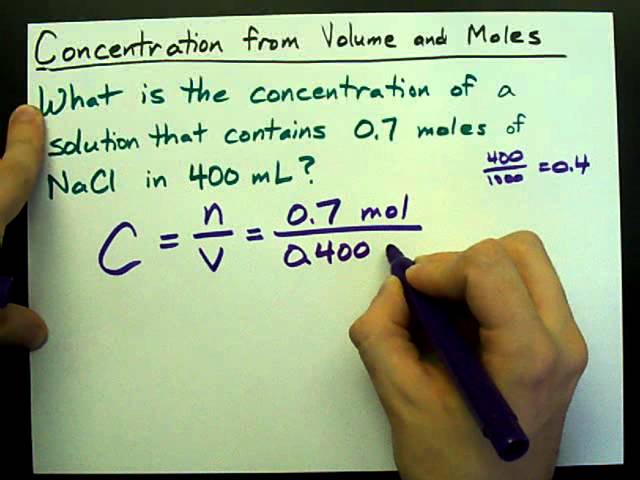how to find concentration of a solution calculator
How do you find the new concentration of a solution calculator?
Concentration of one solution is equal to the molarity times volume of the other solution (M₁V₁ = M₂V₂).
Units should remain constant on both sides of the equation.
This dilution calculator can be used to find missing values needed to dilute a solution.How do I calculate concentration of a solution?
Write out the equation C = m/V, where m is the mass of the solute and V is the total volume of the solution.
Plug in the values you found for the mass and volume, and divide them to find the concentration of your solution.
- Determine the density (g/dm³) of the solute.
- Find the solute's molar mass (g/mol).
- Multiply the molar mass by the molarity.
- Divide the result from Step 3 by the density of your solute.
|
Calculating pH Calculating the Hydronium Ion Concentration from
To calculate the pH of an aqueous solution you need to know the concentration of the On a calculator calculate 10-8.34 |
|
System Of Equation Solution Calculator
Equation Solution Calculator but stop taking place in harmful downloads. Place each piece of potato in a diff erent concentration of sugar solution. 4. |
|
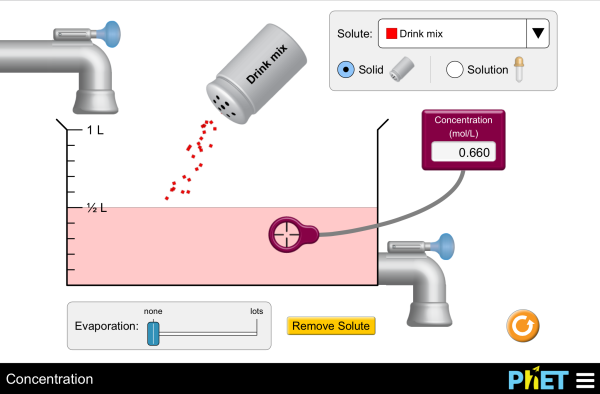
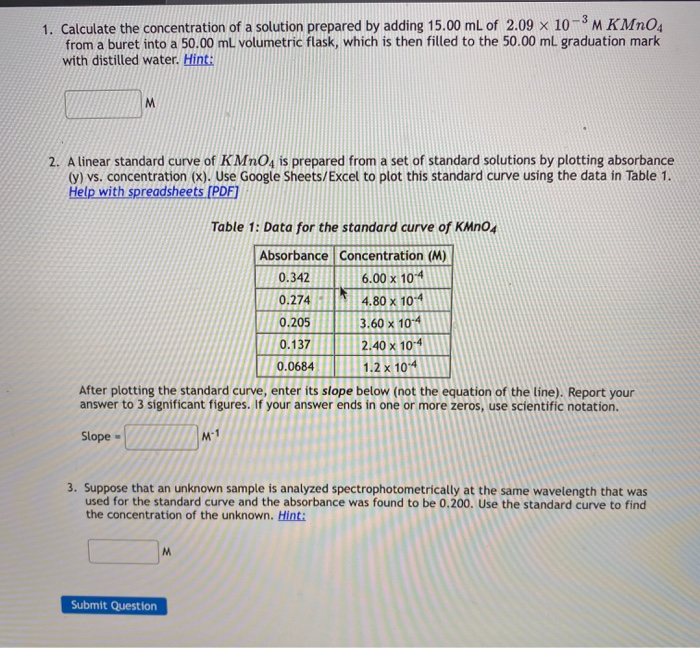
1 Calculation of concentration of a solution The study material
A direct proportionality between the concentration (c) and a related substance amount (n) is used. (*) 1 M solution (read: one molar solution) means that 1 L of |
|
Calculation of concentrated solution viscosities from single?point
Thus the viscosity of a cyclohexanone solution of 26 |
|
Heparin Drip Calculation Reference
Heparin Drip Calculation Reference. (sample calculations for reference only!) Heparin Infusion Rate: Total Units (in IV bag) = Units/hour. |
|
Chemistry of nutrient solution - Use of a Nutrient Solution calculator
Use of a Nutrient Solution calculator 3) Recipe: concentration (milli- or micro-moles per ... ?calculation of the nutrient solution and its cost;. |
|
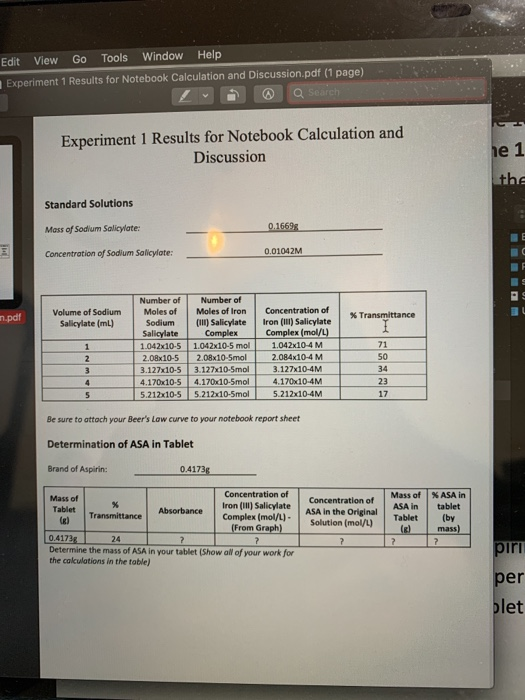
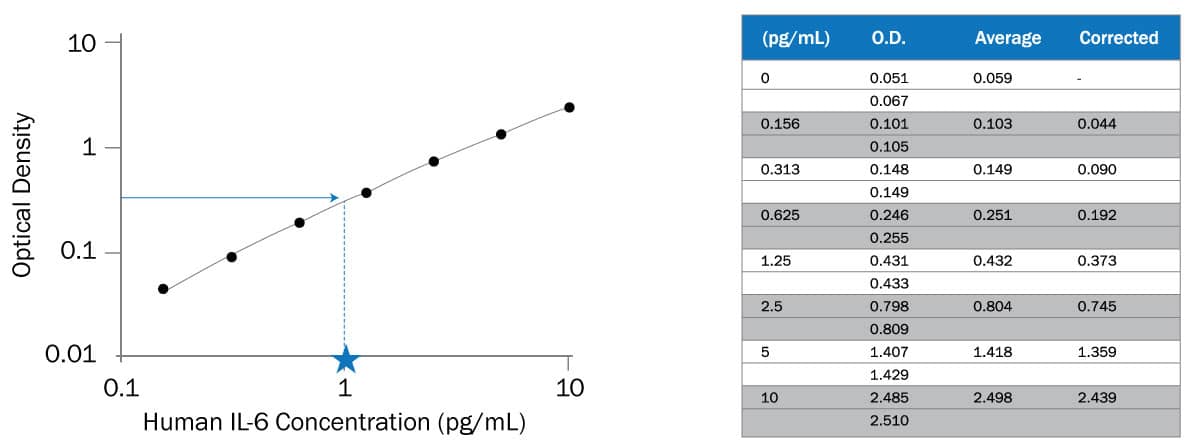
Beers Law: Determining the Concentration of a Solution
Calculations – Complete in your lab notebook. Show all steps! Use significant figures and units. • Calculation for the molarity of the Allura Red stock solution |
|
Medical Calculations
Performing the proportion equation by cross multiplication: Percent solution calculations are used to prepare a specific concentration of solution. |
|
Calculation Refresher Ratio and Proportion Method To solve for x
How can we use the proportion calculation to get the answer? You have KCl in some concentration and ... concentrated KCl solution is to be added. |
|
Nsg 231 Calculating IV Solution Concentration
HCL (Xylocaine) in 5% D/W. The final solution has a volume of 250 ml. You realize that the IV solution contains ____ mg of lidocaine HCL per ml. To find the |
|
1 Calculation of concentration of a solution The study material
Molarity (mol x L-1 = mol x dm-3 = mol/L = mol/dm = M) The molarity can be calculated either using the formula c = n/V (c = molarity, n = substance amount in moles, V = final volume of the solution in L) or directly from the definition* of the molar concentration |
|
Calculating Concentrations and Mixing Reagents
I Mixing a Solution of a Desired Concentration as the number of moles of substance per liter of solvent (molarity, or M/L) Chemical Z? Use the formula: |
|
SOLUTION CONCENTRATIONS
Calculate the solution concentration, as percent by mass, given that 0 290 g of Mass of solution = 0 290 g + 75 00 g = 75 29 g (calculator and correct answer) |
|
Calculating the pH and ion concentrations on a Calculator (TI-83 or
Problem type: given ion concentration, find the pH To obtain the pH of a solution, you must compute the negative log of the hydrogen ion concentration [H+] Step |
|
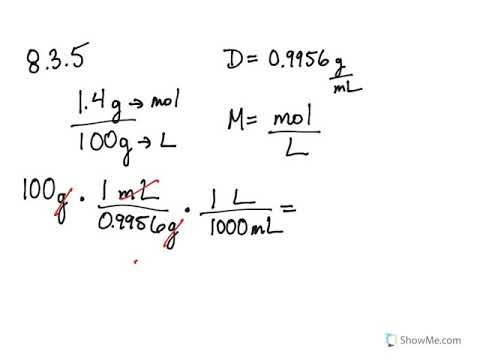
Volume = Mass / Concentration
Always check your units to make sure you‟ve remembered the equation correctly: Molarity = moles/liter; MW = grams/mole, and Volume = Liters So the formula |
|
Dilution Calculator - ppb, ppm, ppt, pph Calculators for preparing
utilized to perform dilution calculations when working with solutions having the dilution factor or fold dilution calculations for solutions with the concentration An alternative and commonly-used notation for this equation is M1V1 = M2V2, |
|
Calculating final, or working, concentrations for - Bates College
10 mai 2012 · V1 = the volume of the stock solution you need; this is your unknown that you solve for C2 = 0 1 M Tris, the final concentration V2 = 2 ml, the |
|
AT A GLANCE/ PHARMACY CALCULATIONS - Mathcentre
Calculating how much ingredient to add to a product to achieve a v/v solution to increase it in strength to a 20 v/v solution? Method AT A GLANCE/ PHARMACY CALCULATIONS NB: When increasing a concentration use the initial and |











/how-to-calculate-normality-609580final2-0d5efa5a961f4fa0a7efc780921faee1.png)