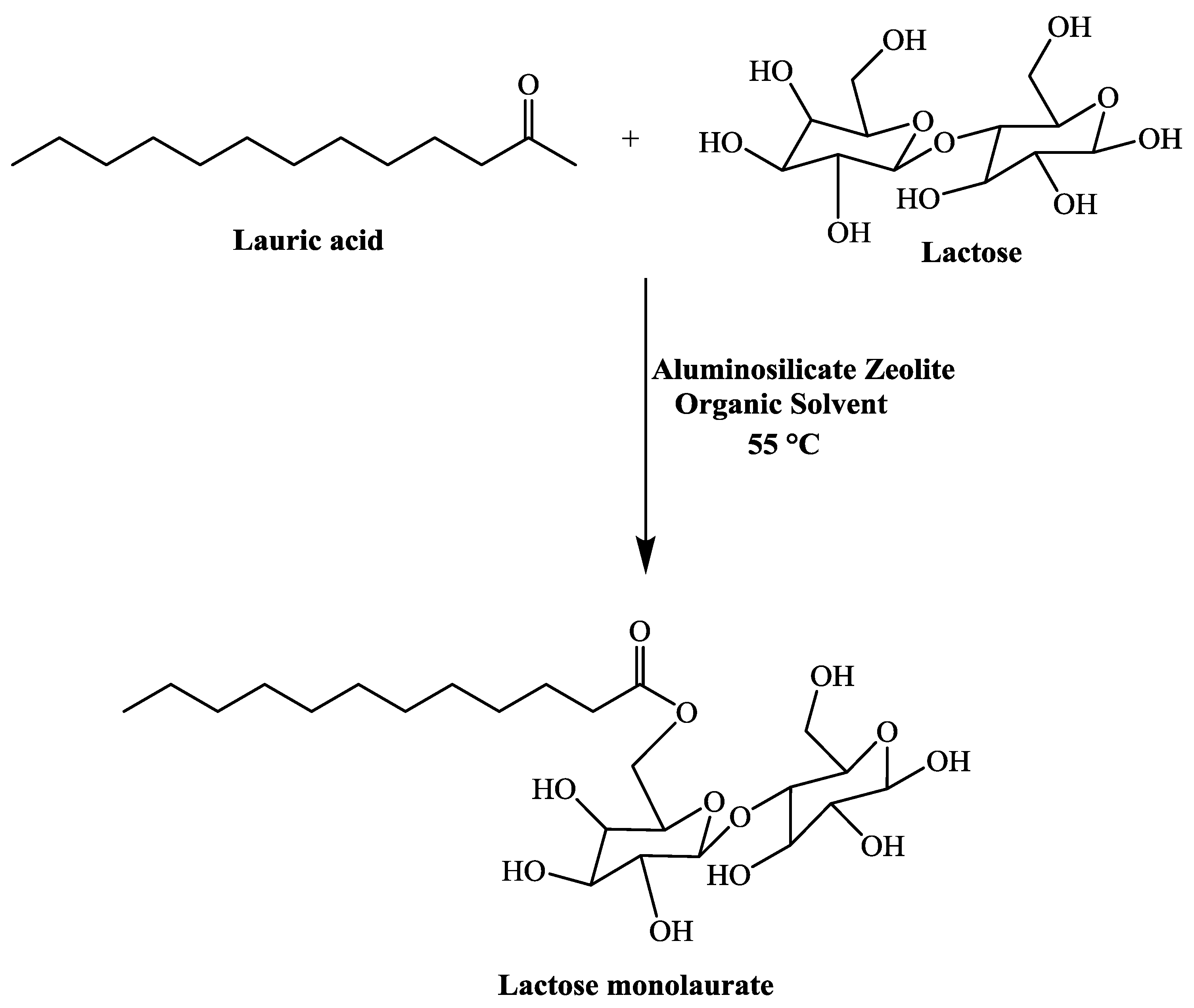
fischer esterification of lauric acid
|
MASSACHUSETTS INSTITUTE OF TECHNOLOGY Department of Chemistry
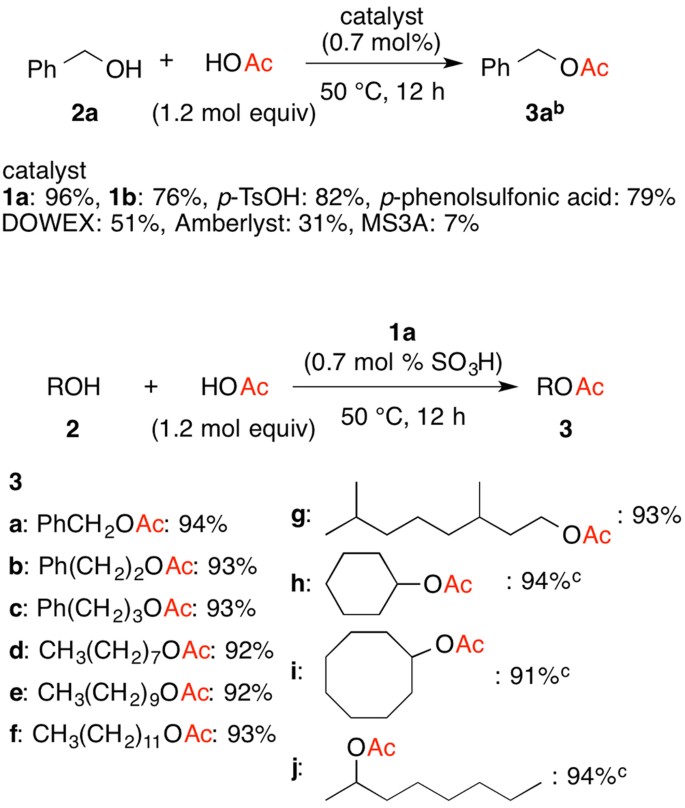
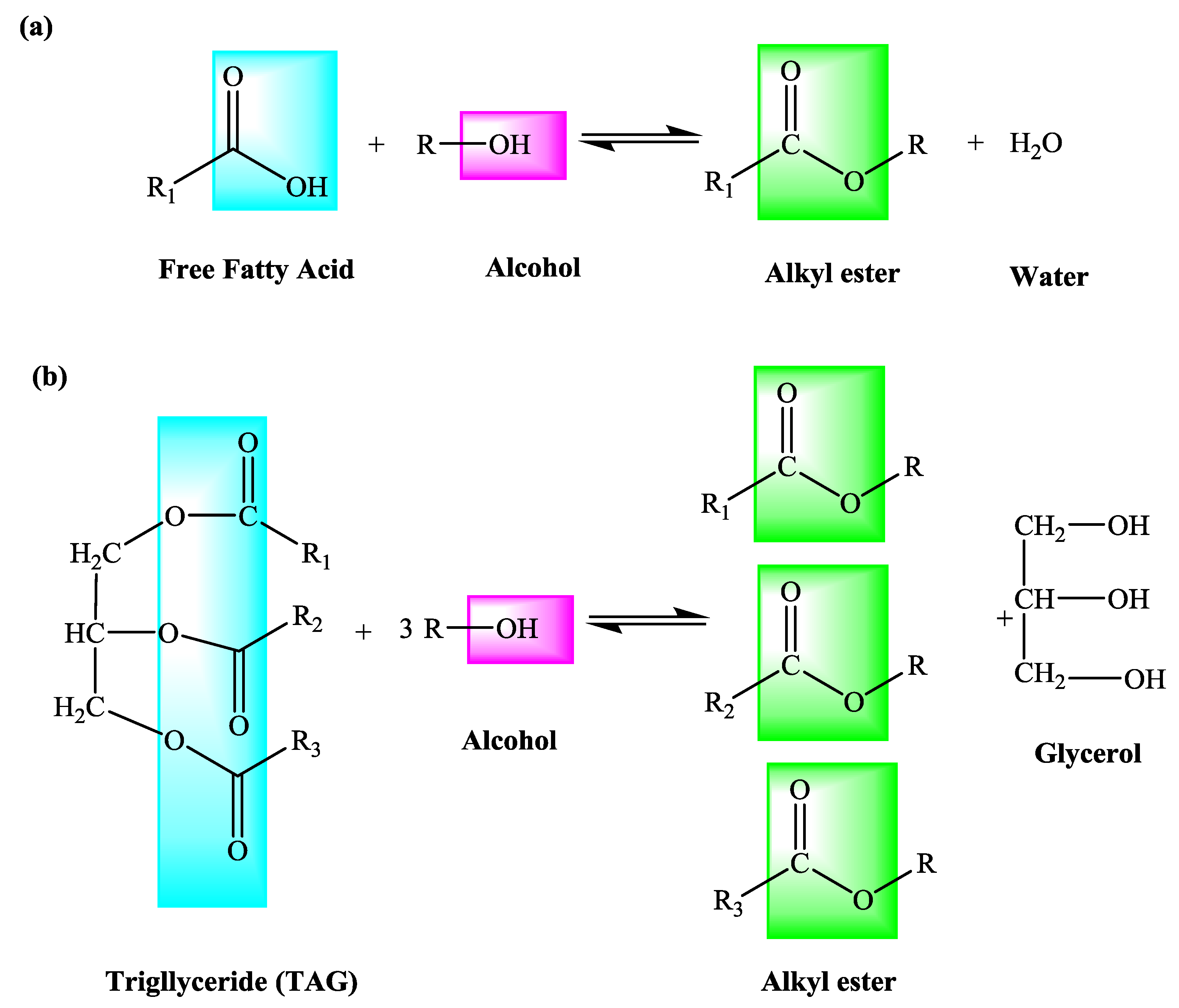
Fischer Esterification A Esterification—Background Esters are found naturally in a variety of fruits vegetables herbs and spices Odors can be complex and generally pleasantly smelling usually the odor of the ester stands out Esters are made by condensing an alcohol with a carboxylic acid |
|
Fischer Esterification
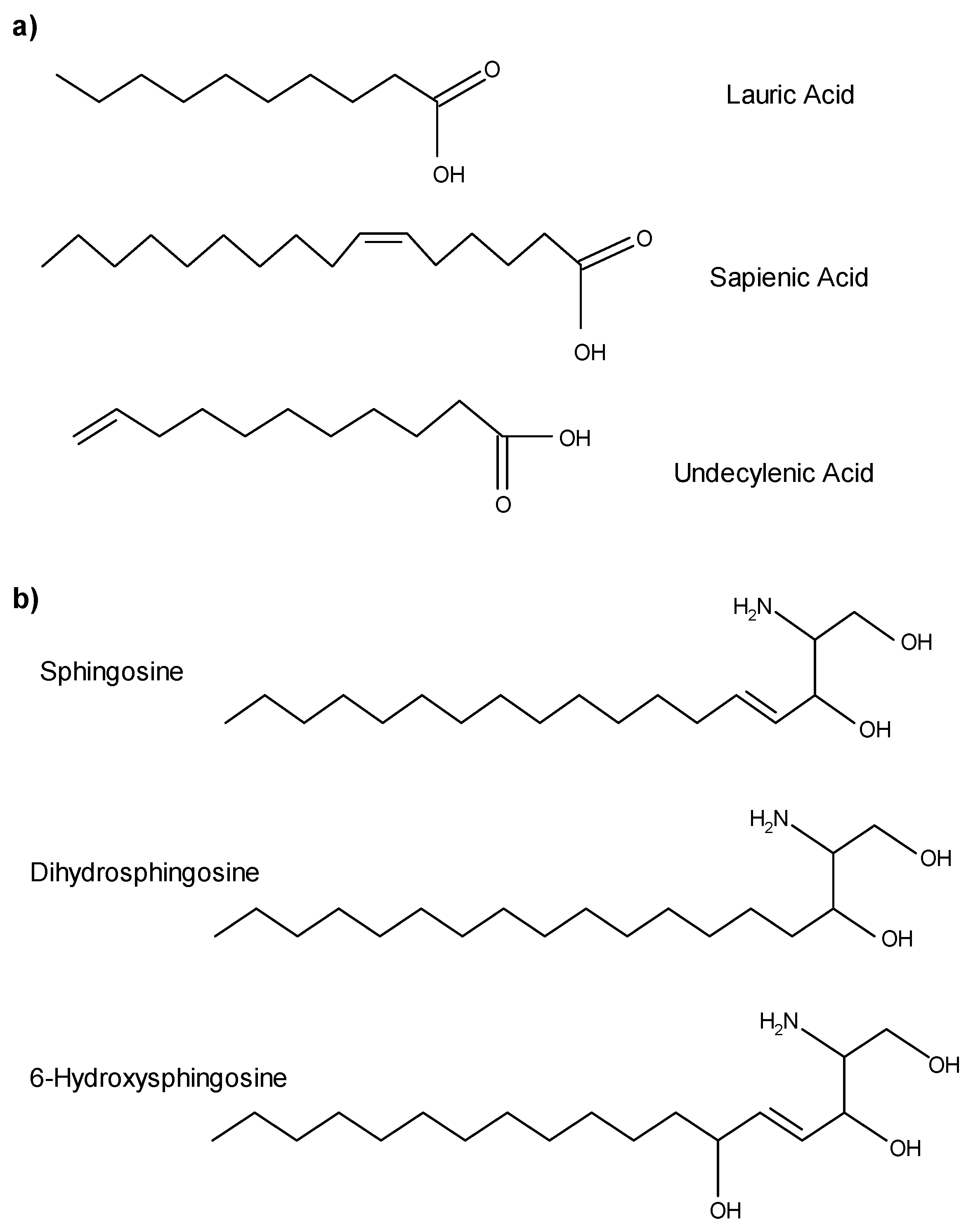
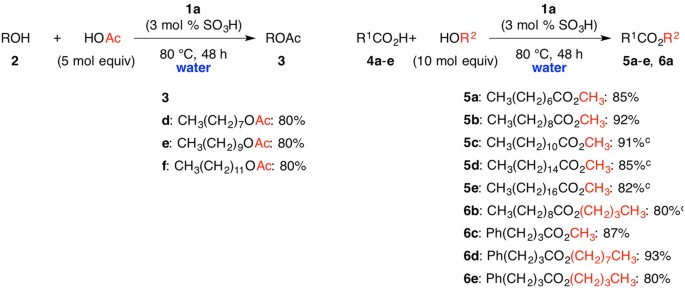
A typical procedure to synthesize esters is the Fischer esterification wherein a carboxylic acid is treated with an alcohol in the presence of a mineral inorganic acid catalyst In this experiment lauric acid (dodecanoic acid) is converted to ethyl laureate Lauric acid is representative of a class of molecules called fatty acids |
What factors affect the reaction chemistry of esterification?
There are several factors that affect the reaction chemistry of esterification. Structure of a molecule and types of functional groups or substitutes associated with carboxylic acids and alcohols determine the speed at which both reagents are esterified as well as the extent of equilibrium.
What are the limitations of Fischer esterification?
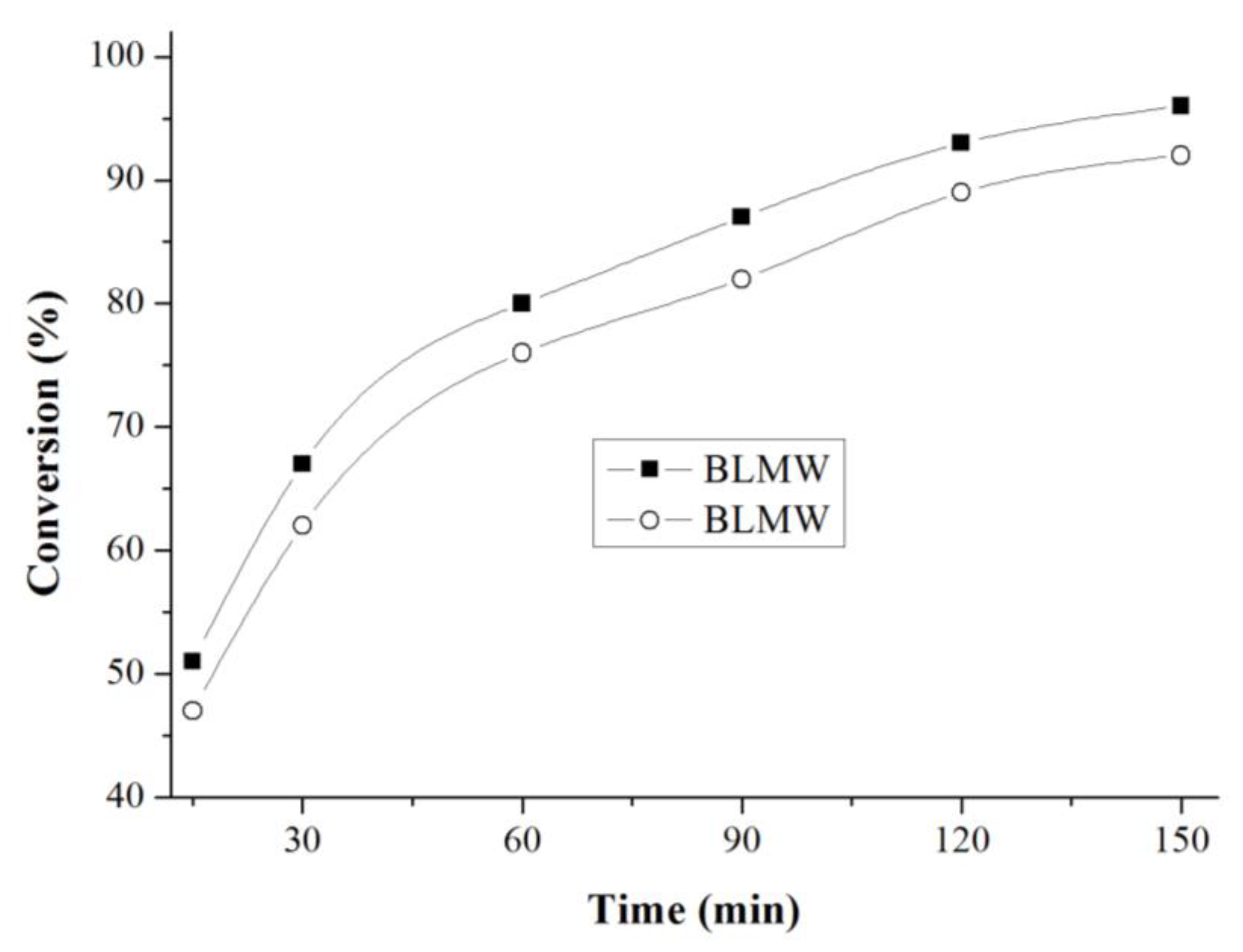
Fischer esterification regarded as the most common and widely practiced process of ester synthesis, faces serious limitations of low conversion and high reaction time attributed largely to establishment of equilibrium. Ester hydrolysis, reverse reaction to esterification, starts by supply of a byproduct- water.
What is a Fisher esterification reaction?
Typical Fisher esterification reaction involves heating a mixture of carboxylic acids and an excess amount of corresponding alcohols in the presence of a catalyst as shown in Eq. (1). The reaction achieves equilibrium after a certain time -- governed by process kinetics and thermodynamics.

Fischer Esterification of Lauric acid

Preparation of esters via Fischer esterification Organic chemistry Khan Academy

Fischer Esterification mechanism. Synthesis of ester from acid and alcohol with mechanism
|
Fischer Esterification
Lauric acid is representative of a class of molecules called fatty acids. These are long straight chain carboxylic acids (C12-C40) found as ester derivatives |
|
IONIC LIQUID SUPPORTED ACID-CATALYSED ESTERIFICATION
NTf2 gave acidic ILs. These acidic ILs were tested as catalysts for esterification reactions. Esterification of alcohol. (methanol) with fatty acid (lauric acid) |
|
Organic Solvent-free Transesterification of Various Starches with
4 janv. 2005 Various Starches with Lauric Acid Methyl Ester and Triacyl Glycerides ... reaction mixture was measured according to Karl Fischer. |
|
University of Malaya
esterification of lauric acid with diethylene glycol were also carried Water content in the sample was analyzed using Mettler Toledo C30 Karl Fischer. |
|
The Reaction of Lauric Acid Esters with Sulfuric Acid
The Reaction of Lauric Acid Esters with Sulfuric Acid. By Robert A. Bauman and Irving J. tán B) until both Karl Fischer aquametry10 and alkalimetry. |
|
334L S-12 syllabus
Fischer Esterification of Lauric Acid. Wade sections 20-10 |
|
Esterification of lauric acid with lauryl alcohol using cross-linked
Esterification of lauric acid with lauryl alcohol using cross-linked enzyme crystals: Solvent effect tained constant as determined with a Karl Fischer. |
|
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters and learn how to predict the products of ester synthesis and hydrolysis reactions. • Learn |
|
Scientific Opinion on the safety of sucrose esters of fatty acids
acids however this evaluation did not consider sucrose esters of lauric acid. The Panel considers that although the toxicological data on the sucrose ester |
|
1. a. The first reaction is an esterification leading to a methyl ester
Sulfuric acid is used as catalyst in the Fischer esterification. It protonates the carbonyl group of the carboxylic acid and not the hydroxyl function. The |
|
Esterification of lauric acid with lauryl alcohol using cross - CORE
In all experiments the water concentration was main- tained constant as determined with a Karl Fischer Titrator (Spectralab MA-101-B, Alfa Instruments, New |
|
Fischer Esterification
Lauric acid is representative of a class of molecules called fatty acids These are long, straight chain carboxylic acids (C12-C40) found as ester derivatives in oils, |
|
IONIC LIQUID SUPPORTED ACID-CATALYSED ESTERIFICATION
was used as catalyst for the esterification reaction of fatty acid Various acids namely (methanol) with fatty acid (lauric acid) using ionic liquid BMI NTf2 combined with and reaction medium for fischer esterification Journal of Molecular |
|
Fischer Esterification - MIT OpenCourseWare
Fischer Esterification: The Organic Synthesis, Isolation, Purification, and Characterization of a Natural Organic Synthesis: Synthesizing an ester from an acid catalyzed reaction of a carboxylic acid and alcohol Benzoic Acid Eye, skin and |
|
Fischer Esterification - MIT OpenCourseWare
Fischer Esterification: The Organic Synthesis, Isolation, Purification, and Characterization of a Natural Organic Synthesis: Synthesizing an ester from an acid catalyzed reaction of a carboxylic acid and alcohol Benzoic Acid Eye, skin and |
|
Kinetic study of lipase catalyzed esterification reactions in water-in
periodically after measuring it with a Metrohm Karl-Fischer titrator Activity Measurements The reaction of the esterification of lauric acid and (-)menthol was |


![PDF] Ionic liquid supported acid-catalysed esterification of PDF] Ionic liquid supported acid-catalysed esterification of](https://i1.rgstatic.net/publication/306076459_Methyl_Laurate_Characterization_from_Enzymatic_Esterification-Transesterification_Process_of_Virgin_Coconut_Oil_VCO/links/5ee763b592851ce9e7e3cc34/largepreview.png)

![PDF] Ionic liquid supported acid-catalysed esterification of PDF] Ionic liquid supported acid-catalysed esterification of](https://d3i71xaburhd42.cloudfront.net/c40957e1468910b731549c416621771ae018d9d5/4-Figure3-1.png)
![PDF] Ionic liquid supported acid-catalysed esterification of PDF] Ionic liquid supported acid-catalysed esterification of](https://d3i71xaburhd42.cloudfront.net/c40957e1468910b731549c416621771ae018d9d5/6-Table2-1.png)
![PDF] Ionic liquid supported acid-catalysed esterification of PDF] Ionic liquid supported acid-catalysed esterification of](https://0.academia-photos.com/attachment_thumbnails/37076818/mini_magick20190305-24145-jzr263.png?1551780226)

![PDF] Ionic liquid supported acid-catalysed esterification of PDF] Ionic liquid supported acid-catalysed esterification of](https://0.academia-photos.com/attachment_thumbnails/34955523/mini_magick20180819-11999-1nx5cg6.png?1534668320)





![PDF] Ionic liquid supported acid-catalysed esterification of PDF] Ionic liquid supported acid-catalysed esterification of](https://pubs.rsc.org/image/article/2019/GC/c9gc00901a/c9gc00901a-s2_hi-res.gif)



















![PDF] Ionic liquid supported acid-catalysed esterification of PDF] Ionic liquid supported acid-catalysed esterification of](https://patentimages.storage.googleapis.com/c0/90/b7/2b79f8bf573547/US20160289161A1-20161006-C00020.png)