font study fsgs
What are the criteria for FSGS?
A working proposal, commonly referred to as the Columbia Classification, distinguishes 5 mutually exclusive morphologic variants: collapsing, tip, cellular, perihilar, and not otherwise specified (NOS), which can be applied to primary and secondary forms of FSGS.
What are the 5 types of FSGS?
What tests are done to find out if I have focal glomerulosclerosis or FSGS? A blood test, urine test, and a kidney biopsy will determine if you have FSGS.
Urine test: A urine test will help find protein and blood in your urine.How do you test for FSGS?
FSGS perihilar variant
Defining criteria include both of the following: a) there must be at least 1 glomerulus with perihilar hyalinosis, with or without sclerosis; and b) more than 50% of glomeruli with segmental lesions must have perihilar sclerosis and/or hyalinosis.
|
NIH Public Access
FSGS; pharmacokinetics; nephrotic syndrome; adalimumab; tumor necrosis factor-?; antifibrotic m2 were eligible to participate in the FONT study. |
|
PODO: Trial Design: Phase 2 Study of PF-06730512 in Focal
3 avr. 2021 The. Novel Therapies to Treat Resistant Focal Segmental Glo- merulosclerosis: Phase II Clinical Trial was conducted by the FSGS/FONT ... |
|
1 KDIGO GN Guideline update – Evidence summary Idiopathic focal
21 août 2018 Idiopathic focal segmental glomerulosclerosis (FSGS) in adults ... Data from the FONT I 2009 study could not be meta-analysed as different ... |
|
Perturbations in podocyte transcriptome and biological pathways
12 sept. 2022 FSGS has been supported by multiple studies. ... Report of the FONT (Novel Therapies for Resistant. FSGS) study group. Am J Kidney Dis ... |
|
Intrinsic tumor necrosis factor-#_#x03B1; pathway is activated in a
16 mai 2019 Activation of TNF? pathway genes occurs in podocytes of FSGS patients ... phase of the Novel Therapies for Resistant FSGS FONT |
| Identification of glomerular and podocyte-specific genes and |
|
Circulating Permeability Factors in Idiopathic Nephrotic Syndrome
glomerulosclerosis (FSGS) and in recurrence after renal transplantation. Evidence for plasma factors the FONT (Novel Therapies for Resistant FSGS) study. |
|
Therapeutic trials in adult FSGS: lessons learned and the road forward
20 mai 2021 Focal segmental glomerulosclerosis (FSGS) ... A study of patients with FSGS in ... glomerulosclerosis: report of the font clinical trial. |
| Biomarkers in Primary Focal Segmental Glomerulosclerosis in |
|
Efficacy of galactose and adalimumab in patients - ResearchGate
Background: Patients with resistant focal segmental glomerulosclerosis (FSGS) who are unresponsive The FONT (Novel Therapies for Resistant FSGS) study |
|
FORM252 FSGS Novel Therapies (FONT-II) Long-term Monitoring
FONT-II F252 Page 1 of 2 FORM252 FSGS Novel Therapies (FONT-II) Long- term Monitoring Form (Form # 252) FAX COMPLETED FORM TO FONT STUDY |
|
FSGS Novel Therapies (FONT-II) - NIDDK Central Repository
22 déc 2011 · FAX COMPLETED FORM TO FONT STUDY DESK UMich (734) 232-2353 NYU (212) 263-4053 FONTII Barcode from tube Date sample was |
|
FSGS - KDIGO
Idiopathic focal segmental glomerulosclerosis (FSGS) in adults Data from the FONT I 2009 study could not be meta-analysed as different outcomes were |
|
Permeability factors in nephrotic syndrome and focal segmental
FSGS Experimental studies have identified podocyte mechan- isms that protect or limit damage rosiglitazone in FSGS: I Report of the FONT Study Group |
|
Glomerular Diseases: FSGS - CJASN
29 août 2013 · Introduction FSGS is the most common primary glomerular histo- erence text ( 4) However, the FSGS: III Report of the FONT study group |
|
DUET: A Phase 2 Study Evaluating the Efficacy and Safety of - JASN
The FSGS partial remission end point was achieved in 28 of sparsentan- treated and 9 of irbesartan-treated patients (P=0 04) After 8 weeks of treatment, BP |












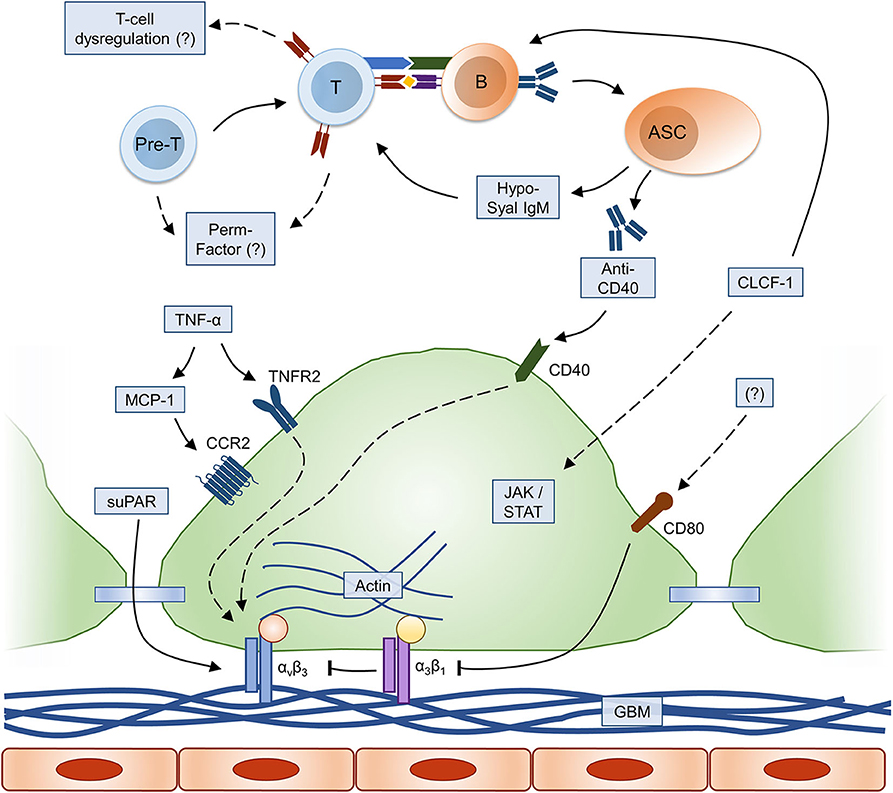



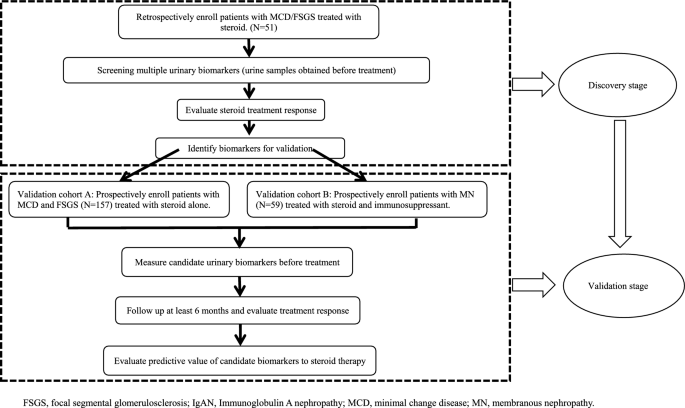












![Full text] Optimal management of primary focal segmental Full text] Optimal management of primary focal segmental](https://els-jbs-prod-cdn.jbs.elsevierhealth.com/cms/attachment/bfe26e51-2204-4c23-824e-f6185b01a2c6/gr1.gif)




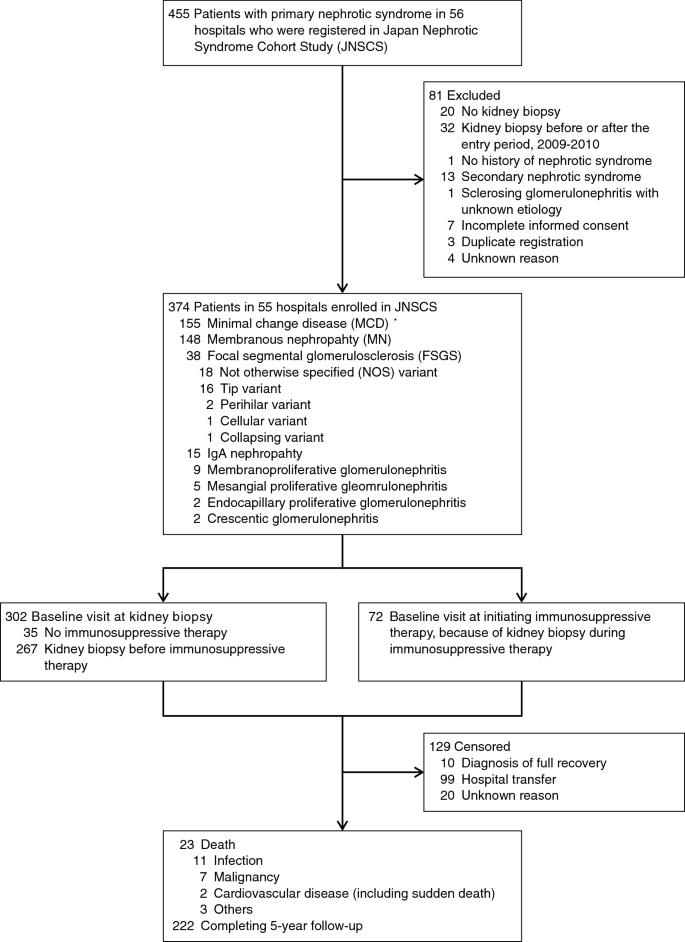
![PDF] Focal segmental glomerulosclerosis: a case study with review PDF] Focal segmental glomerulosclerosis: a case study with review](https://www.mdpi.com/ijms/ijms-21-02095/article_deploy/html/images/ijms-21-02095-ag.png)
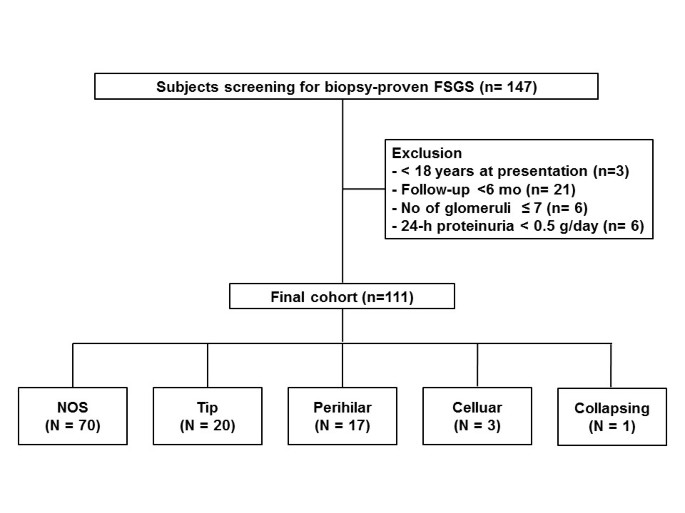


![PDF] Focal segmental glomerulosclerosis: a case study with review PDF] Focal segmental glomerulosclerosis: a case study with review](https://www.mdpi.com/jcm/jcm-07-00265/article_deploy/html/images/jcm-07-00265-g001-550.jpg)









