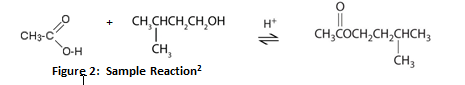fischer esterification sodium bicarbonate
How does sodium bicarbonate remove acid from a solution?
The sodium bicarbonate is very effective in removing acids from solutions. The carbonate ion reacts with the free H+ ions from the acid to make carbonic acid which dissociates into carbon dioxide and water. The sodium reacts with the acetate and sulfate ions to produce a salt that will dissolve in water over the organic ester solution.
What is a carboxylic acid ester?
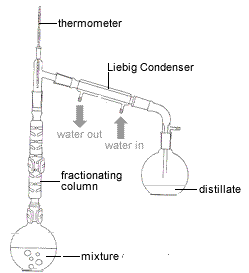
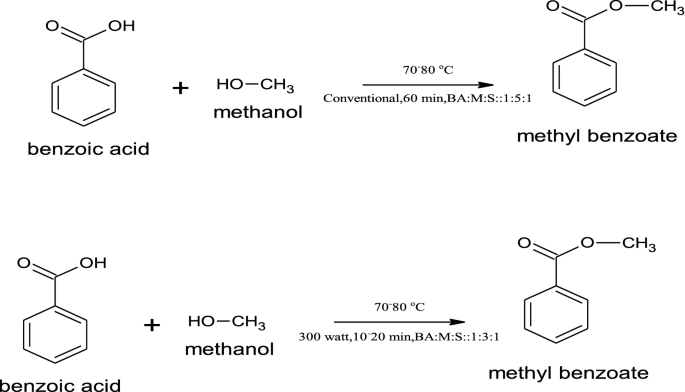
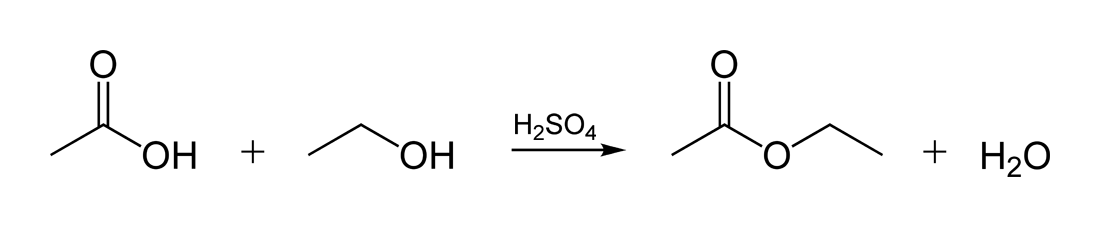
Esters are one of the most common derivatives of carboxylic acids and are widely distributed in both nature and industry. A typical procedure to synthesize esters is the Fischer esterification, wherein a carboxylic acid is treated with an alcohol in the presence of a mineral inorganic acid catalyst.
|
Fischer Esterification
A typical procedure to synthesize esters is the Fischer esterification aqueous sodium bicarbonate solution (a strong base to react with acids). |
|
Experiment 14A: Isopentyl Acetate
14 oct. 2020 This experiment focused on the synthesis of an ester by a Fischer ... 5% aqueous sodium bicarbonate solution was added to the product to ... |
|
PicoSpin™ 45: The Fisher Esterification Reaction Synthesis of
via an esterification reaction between acetic acid and isopentyl alcohol layer with two more 25 mL portions of sodium bicarbonate solution (NaHCO3). |
|
A Visual Organic Chemistry Reaction: The Synthesis of 4-Amino-3
23 juin 2020 nitrobenzoic Acid Methyl Ester via Fischer Esterification ... quenched with saturated sodium bicarbonate solution. |
|
1. a. The first reaction is a Fischer esterification in which the phenol
The portion that dissolves in the organic layer can be extracted with sodium bicarbonate. c. The sulfuric acid is used as catalyst in the reaction. The carbonyl |
|
Synthèse destérification de Fischer dacétate disopentyle (huile de
bicarbonate de sodium (NaHCO3). 9. Après élimination de l'acide acétique lavez la phase organique en utilisant les deux parts. |
|
10. Fisher Esterification: Preparation of Banana Oil
Fischer Esterification Figures 19.32 and 19.33 |
|
Lab 12: Synthesis of an Ester
Esters can be prepared by this method in the presence of an acid catalyst. removed during the extraction by washing with sodium bicarbonate and water. |
|
EXPERIMENT 5 SYNTHESIS OF ESTERS USING ACETIC
saturated aqueous sodium bicarbonate (NaHCO3(aq)) This reaction termed Fischer esterification in honor of its discoverer |
|
Normal and Pseudo Esters of 2-Benzoylbenzoic Acid Types12
Fischer esterification.6 Indeed pseudo methyl. 2-benzoylbenzoate was saturated sodium bicarbonate solution |
|
Synthesis of an Ester: Fischer Esterification The ester is synthesised
The ester is synthesised via a Fischer esterification – the reaction of NaHCO3 R O O + Na CO2 + H2O H2SO4 + 2NaHCO3 Na2SO4 2CO2 + 2H2O + |
|
Fischer Esterification
A typical procedure to synthesize esters is the Fischer esterification, wherein a aqueous sodium bicarbonate solution (a strong base to react with acids) |
|
Lab 12: Synthesis of an Ester - California State University, Bakersfield
Esters can be prepared by this method in the presence of an acid catalyst removed during the extraction by washing with sodium bicarbonate and water |
|
Fischer Esterification - MIT OpenCourseWare
Fischer Esterification: The Organic Synthesis, Isolation, Purification, and sodium bicarbonate solution to remove any traces of the carboxylic acid CO2 gas |
|
Fischer Esterification - MIT OpenCourseWare
Fischer Esterification: The Organic Synthesis, Isolation, Purification, and sodium bicarbonate solution to remove any traces of the carboxylic acid CO2 gas |
|
1 a The first reaction is a Fischer esterification in which the phenol
portion that dissolves in the organic layer can be extracted with sodium bicarbonate c The sulfuric acid is used as catalyst in the reaction The carbonyl group of |
|
Synthesis of Isopentyl Acetate (Banana Oil) - Thermo Fisher Scientific
picoSpin™ 45: The Fisher Esterification Reaction Synthesis of Extract the organic layer with three 25 mL portions of 5 aqueous sodium bicarbonate solution |
|
Esterification - Beyond Benign
The esterification reaction Reference: Fischer Esterification, Organic Chemistry Laboratory Manual, A B Padfas, Hayden-McNeil, Sodium bicarbonate (5 ) |
|
Esters An Introduction
mixture – 7 esters, 3 carboxylic acids, and 7 essential oils (other natural saturated aqueous sodium bicarbonate (caution: evolution of CO2 may occur and |




















![Ph Ch 1261 Fischer Esterification Of Methyl Benzoate 2 [od4p3wgv1wlp] Ph Ch 1261 Fischer Esterification Of Methyl Benzoate 2 [od4p3wgv1wlp]](https://i1.rgstatic.net/publication/323983742_Esterification_process_catalyzed_by_ZSM-5_Zeolite_synthesized_via_modified_hydrothermal_method/links/5ac7702a4585151e80a39f7b/largepreview.png)