how to remove excess alcohol from fischer esterification
|
Fischer Esterification
To drive the equilibrium to make more ester excess alcohol is added following Le Upon cooling of the conical vial remove the spin vane (by using the spin |
How do you remove ethanol from ester?
The most commonly-used method of stripping alcohol from an ester product makes use of an aerated agitation tank, by which an aerated dissolution is effected such that an effective gas-liquid contact area is formed.
How is alcohol removed by esterification?
The key chemical reaction responsible for ethanol removal is esterification through a reaction between ethanol and acetic acid.
Ethanol reacts with acetic acid to produce water and ethyl acetate, which is a harmless ester found in many food products.To drive the equilibrium to make more ester, excess alcohol is added following Le Chatelier's Principle.
In addition, an acid catalyst is needed.
Its role is to facilitate the nucleophilic attack of the alcohol at the carbonyl carbon of the carboxylic acid.
What is Fischer esterification with alcohol in excess?
Fischer Esterification is an organic reaction which is employed to convert carboxylic acids in the presence of excess alcohol and a strong acid catalyst to give an ester as the final product.
This ester is formed along with water.
|
The ester is synthesised via a Fischer esterification – the reaction of
This removes the bulk of the water soluble compounds from the organic layer. Namely excess alcohol (assuming it is water soluble) and/or excess carboxylic acid |
|
Fischer Esterification
To drive the equilibrium to make more ester excess alcohol is added following Le. Chatelier's Principle. Eq.1. In addition |
|
Esters. An Introduction.
Mixing an alcohol with a carboxylic acid will produce no ester. materials because it is easier to remove excess carboxylic acid than excess alcohol |
|
Synthesis of Isopentyl Acetate.pdf
To prepare isopentyl acetate from isopentyl alcohol and acetic acid by the Fischer esterification reaction. Introduction. Esters are derivatives of carboxylic |
|
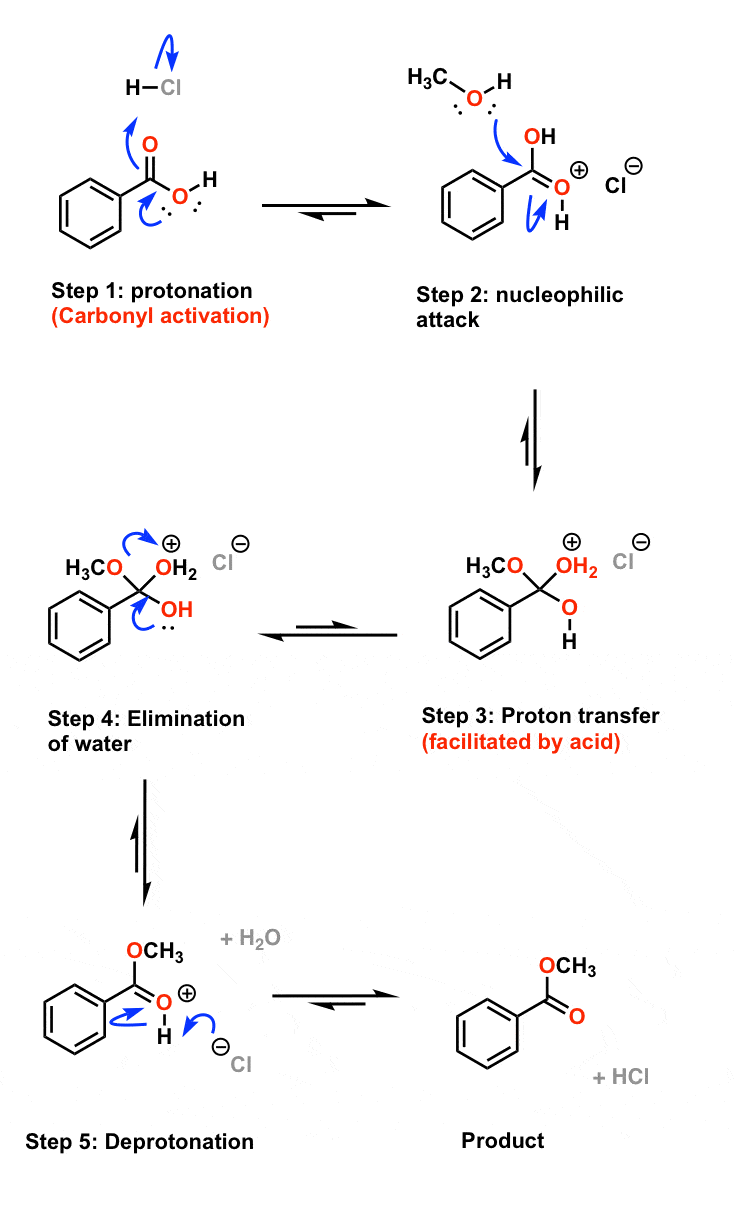
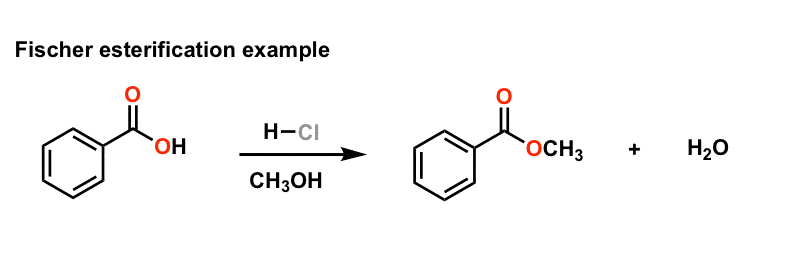
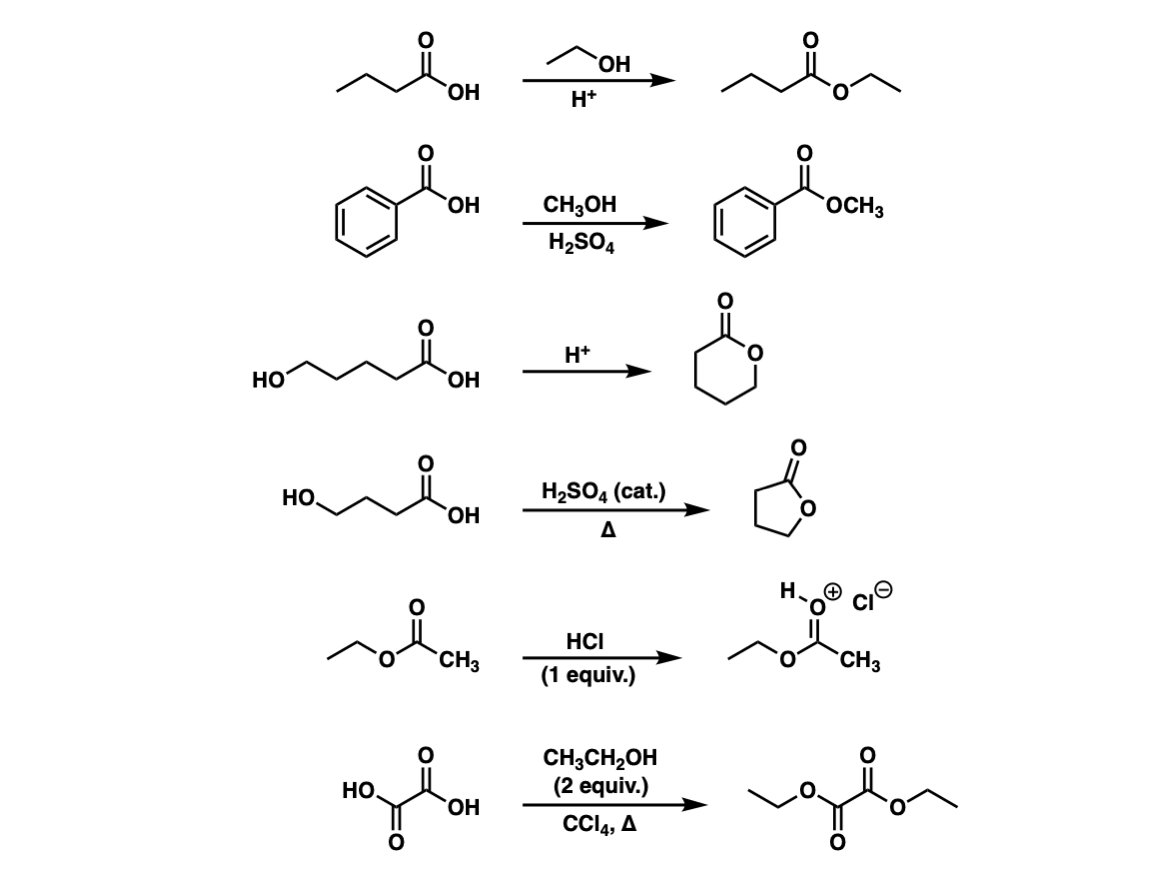
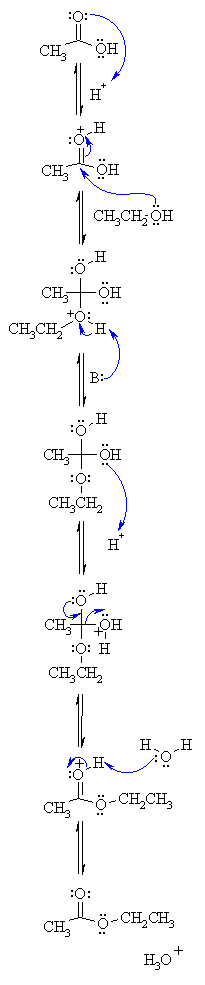
Example: esterification of benzoic acid to methyl benzoate.
MECHANISM (Fischer esterification) This can be accomplished either by removing one or more of the products from the reaction ... 3) excess alcohol. |
|
Lab 12: Synthesis of an Ester
In the isolation procedure much of the excess carboxylic acid and the remaining alcohol will be removed during the extraction by washing with sodium |
|
Synthesis and Investigation of Thermal Properties of Highly Pure
30 juin 2018 Carboxylic esters can be obtained via Fischer esterification of ... paper and the excess organic solvent removed in the rotary evaporator. |
|
EXPERIMENT 5 SYNTHESIS OF ESTERS USING ACETIC
This reaction termed Fischer esterification in honor of its discoverer |
|
PicoSpin™ 45: The Fisher Esterification Reaction Synthesis of
via an esterification reaction between acetic acid and isopentyl alcohol (3-methylbutanol) solution to remove excess acetic acid (Note: Be careful |
|
Synthetic efforts towards the development of a microwave synthesis
reaction received then name of Fischer esterification to honor the remaining excess alcohol and water being discarded in liquid waste container. |
|
Fischer Esterification
A typical procedure to synthesize esters is the Fischer esterification, wherein a To drive the equilibrium to make more ester, excess alcohol is added Remove the cap and immediately and rapidly reassemble the reaction apparatus Acetyl |
|
Synthesis of an Ester: Fischer Esterification The ester is synthesised
The ester is synthesised via a Fischer esterification – the reaction of This removes the bulk of the water soluble compounds from the organic layer Namely , excess alcohol (assuming it is water soluble) and/or excess carboxylic acid, |
|
Esters An Introduction
Mixing an alcohol with a carboxylic acid will produce no ester it is easier to remove excess carboxylic acid than excess alcohol, and because it is difficult |
|
Lab 12: Synthesis of an Ester - California State University, Bakersfield
In this experiment you will react a carboxylic acid and an alcohol under acidic right (in favor of the ester), one of the starting materials must be used in excess R As the carboxylic acid is more easily removed from the reaction mixture, it will |
|
Fischer Esterification - MIT OpenCourseWare
Fischer Esterification: The Organic Synthesis, Isolation, Purification, and Esters are made, by condensing an alcohol with a carboxylic acid The reaction refluxing remove any acid impurities from the ester by extraction with Many of the molecular ions will be formed with enough excess energy to undergo subsequent |
|
Fischer Esterification - MIT OpenCourseWare
Fischer Esterification: The Organic Synthesis, Isolation, Purification, and Esters are made, by condensing an alcohol with a carboxylic acid The reaction refluxing remove any acid impurities from the ester by extraction with Many of the molecular ions will be formed with enough excess energy to undergo subsequent |
|
Synthesis of Isopentyl Acetate
Esterification generally refers to the formation of esters from alcohol and carboxylic Fischer esterification is an example of an acyl transfer reaction After the reaction is complete, you will remove the excess acetic acid and sulfuric acid |
|
Experiment 18 — Synthesis of Esters
acid and an alcohol, a process known as Fischer esterification The reaction is reversible and requires excess reactant and/or removal of water to drive it to |