labcorp antibody test covid fda
If you request a test through your doctor, there is no upfront cost.
If you request a test through our online process, Labcorp will bill the cost test directly to your health plan if you are insured.
While the test itself has no upfront costs if you are insured, there is a $6 non-refundable service fee to PWNHealth.
What is FDA serology test for COVID-19?
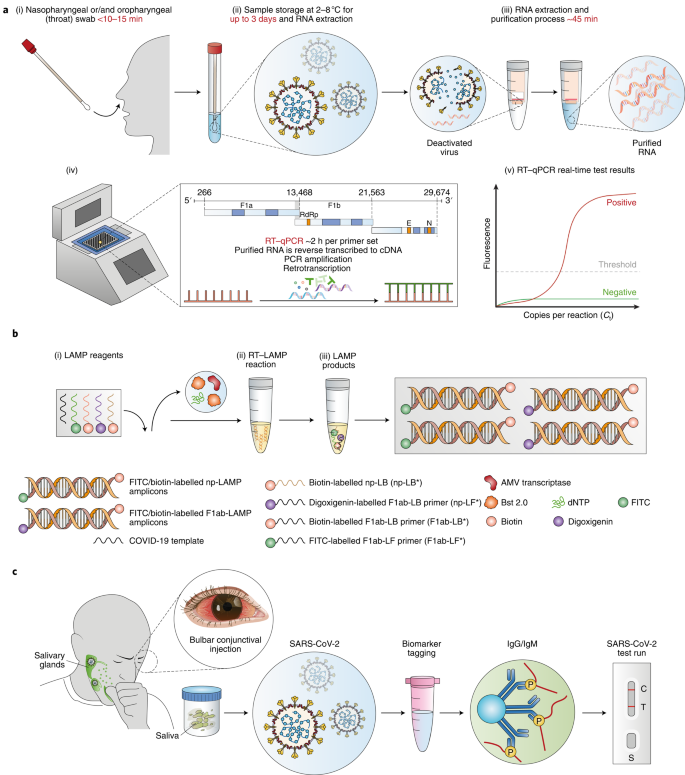
SARS-CoV-2 antibody tests can help identify people who may have been infected with the SARS-CoV-2 virus or have recovered from COVID-19.
Antibody tests should not be used to tell you if you have an active COVID-19 infection.
What is the code for COVID-19 antibody test at labcorp?
164068: SARS-CoV-2 Antibodies, Nucleocapsid Labcorp.
Can antibody test detect COVID?
Antibody testing is usually done to see whether you had COVID-19 in the past and now have antibodies against the virus that causes COVID-19 .
A test to diagnose COVID-19 determines if you currently have the virus that causes COVID-19 .
|
SARS-CoV-2 Antibody Testing Options
What does Labcorp offer for COVID-19 antibody testing? On May 19 2021 |
|
Labcorps COVID-19 RT-PCR Test EUA Summary
21 juin 2022 §263a and meet the requirements to perform high complexity tests |
|
Labcorp VirSeq SARS-CoV-2 NGS Test - FDA
Laboratory Corporation of America (Labcorp). June 10 2022 Labcorp's COVID-19 RT-PCR Test or Labcorp SARS- ... antibody products). |
|
EUA Adaptive tdetect Emergency Use Authorization
2 sept. 2021 The T-Detect COVID Test will be performed at laboratories designated by ... genomic DNA (gDNA) isolated from venous whole blood using ... |
|
Labcorp Seasonal Respiratory Virus RT-PCR Test - EUA Summary
17 mai 2022 §263a and meet the requirements to perform high complexity tests |
|
COVID-19 RT-PCR Test - Letter of Authorization
21 juin 2022 Home Collection Kit or Labcorp COVID-19 Test At Home ... 2020 letter has been revised and reissued by FDA on April 20 |
|
RESOURCE GUIDE - COVID-19 Antibody Testing in a Pandemic
Accessibility & Protocol: The COVID-19 antibody test can be obtained through labcorp.com as well as through a healthcare provider. To make an appointment or get |
|
Labcorp Seasonal Respiratory Virus RT-PCR DTC Test - FDA
16 mai 2022 Anterior nasal swab specimens collected with the Pixel by Labcorp COVID-19+Flu+RSV Test Home. Collection Kit are authorized to be tested with ... |
|
Antibody (Serology) Tests for COVID-19: a Case Study
12 mai 2021 tests without submitting validation data to the FDA. FDA guidance grew more strin- ... LabCorp COVID-19 antibody tests available nationwide. |
|
COVID-19 RT-PCR Test - Patient Fact Sheet
FACT SHEET FOR PATIENTS. Laboratory Corporation of America (“Labcorp”). COVID-19 RT-PCR Test. Updated: June 21 2022. Coronavirus. Disease 2019. (COVID-19). |
|
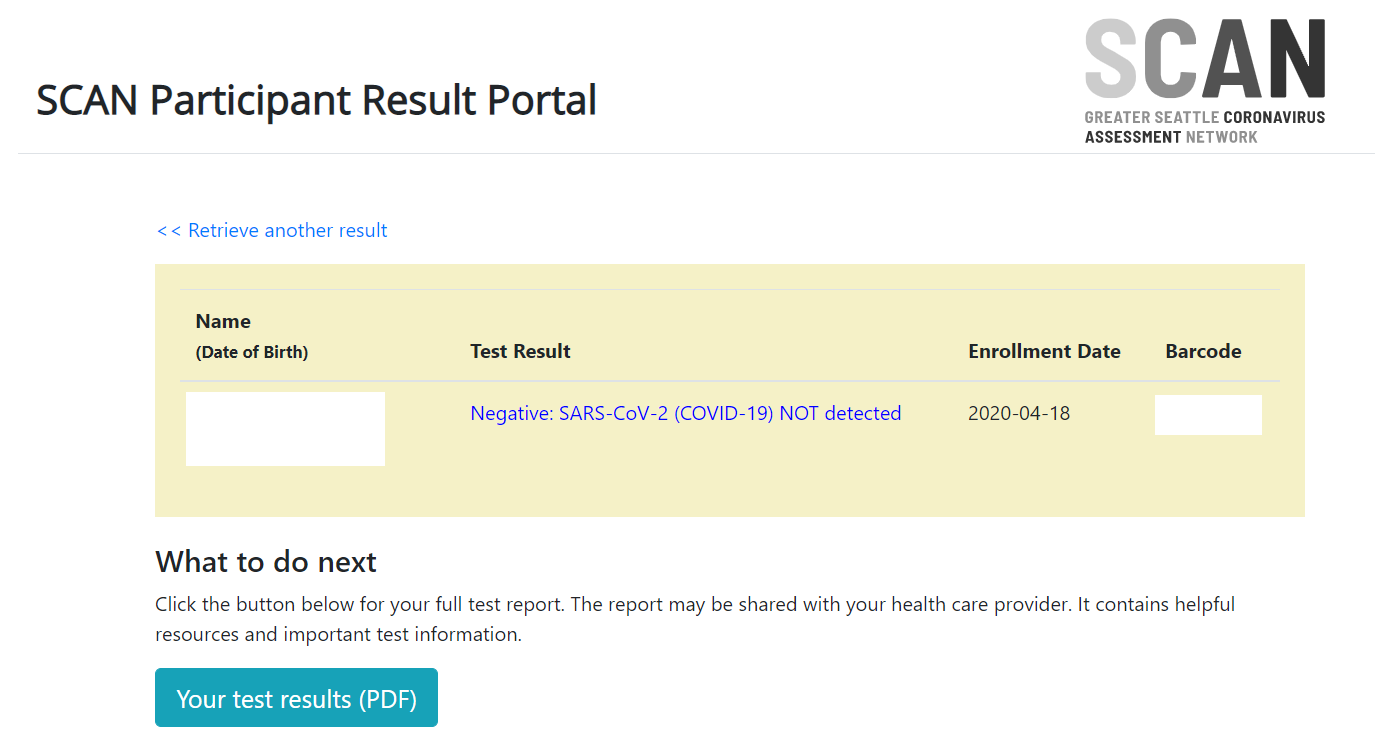
Antibody Test Overview Negative Antibody Results FDA - Labcorp
Your results suggest that you were not exposed to SARS-CoV-2 (COVID-19) or have not yet developed antibodies* Although not having antibodies suggests |
|
Antibody Test Overview Positive Antibody Results FDA - Labcorp
Your results suggest that you've been exposed to SARS-CoV-2 (COVID-19)* Antibodies typically suggest protective immunity from further infection However |
|
Covid-19 Antibody Testing Guide - CSUSM
significance of COVID-19 antibody testing, which is up to date as of the time of publishing Labcorp, Quest Diagnostics, Kaiser Permanente • Accessibility Due to the severity of the COVID-19 pandemic, the FDA has authorized some |
|
SARS-COV-2 ANTIBODY TESTING OPTIONS - Emergency One
LabCorp Helping you provide better patient care SARS-COV-2 ANTIBODY Qualitative antibody testing can provide information on SARS-CoV-2 immune of antibody to SARS-CoV-2, the virus that causes COVID-19, and can help to Test Performance Data, summarized by the FDA: https://www fda gov/medical- |
|
Labcorp - Optum
Labcorp was among the first to offer testing for COVID-19, and did so only five days after through the FDA to produce the same high-quality result SARS- CoV-2 antibody tests are available to be performed by Labcorp's nearly 13,000 |