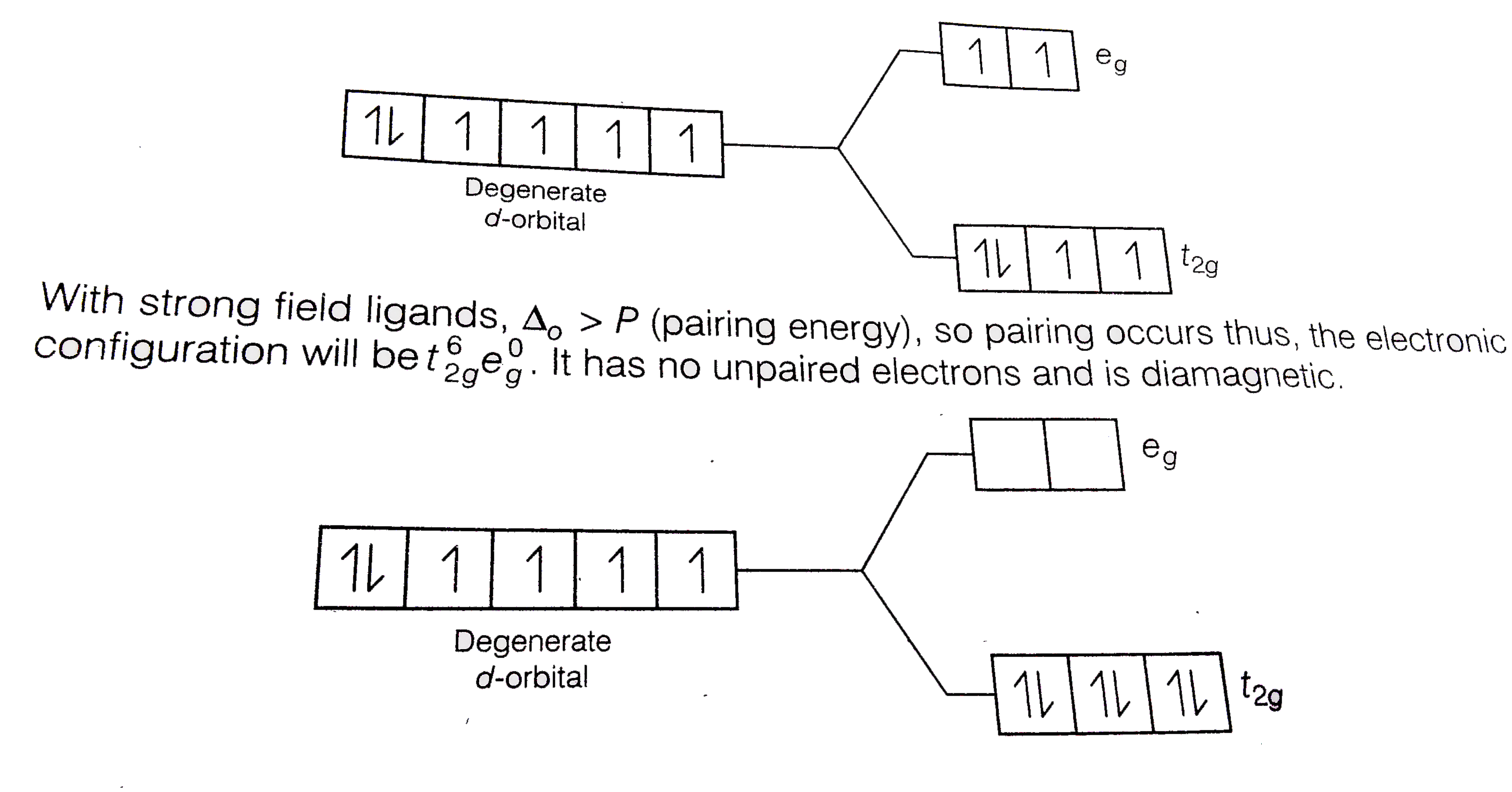ligand field theory pdf
|
Inverted Ligand Field Theory
8 jui 2018 · Inverted ligand field theory suggests an alternative explanation for the spectroscopic properties and chemical reactivity of many high |
|
Introduction to ligand field theory
results of the crystal field theory are also valid in the ligand field theory However since the former theory in some ways is the easier to under stand |
|
Ligand Field Theory CDN
Ligand Field Theory is: ‣ A semi-empirical theory that applies to a CLASS of substances (transition metal complexes) |
|
Crystal (ligand) field theory
Crystal (ligand) field theory • Sphere symmetry • jj-coupling LS- coupling Crystal (ligand) field theory • Orgel's diagrams • Tanabe-Sugano diagrams |
|
Ligand Field Theory
Ligand Field Theory is: ‣ A semi-empirical theory that applies to a CLASS of substances (transition metal complexes) |
|
Ligand Field Theory
A An understanding of how d orbitals are affected by bringing “n” ligands around the metal center MLn n = 6 Octahedral |
|
Ligand Field Theory
23 nov 2015 · Ligand Field Theory In LFT we use metal valence orbitals and ligand frontier orbitals to make metal–ligand molecular orbitals |
|
LIGAND FIELD THEORY
One of the basic ways of applying MO concepts to coordination chemistry is in Ligand Field Theory Ligand field theory looks at the effect of donor atoms on |
|
Coordination Chemistry II: Ligand Field Theory Continued
The nature of the metal ligand π interaction is dependent on the type of ligand • π-donor ligands are ligands with one or more lone pairs of |
What is ligand field theory fundamentals?
Ligand Field Theory can be considered an extension of Crystal Field Theory such that all levels of covalent interactions can be incorporated into the model.
Treatment of the bonding in LFT is generally done using Molecular Orbital Theory.What is the CFT theory?
Crystal field theory (CFT) describes the breaking of orbital degeneracy in transition metal complexes due to the presence of ligands.
CFT qualitatively describes the strength of the metal-ligand bonds.
Based on the strength of the metal-ligand bonds, the energy of the system is altered.Weak field ligands are ones that produce small splittings between the d orbitals and form high spin complexes.
Examples of weak field ligands include the halogens, OH- and H2O.
From a ligand field perspective these ions or molecules all have filled orbitals which can π bond with the metal d orbitals.
What is the ligand field theory?
ligand field theory, in chemistry, one of several theories that describe the electronic structure of coordination or complex compounds, notably transition metal complexes, which consist of a central metal atom surrounded by a group of electron-rich atoms or molecules called ligands.
|
LIGAND FIELD THEORY
One of the basic ways of applying MO concepts to coordination chemistry is in Ligand Field Theory. Ligand field theory looks at the effect of donor atoms on. |
|
Coordination Chemistry II: Ligand Field Theory Continued
Ligand Field Theory Continued. Chapter 10. Wednesday November 25 |
| Introduction to ligand field theory. |
|
Crystal (ligand) field theory
Crystal (ligand) field theory. Literature. • C.E. Housecroft A.G. Sharpe: Inorganic chemistry. Page 2. 2. Title page. Crystal (ligand) field theory. • Sphere |
|
Ligand Field Theory
Ligand Field Theory is: ‣ A semi-empirical theory that applies to a CLASS of substances (transition metal complexes). |
|
Ligand Field Theory
Ligand Field Theory is: ‣ A semi-empirical theory that applies to a CLASS of substances (transition metal complexes). |
|
Ligand Field Theory
Ligand Field Theory is: ‣ A semi-empirical theory that applies to a CLASS of substances (transition metal complexes). |
|
Coordination Compounds
(vi) It does not distinguish between weak and strong ligands. The crystal field theory (CFT) is an electrostatic model which considers the metal-ligand bond to |
|
Eurasian Journal of Science & Engineering ISSN 24
٠١/٠٩/٢٠١٧ Spectra were measured from 1100 nm to 220 nm at ambient temperature. 3. Theory. The ligand field splitting parameter differs methodically with ... |
|
BSc Chemistry
The Ligand Field Theory explains the covalent bonding of metal and ligands with the help of molecular orbital (MO) diagram. 3. Delocalized approach To Bonding: |
|
Coordination Chemistry II: Ligand Field Theory Continued
25-Nov-2015 Only the d4 through d7 cases can be either high-spin or low spin. ? < ?. ? > ?. Weak-field ligands: - Small ? High spin complexes. |
|
Ligand Field Theory - BRIAN N. FIGGIS
6.2.2 Ligand Field Theory as Extended CFT. 6.2.2.1 The identification of A in qualitative MO theory. 6.2.2.2 The concept of an 'effective' d-orbital set. |
|
Introduction to ligand field theory.
however that the ligand field theory offers only a model of nature |
|
Crystal (ligand) field theory
Crystal (ligand) field theory. Literature Crystal field. Schrödinger equation ... Splitting of states for electron configuration d2 – sphere symmetry. |
|
Ligand Field Theory
Octahedral Case: Ligands along x y |
|
Ligand Field Theory
Ligand Field Theory is: ? A semi-empirical theory that applies to a CLASS of substances (transition metal complexes). |
|
B.Sc. III YEAR INORGANIC CHEMISTRY-III
Unit- 2 Metal Ligand bonding in transition metal complexes 2.8 Ligand field theory and molecular orbital theory. 2.9 Summary. 2.10 Terminal questions. |
|
Coordination Compounds
(vi) It does not distinguish between weak and strong ligands. The crystal field theory (CFT) is an electrostatic model which considers the metal-ligand bond to |
|
Ligand Field Theory: An ever-modern theory
Ligand Field Theory: An ever-modern theory. To cite this article: Claude A Daul 2013 J. Phys.: Conf. Ser. 428 012023. View the article online for updates |
|
?-Bonding and Molecular Orbital Theory
The combination of ligand orbitals overlapping with dxz-orbital of the metal center. Molecular orbitals and the effect of ?-bonding on crystal field splitting |
|
LIGAND FIELD THEORY
chemistry is in Ligand Field Theory Ligand field theory looks at the effect of donor atoms on the energy of d orbitals in the metal complex There are two ways in |
|
LIGAND_FIELD_THEORYpdf
A ligand field theory considers the interaction between a central transition metal atom and its ligands, in particular the part arising from the immediate donor atoms |
|
Introduction to ligand field theory
however, that the ligand field theory offers only a model of nature, with all between the central ion and the ligands in terms of some electronic theory? |
|
Ligand Field Theory - CDN
Ligand Field Theory is: ‣ A semi-empirical theory that applies to a CLASS of substances (transition metal complexes) ‣ A LANGUAGE in which a vast number |
|
Coordination Chemistry II: Ligand Field Theory - UCI Chemistry
25 nov 2015 · Metal ions typically have some valence electrons that can be accommodated in the metal d orbitals • d0 ions – Ti4+, Zr4+, V5+, Ta5+, Cr6+, |
|
Ligand field theory
repulsion of ligands and electrons in d-orbitals along the axis => splitting of the five degenerated energy levels into two different levels, where the lower one |
|
Ligand Field Theory - IOPscience
Ligand Field Theory: An ever-modern theory To cite this article: theory is the fact that the more accurate molecular orbital theory does generally over-estimate |
|
Crystal (ligand) field theory
1 Title page Crystal (ligand) field theory Literature Crystal field Schrödinger Splitting of states for electron configuration d2 – sphere symmetry 1 Hund 2 |
|
Crystal Field Theory
The interactions between the metal ion and the ligands are purely electrostatic ( ionic) •The ligands are regarded as point charges •If the ligand is negatively |











![3 Ligand Field Theory-ippt [en5ke2y2j5no] 3 Ligand Field Theory-ippt [en5ke2y2j5no]](https://demo.vdocuments.mx/img/378x509/reader024/reader/2021010605/575094be1a28abbf6bbbb479/r-1.jpg)