labcorp antibody test covid fda approval
Is a PCR test the same as an antibody test?
There are three types of tests available for COVID-19: polymerase chain reaction (PCR), antigen, and antibody (serology) testing.
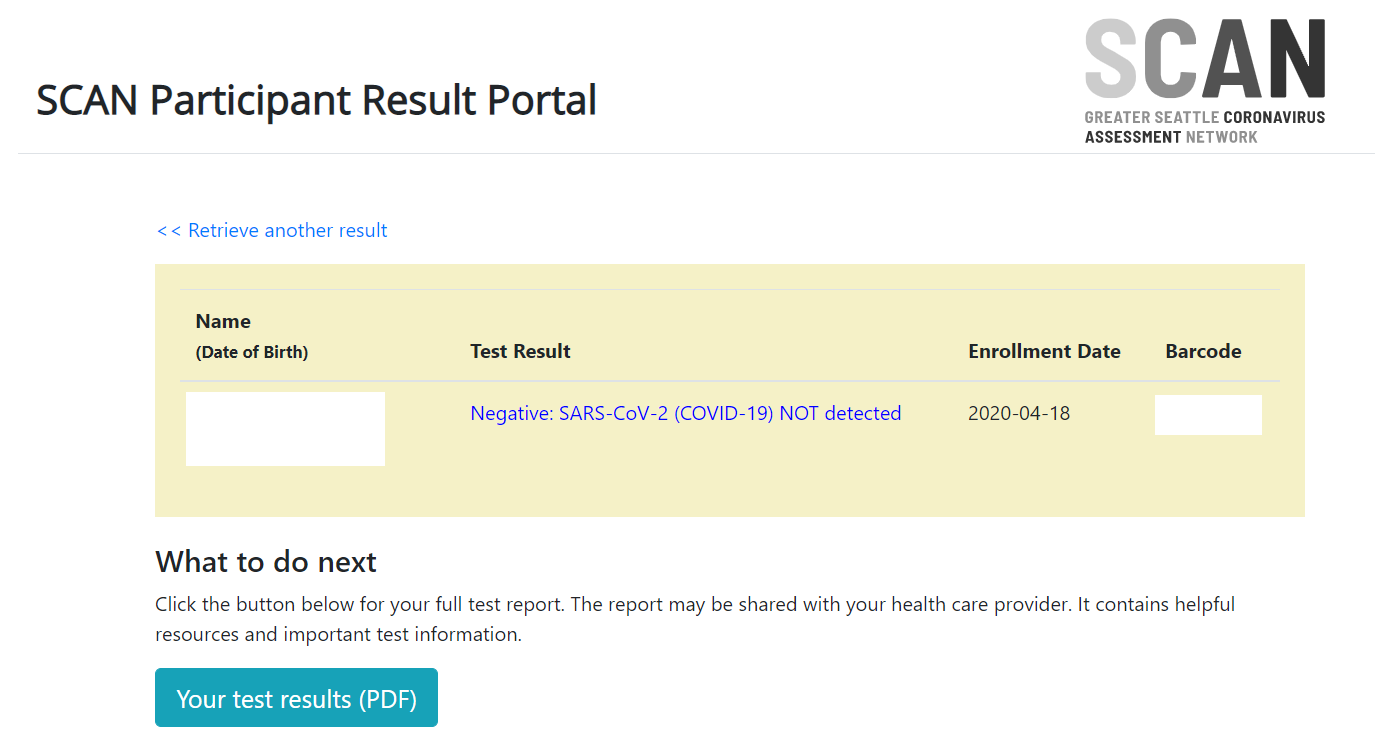
PCR and antigen tests detect whether a person is currently infected, and serology detects whether a person had an infection in the past.Can you do an antibody test for COVID?
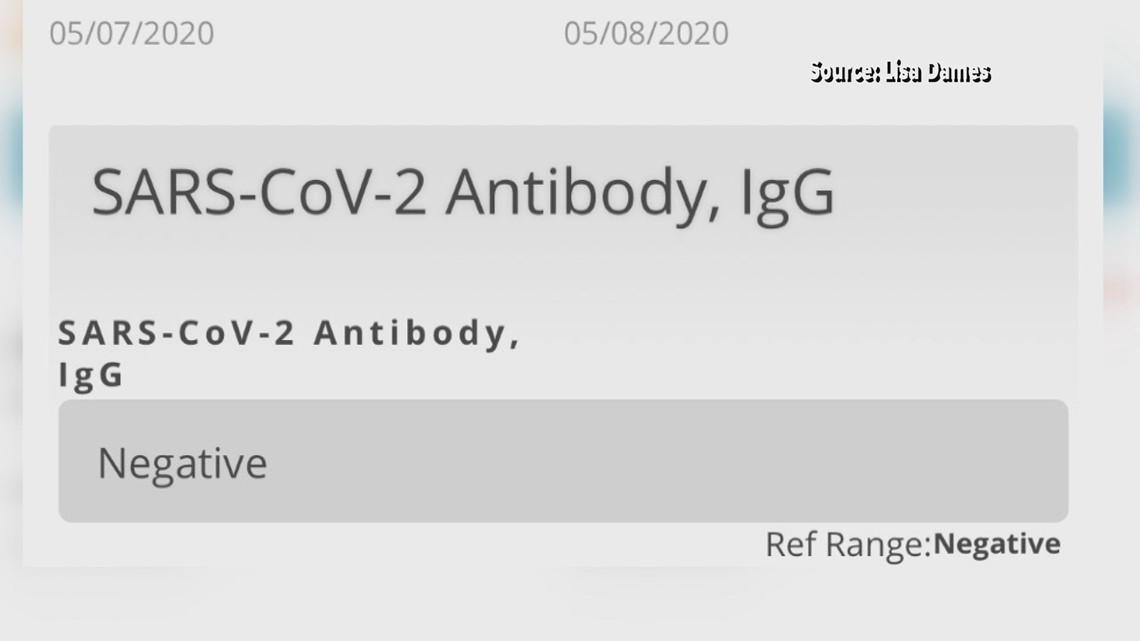
SARS-CoV-2 antibody tests can help identify people who may have been infected with the SARS-CoV-2 virus or have recovered from COVID-19.
Antibody tests should not be used to tell you if you have an active COVID-19 infection.
Scientists continue to learn more about COVID-19 and COVID-19 immunity.Risks.
Results of COVID-19 antibody tests may not always be accurate.
Results may not be accurate if the test was done too soon after infection or the test quality is uncertain.
At the start of the pandemic, there was a rush to get antibody tests on the market.
|
COVID-19 RT-PCR Test - Letter of Authorization
Jun 21 2022 Such records must be made available to FDA for inspection upon request. Labcorp (You). G. Your product must comply with the following labeling ... |
|
EUA Adaptive tdetect Emergency Use Authorization
Sep 2 2021 The T-Detect COVID Test will be performed at laboratories designated by ... genomic DNA (gDNA) isolated from venous whole blood using ... |
|
COVID-19 RT-PCR Test - Healthcare Provider Fact Sheet
Jun 21 2022 consistent with the Emergency Use Authorization (EUA). ... COVID-19 Test Home Collection Kit or Labcorp. COVID-19 Test At Home Collection ... |
|
Labcorp VirSeq SARS-CoV-2 NGS Test - FDA
Jun 10 2022 Laboratory Corporation of America (Labcorp) ... Labcorp's COVID-19 RT-PCR Test or Labcorp SARS- ... A cleared or approved test should be. |
|
COVID-19 RT-PCR Test - Patient Fact Sheet
Jun 21 2022 This test is not yet approved or cleared by the. United States FDA |
|
Improved Syphilis Reverse Screening Cascade
tests detect antibodies to specific antigenic components of Treponema pallidum.6 These antibodies ... Treponemal tests that are currently FDA-approved. |
|
COVID-19 ANTIBODY TEST NOW DIRECTLY AVAILABLE
May 5 2020 The COVID-19 IgG antibody blood test detects the presence of antibodies to the virus and ... This test has not been FDA cleared or approved. |
|
Labcorp Seasonal Respiratory Virus RT-PCR Test - EUA Summary
May 17 2022 Labcorp COVID-19+Flu+RSV Test Home Collection Kit ... For use under Emergency Use Authorization (EUA) only. For prescription use only. |
|
Standard Roche Letter Template
May 19 2020 Roche highly accurate antibody test for COVID-19 goes live at more than ... antibody test received Emergency Use Authorization from the FDA. |
|
RESOURCE GUIDE - COVID-19 Antibody Testing in a Pandemic
COVID-19 ANTIBODY TESTING. What is Antibody Testing? EMERGENCY USE AUTHORIZATION (EUA). FDA approval can take years for in vitro diagnostic or IVD. |
|
SARS-COV-2 ANTIBODY TESTING OPTIONS - Labcorp
Qualitative antibody testing can provide information on SARS-CoV-2 immune tests determine the presence of antibody to SARS-CoV-2, the virus that causes COVID-19, and can help to These tests have not been FDA cleared or approved |
|
Covid-19 Antibody Testing Guide - CSUSM
significance of COVID-19 antibody testing, which is up to Labcorp, Quest Diagnostics, Kaiser FDA approval can take years for in vitro diagnostic or IVD |
|
Screening and testing - NC Chamber
The COVID-19 Serological Antibody test determines the presence of LabCorp's COVID-19 PCR test has not been FDA cleared or approved, has been |
|
COVID-19
We will talk more about testing (including antibody testing) For serology, we are referring patients for the Labcorp Roche COVID-19 antibodies testing This specific antibody test has approval for FDA Emergency Use and has a 99 8 |