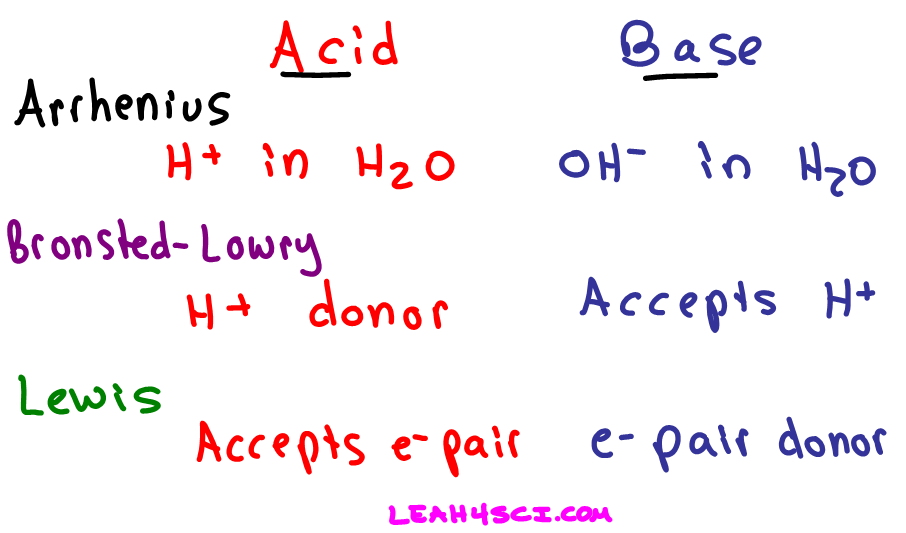lewis acids and bases practice problems
|
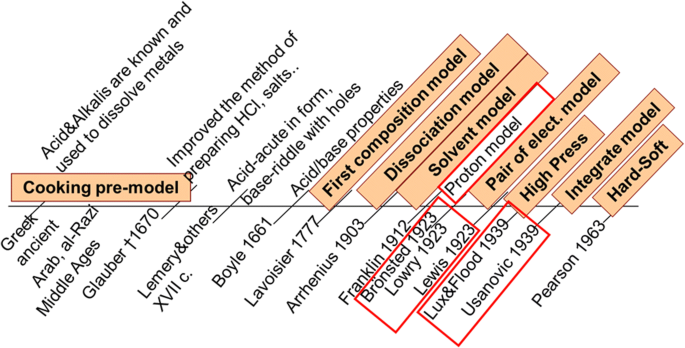
Chem 1B Practice problems for Exam 2
Chem 1B Practice problems for Exam 2 Once again this is not a “practice test Substances that can behave as either Lewis acids or bases are called : a |
Is Fe3+ a Lewis acid or base?
In this reaction the ferric ion (Fe3+) is acting as a Lewis Acid and the oxalate anion (C2O42-) as a Lewis Base.
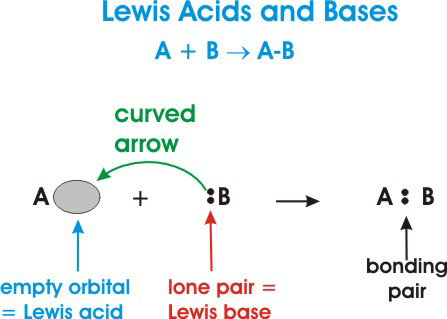
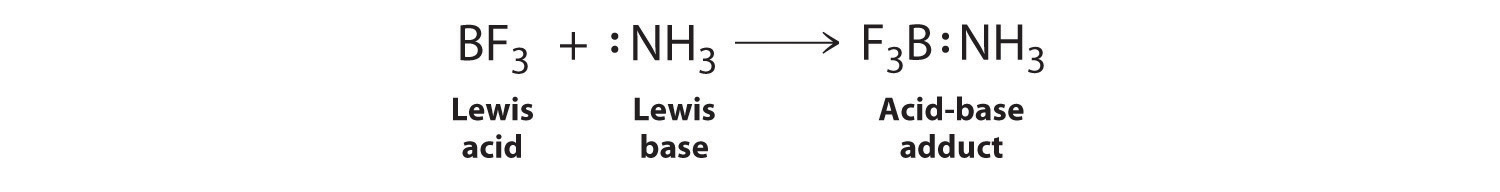
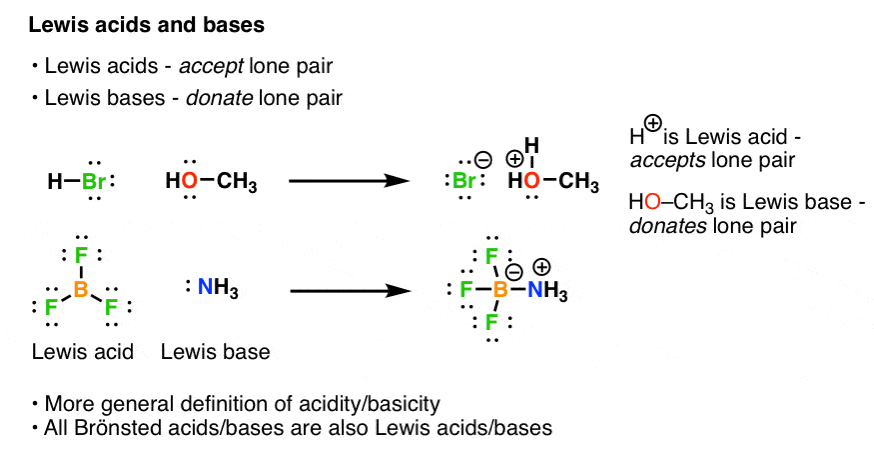
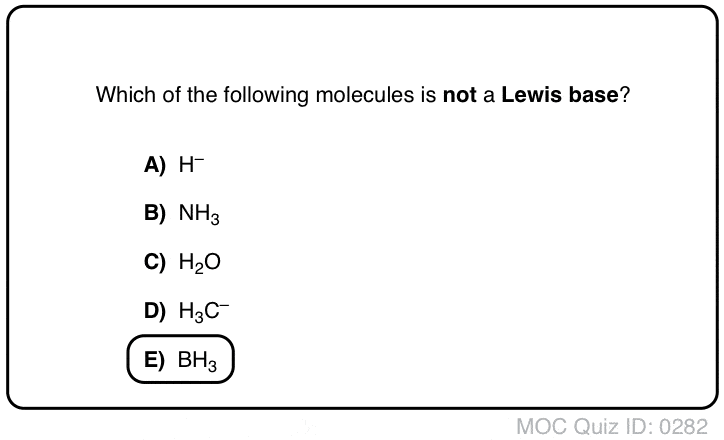
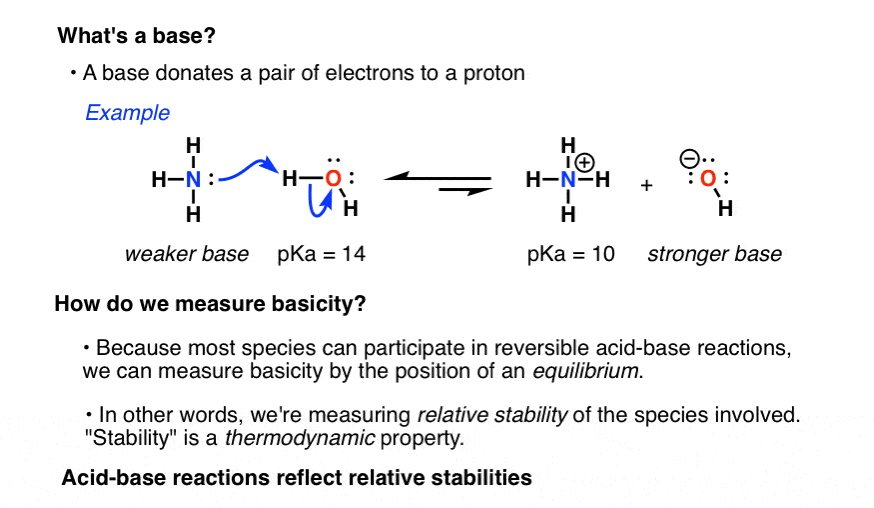
A Lewis Acid is an electron pair acceptor and a Lewis Base is an electron pair donor.
The oxalate anion is donating electrons to the vacant 3d and 4s orbitals on iron.A Lewis acid is any species (molecule or ion) that can accept a pair of electrons, and a Lewis base is any species (molecule or ion) that can donate a pair of electrons.
A Lewis acid-base reaction occurs when a base donates a pair of electrons to an acid.
What is an example of a Lewis acid-base reaction?
The interaction between a magnesium cation (Mg+2) and a carbonyl oxygen is a common example of a Lewis acid-base reaction.
The carbonyl oxygen (the Lewis base) donates a pair of electrons to the magnesium cation (the Lewis acid).
How do you find the Lewis acids and bases?
A Lewis Acid will have vacant orbitals.
All cations are also Lewis Acids since they accept electrons.
Lewis Base: A compound that is an electron-pair donor, or a compound that has a pair of electrons that are able to bond with an electron-acceptor.
A Lewis Base will have lone-pair electrons.
|
Chem 1B Practice problems for Exam 2 Once again this is not a
The chemical equations below show the reaction of Al(OH)3 as a Lewis acid and as Substances that can behave as either Lewis acids or bases are called :. |
|
Test2 ch17a Acid-Base Practice Problems
Acid-Base Chemistry. Extra Practice Problems. General Types/Groups of problems: Conceptual Questions. Acids Bases |
|
Practice Problems -? Answers 1. Draw the structure of the conjugate
2. Draw the structure of the conjugate base of each of the following ions or compounds. 3. Classify each compound as a Lewis acid or Lewis base in the |
|
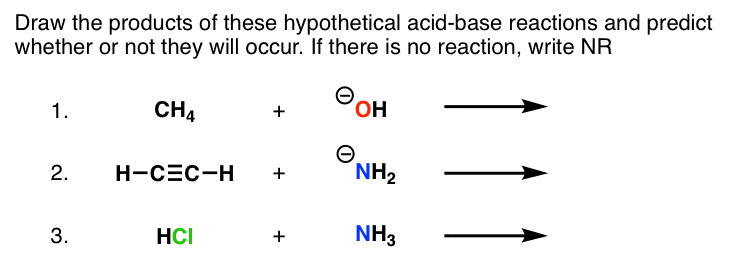
1. For each of the species below identify the most acidic proton and
PRACTICE PROBLEMS FOR BRONSTED-LOWRY ACID-BASE CHEMISTRY corresponding conjugate acid. You might want to draw detailed Lewis formulas in some. |
|
POLYPROTIC ACIDS AND BASES: Very important!
Sample Problem 18.15. Identify the Lewis acids and bases: a) H+. + OH-. ? H2O acid base adduct (OH |
|
18 Acids and Bases
Section 18.1 Introduction to Acids and Bases pages 634–643. Practice Problems pages 635–640. 1. Write balanced equations for reactions between. |
|
OChem1 Practice Problems
Draw Lewis formula structures for molecules with the following molecular formulae. Klein Chapter 3 : Acids and Bases Worksheet. Concepts. |
|
Organic Chemistry Jasperse Acid-Base Practice Problems
Acid-Base Practice Problems. A. Identify each chemical as either an “acid” or a “base” in the following reactions and identify “conjugate” relationships. |
|
Tentative Daily Schedule - CHEM 125 Fall 2012 Atomic Structure
30 ago 2018 Suggested Practice Problems: ... o Practice Packing Problems: Workbook 66-71 ... o Passport: Lewis Acid/Base |
|
A b c mL of Base added
Be able to do the calculations above for a weak base + strong acid problem. For example what is the pH at the equivalence point for the titration of a 25.0mL |
|
Chem 1B Practice problems for Exam 2 Once again, this is not a
The chemical equations below show the reaction of Al(OH)3 as a Lewis acid and as a Substances that can behave as either Lewis acids or bases are called : |
|
Acid-Base Practice Problems
Acid-Base Practice Problems A Identify each chemical as either an “acid” or a “ base” in the following reactions, and identify “conjugate” relationships |
|
Practice Problems -‐ Answers 1 Draw the structure of the conjugate
Draw the structure of the conjugate base of each of the following ions or compounds 3 Classify each compound as a Lewis acid or Lewis base in the reactants |
|
Acid Base Practice Test - Houston ISD
Acid Base Practice Test ____ 1 Acids Which of the following chemical reactions represents an acid-base reaction? a HBr + KOH Lewis acid d traditional |
|
Chapter 11 – Acids and Bases – Practice Problems Section 111
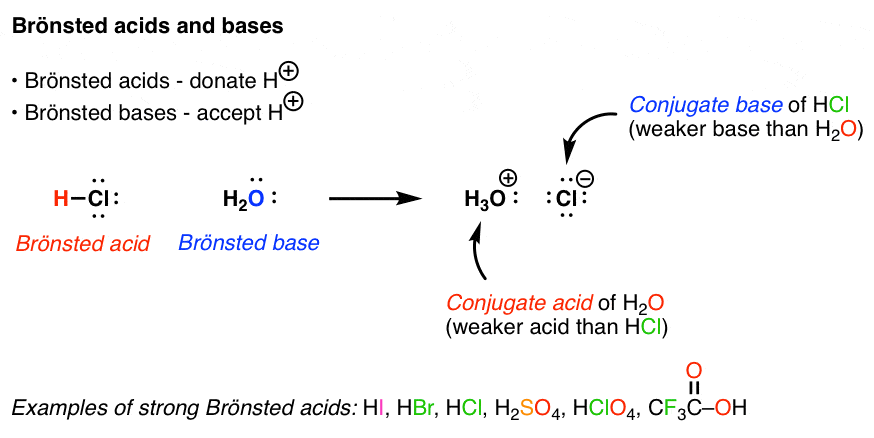
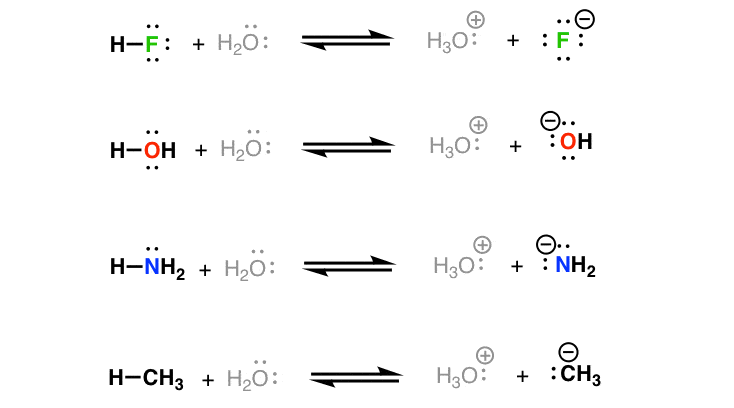
Identifying Conjugate Acid-Base Pairs • According to Bronsted-Lowry theory, a conjugate acid-base pair consists of molecules or ions related by the loss of one |
|
18 Acids and Bases
Write balanced equations for reactions between the following Explain why many Lewis acids and bases are Write ionization equations and acid ionization |
|
Acids Lewis Acids and Bases Lewis Acids Lewis acids: H+ Cu2+
Problem: Some acids do not contain protons Example: Al3+ (aq) = ≈ pH 3 Deodorants and acid loving plant foods contain aluminum salts Lewis Acids and |
|
Questions ACID-BASE THEORIES - Chemguide
Use the following equations to help you to explain what is meant by the statement that “water is amphoteric” 5 a) Define the terms Lewis acid and Lewis base b) |
|
Practice Problems For Bronsted Lowry Acid Base Chemistry
test acids and bases types of reactions siyavula acid base practice problems what is an example of a bronsted lowry acids practice a p chemistry practice test |
|
Chapter 12 – Acid-Base Chemistry - WebAssign
Curved Arrows in a Mechanism–an Example A video or simulation is available online The Lewis acid-base reaction between SO3 and H2O to form H2SO4 is |